Imaging & Artificial Intelligence for Assessing Quality of a Biomanufactured Retinal Pigment Epithelium
Background: We have developed methods to assess quality of manufactured retinal pigment epithelium (RPE). The work was a collaboration with Kapil Bharti (National Eye Institute, NEI/NIH) who is advancing a tissue engineering therapy to treat age-related macular degeneration (AMD). AMD is the leading cause of vision loss in adults and one its major causes is the degeneration of a supportive layer of cells called the RPE. A tissue-engineered RPE is being developed to repair the degenerative RPE tissue in the patient. This strategy circumvents the immune system by using the patient's own cells to make healthy RPE through "induced-pluripotent stem cell" technology (iPSC). A clinical trial is being planned to test this approach in humans. It is important to conduct a portion of clinical trials in the public domain so that a "behind the scenes" view of the development and manufacturing process can benefit other cell therapy stakeholders. This is key since the cell therapy industry groups, Alliance for Regenerative Medicine (ARM) and Regenerative Medicine Foundation (RMF) have identified a lack of reliable methods for characterizing cell-based products as possibly the single greatest challenge for the field (ARM Report, 2014; RMF Report, 2015).
Imaging & Artificial Intelligence (AI) to Assess Tissue Quality: We have collaborated with Peter Bajcsy (NIST/ITL) and Kapil Bharti (NIH/NEI) to develop an approach for non-invasively assessing quality of clinical-grade tissue-engineered human retinal pigment epithelium (RPE). The method uses quantitative brightfield absorbance imaging (QBAM) and artificial intelligence (AI). Absorbance imaging was implemented in Bharti’s GMP facility where we noninvasively collected brightfield images of the clinical-grade constructs. Constructs were imaged using absorbance imaging and traditional RPE potency test data were collected. The RPE potency tests were measurements of polarized secretion of vascular endothelial growth factor (VEGF) and measurements of trans-epithelial resistance (TER). Peter Bajcsy's team directed the image processing, image analysis and implementation of AI methods. AI algorithms were trained to predict potency (VEGF and TER) from the absorbance images. The trained AI algorithm made correct predictions for 35 of 36 test image data sets.
"This work demonstrates how a garden variety microscope, if used carefully, can make a precise, reproducible measurement of tissue quality."

Absorbance Measurements Can Be Made on an Absolute Scale: Absorbance imaging works for RPE because they express melanin and become pigmented. Pigmentation can be measured by absorbance imaging, which is advantageous since absorbance measurements can be made on an absolute scale. To generate absorbance images, brightfield image captures of the constructs are divided by image captures of background (culture dish without constructs but with the light source on). This is a ratiometric approach which enhances comparability. Further, optical filters are widely available and can be used as reference materials to benchmark absorbance measurements. These attributes enable direct comparison of absorbance measurements made in different labs, with different imaging systems and by different operators, which makes this attractive for a biomanufacturing application where quantitative, comparable data are required for establishing manufactured tissue quality.
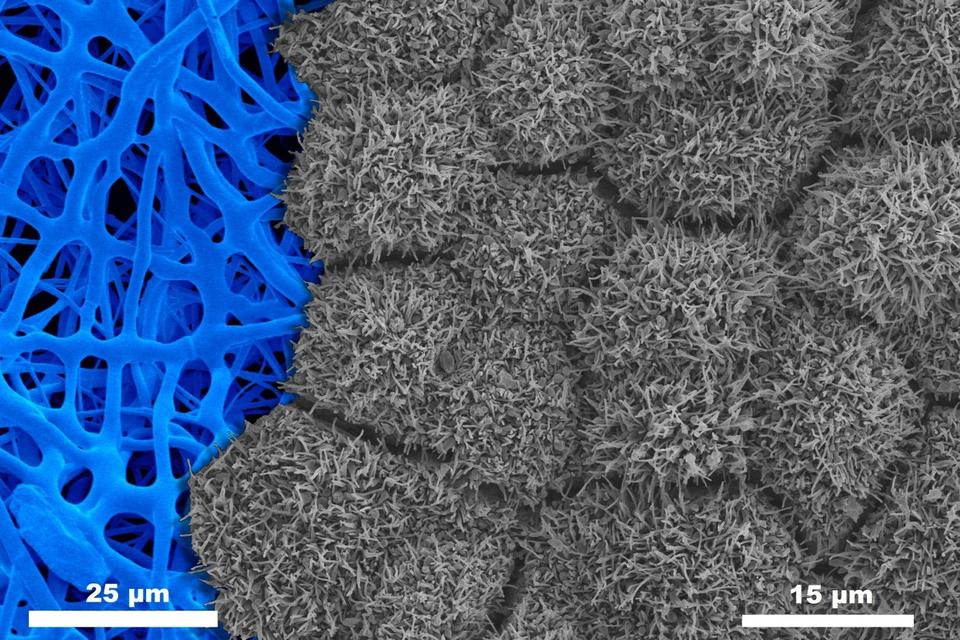
Nanofiber Characterization: Since tissue-engineered RPE are delicate and challenging to handle, they are being grown on fibrous scaffolds to facilitate their handling and implantation into the eye. We have developed an automated, open-source, validated algorithm for assessing nanofiber scaffold structure called DiameterJ.
Publications:
- Schaub NJ, Hotaling NA, Manescu P, Padi S, Wan Q, Sharma R, George A, Chalfoun J, Simon M, Ouladi M, Simon Jr CG, Bajcsy P, Bharti K (2020) Deep learning predicts function of live retinal pigment epithelium from quantitative microscopy. Journal of Clinical Investigation 130, 1010-1023. https://doi.org/10.1172/JCI131187
- NIST Press Release
- NIH Press Release
- Commentary at Journal of Clinical Investigation
- Potency & Absorbance Imaging Data: The data from this work was made publicly available so that others may use it to develop improved algorithms for predicting tissue potency (VEGF & TER) from absorbance images.
- Plugins
- Hotaling NA, Khristov V, Wan Q, Simon Jr CG, Bharti K. Nanofiber scaffolds for use in development of a tissue engineered retinal pigment epithelium. Journal of Ocular Pharmacology & Therapeutics 32, 272-285. https://doi.org/10.1089/jop.2015.0157
Slides: RPE slides.pdf
Contributors: Nicholas Schaub, Nathan Hotaling, Peter Bajcsy, Kapil Bharti, Michael Halter, Carl Simon

