SI Units

SI Model
Familiarity with the elements and structure of the International System of Units (SI), commonly known as the metric system, will prepare students to successfully make measurements in science, technology, engineering and mathematics (STEM) applications. The major SI elements are illustrated in this model.

Defining Constants
The SI rests on a foundation of seven (7) defining constants: the cesium hyperfine splitting frequency, the speed of light in vacuum, the Planck constant, the elementary charge (i.e. the charge on a proton), the Boltzmann constant, the Avogadro constant, and the luminous efficacy of a specified monochromatic source. NIST provides values and a searchable bibliography for the fundamental physical constants.
Base and Derived Units
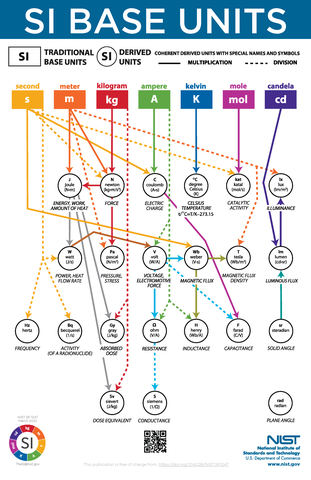
Definitions of all seven (7) SI base units are expressed using an explicit-constant formulation and experimentally realized using a specific mises en pratique (practical technique).
The SI is made up of 7 base units that define the 22 derived units with special names and symbols, which are illustrated in NIST SP 1247, SI Base Units Relationship Poster. The SI plays an essential role in international commerce and is commonly used in scientific and technological research and development. Learn more about the SI in NIST SP 330 and SP 811.
The seven SI base units, which are comprised of:
- Length - meter (m)
- Time - second (s)
- Amount of substance - mole (mol)
- Electric current - ampere (A)
- Temperature - kelvin (K)
- Luminous intensity - candela (cd)
- Mass - kilogram (kg)
Together the base units and derived units with special names (twenty-nine units) form the core set of SI units. All other SI units are combinations of some of these twenty-nine units. Any of the base units and derived units with special names can be constructed directly from the seven defining constants. Derived units with generic names reflect their mathematical derivation, such as area (m2), volume (m3), velocity (m/s), and acceleration (m/s2).

Prefixes
Twenty-four prefixes ranging from 1030 to 10−30, are currently recognized for use. Values of quantities are expressed using Arabic symbols for numbers paired with a unit symbol, often a prefix symbol that modifies unit magnitude.
Writing with the SI
A benefit of the International System of Units (SI) is that written technical information is effectively communicated, overcoming the variations of language, including spelling and pronunciation. Values of quantities are expressed using Arabic symbols for numbers paired with a unit symbol, often with a prefix symbol that modifies unit magnitude. Learn more about Writing with SI.
Scientists, Technologists, Engineers, and Mathematicians write with the SI. SI writing style is an essential literacy skill used by STEM professionals and the public to interpret, evaluate, synthesize, communicate, and apply measurement information. For STEM professionals, SI skills are necessary to plan investigations, collect and document research data observations, analyze results, clearly communicate, share research, gain new information by interpreting the findings of others, develop technological solutions to improve the quality of life, design and build critical infrastructure, and develop new products for the marketplace. Using SI writing style helps prevent miscommunication, misinterpretation of data, and calculation errors. SI writing style is also important for consumers who will interpret and use quantities to make daily life decisions.

The International System of Units (SI), commonly known as the metric system, is the international standard for measurement. The International Treaty of the Meter was signed in Paris on May 20, 1875 by seventeen countries, including the United States and is now celebrated around the globe as World Metrology Day. NIST provides official U.S. representation in the various international bodies established by the Meter Convention: CGPM - General Conference on Weights and Measures; CIPM - International Committee for Weights and Measures; and BIPM - The International Bureau of Weights and Measures.
EXPLORE THE METRIC PROGRAM
Becoming Familiar with SI | Everyday Estimation | U.S. Metrication | U.S. Metrication FAQs | Prefixes | Metric Kitchen | SI Education and Training | SI Publications | Understanding Metric | Writing with Metric Units | National Metric Week | NEST-R (STEM Registry) | NIST Education Resources
Contacts
-
Metric Program


