Summary
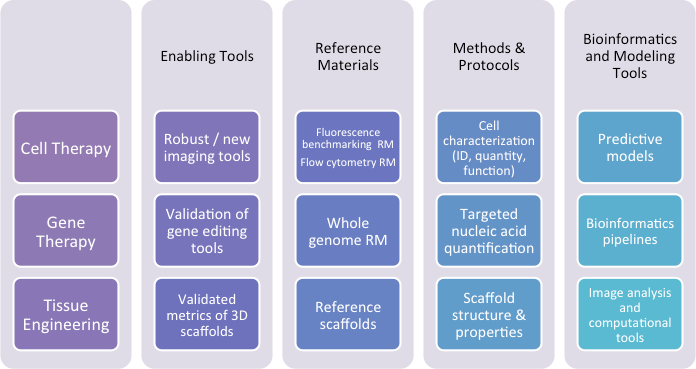
The NIST Regenerative Medicine and Advanced Therapies (RMAT) laboratory programs are focused on developing innovative measurement assurance solutions and technologies to accelerate translation and commercialization of advanced therapy products. Our measurement infrastructure provides enabling tools, methods and protocols, bioinformatics and modeling tools, as well as documentary standards and reference materials.
Our programs are designed to support underpinning measurement challenges for a wide range of product types (cell therapy, gene therapy, and tissue engineered) produced by different manufacturing processes. We enable high confidence measurements via common platforms (e.g., sequencing, flow cytometry, microscopy) and help the community connect those measurements with the quality attributes needed in process development, product characterization, and testing.
Description
Examples of Current Activities:
Cell Counting and viability are key measurements critical for decision making from R&D to manufacturing. We recently developed an approach for evaluating the quality of cell counting measurements through experimental design and statistical analysis that does not require the use of a reference material (reference control) and is agnostic of the cell type and counting method. A two-part ISO standard has been produced as an outcome of this effort. These standards will facilitate method validation and method bridging.
- ISO 20391-1:2018 - Biotechnology -- Cell counting -- Part 1: General guidance on cell counting methods
- ISO 20391-2:2019 Biotechnology — Cell counting — Part 2: Experimental design and statistical analysis to quantify counting method performance
Copy Number Standards: Accurate measurement of copy numbers is important for clinical diagnostics and as a critical quality attribute (CQA) for many advanced therapies and raw/ancillary materials. NIST has deep expertise in providing reference materials with certified copy number values to ensure that the procedures and analytical measurements are working properly. In addition, working with global Metrology Institutes, NIST contributed to the publication of an ISO standard on quantitative qPCR and ddPCR measurements.
Flow Cytometry is widely used in measurement of cells, including expressed proteins, and other cellular attributes and processes. The NIST Quantitative Flow Cytometry Consortium enables the development of comparable data across labs via standardized protocols and reference materials such as fluorescence standards for calibrating beads and lyophilized cell reference materials. A CLSI documentary standard is under development.
Gene Delivery Systems through viral and non-viral vector means provide gene therapies mechanisms to deliver genes of interest into a patient’s cells and cure previously untreatable diseases. There is a need for characterization and quality measurements of gene delivery system particles. We are developing methods to measure particle heterogeneity and provide advanced characterization of particle physical and infectious titers.
Genome Editing products and processes need reliable methods for assuring the results of editing and adding confidence of safety and likely effectiveness. We are working with stakeholders, including through NIST-organized workshops, to identify pre-competitive technology, measurement, and standards needs and solutions. The current program focuses on evaluating assays and informatics tools, and the development of next-generation reference materials. We have established a NIST-led Genome Editing Consortium to develop measurement solutions and standards to advance this technology space.
Live Cell Imaging tools provide spatial information and population heterogeneity on large numbers of individual cells over time. We are producing high quality data obtained using benchmark materials to develop predictive models based on stochastic fluctuations and to provide reference data. These efforts will support methods to validate putative molecular markers for pluripotency and differentiation and for evaluating markers of product potency including CAR-T therapy products. We are also developing a suite of advanced imaging capabilities, including broadband coherence RAMAN spectroscopy (BCARS) imaging, for characterizing cell state and cell-biomaterial interactions.
Rapid Microbial Testing Methods (RMTM) are urgently needed for the advanced therapy industry. NIST has established an RMTM Consortium to convene stakeholders and develop measurement assurance tools and solutions, including reference materials and best practices, to support product safety assessments. NIST is also working with stakeholders to develop an ISO standard to clarify risk assessment methodologies and validation of selected fit-for-purpose assays.

