Summary
The NIST Rapid Microbial Testing Methods (RMTM) Consortium addresses the need for measurements and standards, including reference materials, to increase confidence in the use of rapid testing for microbial contaminants in regenerative medicine and advanced therapy products.
CONSORTIUM LAUNCH
A virtual half-day consortium launch workshop, held on September 17, 2020, included presentations and panel discussions from subject matter experts. The workshop agenda, abstracts, and full recording are available on the workshop website.
MEMBER BENEFITS
- Access to a neutral forum to address pre-competitive needs
- Participation in the development of reference materials, methods and protocols, and interlaboratory studies
- Access to tools developed by the Consortium ahead of public release
- Institutional representation on Consortium steering committee
BECOME A MEMBER
- Complete the Letter of Interest Form
- Participants will sign a Cooperative Research and Development Agreement (CRADA); Federal agencies may join under a Letter Agreement (LA)
- No cost to join the Consortium
Notice of NIST's Rapid Microbial Testing Method Consortium
Description

The safety and quality of advanced therapies, including cell therapy, gene therapy, and tissue engineered products, must be maintained prior to patient administration. The culture-based compendial methods currently used to assess product purity (specifically to ensure absence of microbial contamination) typically take weeks, which is inadequate for patients in urgent need of life-saving therapies. These methods are also incompatible with products that have a limited shelf-life and cannot meet good manufacturing practices required in process control and release testing. Alternative rapid microbial testing methods are needed to ensure fit for purpose safety assessments for this broad class of advanced therapeutics. The NIST RMTM Consortium brings together experts across the regenerative medicine field including stakeholders in industry, academia and government to address this need.
CONSORTIUM GOALS AND ONGOING EFFORTS
- Identify and/or design fit for purpose microbial cell reference materials, and develop methods to characterize cell properties beyond colony forming units.
- Develop guidance on the use of reference materials in the qualification and/or validation of rapid microbial testing methods for advanced therapy products.
- Design interlaboratory studies based on candidate reference materials to evaluate materials and support the development of best practices and standard methods.
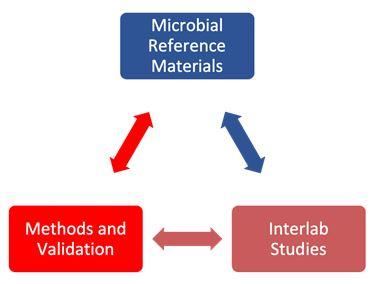
CONSORTIUM ORGANIZATION
The RMTM Consortium Consists of 3 Working Groups
WG01 MISSION: The mission of the Reference Material Working Group (WG01) is to identify and facilitate the development, characterization, and qualification of reference materials (RMs) to support the wide adoption of new and existing Rapid Microbiology Test Methods (RMTMs) within the Advanced Therapy Industry.
WG02 MISSION: The mission of the Methods and Validation Schemes Working Group (WG02) is to develop a framework for the validation of methods to support the wide adoption of new and existing Rapid Microbiology Test Methods (RMTMs) by the Advanced Therapy Industry.
WG03 MISSION: The mission of the Interlaboratory Study Design and Implementation Working Group (WG03) is to design and implement interlaboratory studies to assess the analytical performance of various RMTMs while also evaluating the performance and fitness for purpose of candidate reference materials.
CONSORTIUM IMPACT
This consortium is expected to lead to measurement assurance tools for RMTM and increased confidence in their use for advanced therapy products due to:
- Stakeholder input into the design of microbial reference material(s)
- Data from interlaboratory studies to support development of best practices and standard methods, including ISO standards (e.g., ISO/CD 24190 -- Biotechnology — Analytical Methods — Risk-based approach for method selection and validation for rapid microbial detection in bioprocesses)
- Improved rapid microbial measurement capabilities
CONSORTIUM MODEL
- Convenes industry, academia, and government to identify and address measurement and standards needs across the rapid microbial testing methods field
- Enables members to work with NIST to develop measurement solutions and standards
- Leverages NIST expertise in measurement science, standards development, reference materials, technology development, and basic research
- Collaborates with related programs at other organizations
WHY NIST
- Cross-disciplinary expertise in engineering and the physical, information, chemical, and biological sciences
- As a non-regulatory agency of the U.S. Department of Commerce, NIST does not impose standards; standards are accepted by consensus
- Neutral convener for industry consortia, standards development organizations, federal laboratories, universities, public workshops, and interlaboratory comparability testing

