Material Matters
The Quarterly Magazine of NIST's Material Measurement Laboratory
Fall 2017
Measuring Materials to Improve Lives
In this issue
- A Message from the MML Director
- A New Way to Test Body Armor
- Measuring Liquid Body Armor
- NIST, NFL, GE and Under Armour Announce Dynamic Research and 6D Helmets as Grand Prize Winner in Head Health Challenge III
- New NIST Tool Aims to Improve Accuracy of Test to Determine Cardiovascular Disease Risk
- How the Cone Snail’s Deadly Venom Can Help Us Build Better Medicines
- New Method for Identifying Carbon Compounds Derived from Fossil Fuels
- NIST’s Quick Test May Speed Antibiotic Treatment and Combat Drug Resistance
- Assessing the Higher Order Structure of Monoclonal Antibodies
- Outreach and Partnering
- Recent Awards
- NIST Standard Story: Matters of the Heart
A Message from the MML Director

Director*
Material Measurement Laboratory
NIST
A key part of NIST’s mission is advancing measurement standards and technology to improve the quality of our lives, and little is more central to the quality of our lives than our personal safety and security. Across MML’s research portfolio in the biological, chemical, and materials sciences, from advanced materials development to chemical identification reference data and clinical diagnostic standards, our work helps industry ensure and enhance our personal safety.
MML scientists engage in a wide-range of security-related research, from developing and standardizing contraband screening technologies to developing materials that can protect better, and much more. This past summer MML updated its Mass Spectral Library, used by virtually every manufacturing sector to identify chemicals, and in the identification and detection of chemical weapons. MML researchers also recently demonstrated screening techniques to help prevent accidental exposure to synthetic opioids, including fentanyl, helping law enforcement address the opioid crisis.
In addition to our research focused on threats posed by contraband materials, MML research also helps keep us safe in our everyday lives. With cutting-edge instruments, methods and standards, we accurately determine the composition, structure and properties of materials, and explore how new materials might outperform those used in existing products. In this issue of Material Matters you can read about recent research that gives us a better understanding of the fibers used in modern body armor, as well as an effort to develop better materials for helmets and pads to protect against traumatic brain injury.
Other research highlighted in this issue demonstrates less direct, but no less important ways that MML research helps promote our welfare – exploring better treatments for diseases like Alzheimer’s, a new test for evaluating antibiotics more quickly, improving a blood test used to evaluate the risk of heart disease, and more accurately determining the sources of carbon dioxide in the atmosphere.
Body, mind, heart – our research is leading to enhancements that promise to help protect us and improve our lives in innumerable ways. Across all of these sectors and disciplines, MML research shares a desire and drive to harness materials to improve human life, making us safer and more secure, so we can focus on the things that really matter.
* During the search for a new MML director, Michael Fasolka, long-time MML deputy director, is acting director of MML.
A New Way to Test Body Armor
October 16, 2017

Scientists at the National Institute of Standards and Technology (NIST) have developed a new way to investigate the high-performance fibers used in modern body armor. Described in the Journal of Polymer Science, the research may help increase confidence in the apparel that protects military units, police departments and public figures from gunfire. It may also lead to the development of new, lighter weight materials for body armor in the future.
High-performance polymer fibers have been used in ballistics applications for more than 40 years. Traditionally, these fibers are woven together into a fabric and then layered 15-20 times over to make a vest with a thickness of anywhere from about 6 to 13 millimeters (a quarter to half an inch). Although effective at stopping or slowing down bullets, users have sometimes found these vests, which are worn either under or over clothing, to be heavy and bulky—akin to wearing 15 to 20 shirts at once on a hot summer day. Many would like a more comfortable alternative.
The testing of soft body armor has been a big concern because the deployment of a new kind of fiber—believed to be superior to the previous material—unexpectedly failed in 2003, resulting in
Measuring Liquid Body Armor
At the Annual Conference of the North American Thermal Analysis Society, from August 7 to 10, 2017 in Newark, Delaware, Ran Tao of MML’s Security Technologies Group presented on her recent work of shear thickening fluids. Shear thickening fluids composed of silica nanoparticles and polyglycol (a liquid polymer) have been investigated as performance enhancement additives for soft body armor. Such material is ideal for damping and energy absorption applications because, at high rate, the fluid undergoes a fluid-like to solid-like transition and, in doing so, the impact energy is dissipated. Tao’s research highlighted a method of visualizing the breakdown of fumed silica agglomerates during measurement. Using different data presentation and analysis methods of large amplitude oscillatory shear rheology, the stress instabilities, as an indication of agglomerates breakdown, can be readily obtained. The results indicate that those shear thickening fluids containing fractal fumed silica are not ideal candidates as liquid body armor due to the irreversible agglomerate breakdown under
New NIST Tool Aims to Improve Accuracy of Test to Determine Cardiovascular Disease Risk
September 19, 2017
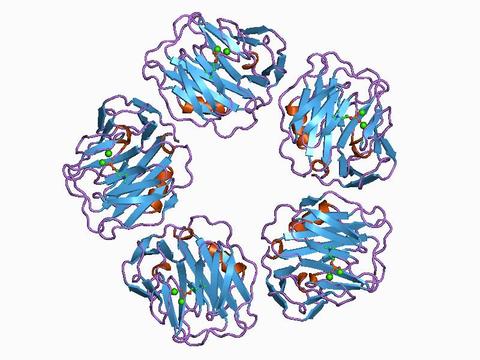
Cardiovascular disease caused one out of three deaths in the United States in 2016, and for decades it has been the leading killer for both men and women. Hoping to reduce these numbers, researchers at the National Institute of Standards and Technology (NIST) have developed a new Standard Reference Material (SRM) that can improve the results of a common blood test used to assess a person’s risk of heart disease.
The blood test measures C-reactive protein (CRP), which is a marker for inflammation in the body. While the precise relationship between slightly elevated CRP levels and cardiovascular disease is still being determined, research suggests that inflammation in arteries can lead to plaque buildup, and then to heart attacks and strokes. Some studies indicate that high-sensitivity CRP (hsCRP) tests—which detect minute amounts of the protein in blood—may have advantages for predicting heart disease when cholesterol counts are normal.
The hsCRP test kits are made from antibodies that attach to CRP in a blood sample like a lock and key to provide an accurate count of the protein. However, depending on their source and quality, some of these antibodies attach better than others, leading to variable results between batches and kit makers
How the Cone Snail’s Deadly Venom Can Help Us Build Better Medicines
October 10, 2017

Cone snails have inspired humans for centuries. Coastal communities have often traded their beautiful shells like money and put them in jewelry. Many artists, including Rembrandt, have featured them in sketches and paintings. Now, scientists at the National Institute of Standards and Technology (NIST) are finding these deadly predators inspiring, too, as they seek new ways to cure old medical problems using the poisonous snails as models.
“This is the same venom used to kill dinosaurs in ‘Jurassic Park,’” says NIST biochemist Frank Marí, with a chuckle. “It is scary stuff, but that power could be used for a different kind of good in real life.”
Like all NIST scientists, Marí measures things. Specifically, he measures RNA and the associated proteins at work inside marine animals. As technology has improved over the years, he and his team have become better able to examine, analyze and catalog the molecules at work in some of the ocean’s lesser-known creatures, including cone snails. This year, his lab made several significant discoveries about their venom, discoveries that might ultimately lead to the development of new medicines for hard-to-treat diseases. By imitating the way that these small, quiet creatures deliver poison, scientists may be able
NIST’s Quick Test May Speed Antibiotic Treatment and Combat Drug Resistance
September 22, 2017

BOULDER, Colo.—Researchers at the National Institute of Standards and Technology (NIST) have demonstrated a potential new tactic for rapidly determining whether an antibiotic combats a given infection, thus hastening effective medical treatment and limiting the development of drug-resistant bacteria. Their method can quickly sense mechanical fluctuations of bacterial cells and any changes induced by an antibiotic.
Described in Scientific Reports, NIST’s prototype sensor provides results in less than an hour, much faster than conventional antimicrobial tests, which typically require days to grow colonies of bacterial cells. Delayed results from conventional tests allow dangerous infections to progress before effective treatments can be found and provides a time window for bacteria to develop drug resistance.
Improperly prescribed antibiotics and antibiotic-resistant bacteria pose serious threats to public health. At least 2 million illnesses and 23,000 deaths are attributed to antibiotic-resistant bacterial infections in the United States each year, according to a 2013 report from the Centers for Disease Control and Prevention.
One solution may be the new NIST sensing approach, based on a quartz-crystal resonator whose vibrations vary in measurable ways when particles on the surface change. The method, which involves bacterial cells adhered to a resonator, represents a new way of using these supersensitive crystals, which NIST researchers previously demonstrated for applications such as measuring carbon nanotube purity.
The new NIST technique senses
Assessing the Higher Order Structure of Monoclonal Antibodies
The sale of innovative and generic biologic medicines is a major driver of U.S. economic activity. Ten of the top-selling 20 drugs are protein therapeutics. Of these, seven are monoclonal antibodies (mAbs), by far the largest class of protein therapeutics. The development and manufacture of therapeutic mAbs presents many analytical challenges. Characterization of mAbs requires assessing their higher order structure, since misfolding or aggregation can lead to loss of efficacy or cause potentially life-threatening immune responses. Techniques for accurate and precise characterization of mAb higher order structure for establishing consistency in drug manufacturing, detecting process-related drug-product variations and establishing comparability of biosimilars to innovator reference products are therefore of great interest to regulators and the biopharmaceutical industry. An MML team of Luke Arbogast, Frank Delaglio, John Schiel and John Marino has developed a nuclear magnetic resonance (NMR) method that produces spectral ‘fingerprints’ to establish higher order structure comparability between mAb samples. This work is described in a recently published article entitled “Multivariate Analysis of Two-Dimensional 1H, 13C Methyl NMR Spectra of Monoclonal Antibody Therapeutics To Facilitate Assessment of Higher Order Structure,” that was selected by American Chemical Society journal editors to be featured in ACS Editors’ Choice.
Using a benchmark 2D NMR experiment, with principle component analysis applied directly to the spectral data matrix, spectra from highly similar species are successfully discriminated, with low limits of detection, that cannot be distinguished by visual inspection or simple intensity based statistical approaches. The MML team is further able to use this approach to identify the sources of spectral variation between species and assign these differences to alterations in structure. The team also previously demonstrated that the NMR analysis can be performed in an hour or less, which makes this a practical method for higher order structure determination in the manufacturing environment.
Outreach and Partnering
NIST-EPA Inter-Agency Agreement on Fecal Waste Contaminants in Water
Scott Jackson, leader of MML’s Complex Microbial Systems Group is working with scientists from the U.S. Environmental Protection Agency’s (EPA) National Risk Management Research Laboratory, Water Supply and Water Resources Division to develop molecular genetic reference materials for validating analytical methods developed at the EPA for the detection of fecal waste contaminates in our waterways. Fecal microbes are the most common biological contaminants in U.S. waters and pose serious public and ecological health risks. A nationwide network of regional and state laboratories uses the EPA-developed methods to monitor local recreational waters for safety and for evaluating best management practices. The NIST-developed reference material will be used to assess the analytical sensitivity and specificity of the EPA-developed methods as well as ensuring interlaboratory competency through annual performance testing.
CDC Human Microbiome Disruption Meeting
On September 18, 2017 MML Group Leader Scott Jackson visited the Centers for Disease Control and Prevention in Atlanta, to develop a set of standard practices for assessing, during drug development, the degree and significance of human microbiome disruption or restoration resulting from exposure to medications such as antibiotics, microbiome protectants, or microbiome restoratives. Several pre-calls with invited participants were hosted in advance to develop a starting draft of standard practices and define current gaps in approaches to assess drug-related microbiome disruption and gaps in evidence linking potential microbiome indices to outcomes. During this meeting, Jackson gave a presentation on the ongoing activities at NIST surrounding standards for microbiome measurements as well current antibiotic resistance research. The meeting was hosted by the CDC’s Division of Healthcare Quality Promotion.
NIST Joins Joint Agency Microbiome Group
Last year the FDA and NIH partnered to form the Joint Agency Microbiome Working Group. The group, composed of scientists interested in microbiome research, recently expanded their strategic document to officially include NIST as a member. MML’s Scott Jackson is representing NIST in the group. The goals of the group are to:
- Promote trans-agency collaborations in advancing microbiome related science
- Keep the three agencies appraised of current microbiome-related research
- Keep the three agencies appraised of scientific gaps related to regulatory questions/ needs
- Keep the three agencies updated on past and upcoming microbiome related meetings
- Provide updated general information about the regulation of microbiome-related resources such as probiotics and fecal microbial transplants
- Coordinate minisymposium or retreat to share intramural research
Industry Engagement on Neutron and X-Ray Measurements
During the week of September 18, 2017, MML staff visited, in succession, the Toyota Research Institute of North America (Ann Arbor, Michigan), Dow Chemical (Midland, Michigan), and the former Dow Corning (Midland, Michigan). Both Toyota and Dow Chemical are members of the NIST public-private consortium nSoft, and Dow Chemical has had a 20+ year cooperative research and development agreement with the NIST Synchrotron Science Group located at Brookhaven National Laboratory. After recent visits to the Gaithersburg campus, there was a request for a reverse site visit to discuss emerging capabilities in neutron and X-ray measurements. In addition to interests in characterization of membranes for water purification and energy storage, all three companies expressed great interest in applying new NIST capabilities for X-ray spectroscopic microscopy to the characterization of catalysts.
NIST Agreement on Non-Animal Assay for Assessing Skin Sensitivity to Chemicals
Scientists from MML’s Biosystems and Biomaterials Division will work with scientists from the U.S. Consumer Product Safety Commission (CPSC) through an interagency agreement to improve the measurement assurance of a non-animal alternative assay focused on assessing the potential skin sensitization risk for chemicals. This assay is currently being evaluated with a three-laboratory comparison through the Interagency Coordinating Committee for the Validation of Alternative Methods (ICCVAM). The work being conducted at NIST will focus on how to use measurement science approaches (cause and effect analysis, robustness testing, and quantifying different sources of variability) to enhance the quality of the assay. This work has already uncovered unexpected sources of variability that were not highlighted in the original protocol (e.g., photodegradation of assay reagents, variable quality of key reagents among suppliers, impact of different cuvettes) and enabled the design of potential in-line process control measurements to ensure confidence in the assay result. Furthermore, the NIST/CPSC team will evaluate the feasibility of using this assay to test more complex compounds such as nanomaterials. This work and the interlaboratory comparison results will be used to statistically determine specification ranges to ensure assay performance.
MML’s Applied Chemicals and Materials Division Begins SBIR Phase II with Innoveering
On September 5, 2017, Elisabeth Mansfield, leader of MML’s Thermophysical Properties of Fluids Group kicked off a Phase II Small Business Innovation Research (SBIR) project with Innoveering LLC to design a high-temperature, low-uncertainty pressure sensor. Innoveering is a small business based in New York that has a history of designing pressure sensors for oil and gas applications. They are applying their knowledge developed for previous applications to the development of a new sensor for NIST. The Thermophysical Properties of Fluids group has an interest in a new pressure sensor to overcome some of the challenges with current pressure sensor size and calibrations. The new pressure sensor is much smaller (1 in x 1 in), has a pressure range up to 7MPa, and is based on MEMS technology. It is expected that this sensor will allow NIST to develop smaller instrumentation for thermophysical property measurements and improve the uncertainties in current instrumentation. The finalized sensor is expected to be delivered at NIST by August 31, 2019. The SBIR program encourages domestic small businesses to engage in federal research/research and development that has the potential for commercialization.
MML Leads Microscopy & Microanalysis Symposium on Advances in Scanning Electron Microscopy
Bob Keller, leader of MML’s Nanoscale Reliability Group, teamed with Professors Raynald Gauvin (McGill University) and Shirin Kaboli (University of Nevada-Las Vegas) to organize and run a symposium entitled Advances in Scanning Electron Microscopy: Transmission Modes and Channeling Effects at the Microscopy & Microanalysis Meeting, held in St. Louis, August 7-9, 2017. The Symposium drew many of the world’s experts in both transmission and electron channeling methods, which share numerous common aspects of electron scattering and detection physics. This was the first symposium at the Microscopy & Microanalysis conference that included transmission scanning electron microscope (SEM) methods as a primary focus. The historically uncommon transmission approach has seen rapid research and commercial growth during the past five years, since the NIST development of transmission-EBSD (aka transmission Kikuchi diffraction) was first reported. Transmission-centric topics included imaging methods, electron diffraction methods, and electron energy loss spectrometry in the SEM, for characterization of a wide variety of substances, including metals, semiconductors, ceramics, rocks and minerals, and biological material. Channeling-centric topics included electron channeling contrast imaging and electron channeling pattern methods, used primarily for studies of deformation in metals and semiconductors.
Recent Awards

Stafford Selected as 2017 American Physical Society Fellow
MML project leader Chris Stafford (Functional Polymers Group) has been selected as a 2017 Fellow of the American Physical Society (APS). Stafford is being recognized for “developing innovative techniques to enhance physical understanding of polymer thin films, including combinatorial arrays with controlled gradients, buckling methods to quantify modulus, and layer-by-layer methods to deduce structure-property relationships in polymer membranes.” APS Fellowships are a distinct honor given after extensive review by the Fellowship committee of the appropriate APS division, topical group or forum, and by the APS Fellowship Committee.

Marino Honored by American Chemical Society for Talk
John Marino, leader of MML’s Biomolecular Structure & Function Group, was honored by the Division of Biochemical Technology (BIOT) in the American Chemical Society (ACS) with the “Best of BIOT” award for his talk presented at the 2017 ACS National Meeting in San Francisco titled “Addressing the challenge of higher-order structure assessment of biologics with 2D NMR.” In addition to the honor, Marino was featured in the “Best of BIOT” webinar series.

MML Team Receives Netexplo Award for Global Innovation
The Netexplo Observatory has awarded Matthew Staymates (left, left), Jessica Staymates, Bill MacCrehan, Greg Gillen, Roderick Kunz, Thomas Mendum, Ta-Hsuan Ong, Geoffrey Geurtsen, and Brent Craven a Netexplo Innovation 2017 award for their work in understanding canine olfaction with an artificial dog’s nose and implications for biomimicry and improved contraband detection. The Netexplo awards 10 individuals/organizations whose work represents the 10 most innovative and promising global digital initiatives of the year. The award ceremony was held at the United Nations Educational, Scientific and Cultural Organization (UNESCO) house in Paris, France on April 26 and 27, 2017.

Glover Named ASTM Emerging Professional
Jack Glover of MML’s Security Technologies Group was recently selected as an ASTM International Emerging Professional for his contribution to the revision of the ASTM F792, a widely-used international standard that defines test methods for assessing the imaging performance of the X-ray systems that are used to inspect small baggage, such as the carry-on baggage in air transportation. This competitive recognition is awarded for new ASTM members who have demonstrated the potential to be committee leaders. Glover improved existing test methods, developed new test methods, developed algorithms to automatically compute values for the performance metrics, updated and identified new performance metrics, and designed and validated test artifacts. He was also the chair of the F792 working group that revised ASTM’s F792 standard.
Gorham Named ASTM Emerging Professional
Justin Gorham of MML’s Nano Materials Research Group has recently been selected as an ASTM International Emerging Professional. This competitive recognition is awarded for new ASTM members who have demonstrated the potential to be committee leaders. Gorham joined ASTM in 2015 and has contributed significantly to the development of one published standard and one work item in Committee E56 on Nanotechnology, Subcommittee E56.06 on Nano-Enabled Consumer Products. These are A Standard Guide for Tiered Approach to Detection and Characterization of Silver Nanomaterials in Textiles and A New Test Method for Detection of Total Silver in Textiles by ICP Analysis. These two standards are critical to industries that manufacture textile products containing silver nanomaterials and to agencies that regulate such products. In addition to his standards work with ASTM, Gorham has co-led the production of a NIST silver nanoparticle reference material.

Chiang Named Distinguished Speaker by the Society of Petrophysicists
Wei-Shan Chiang, a NIST guest researcher (sponsored by Aramco) at the NIST Center for Neutron Research, has been named a Distinguished Speaker by the Society of Petrophysicists and Well Log Analysts (SPWLA) for 2017-2018. She was selected for her paper entitled “Simultaneous Neutron and X-ray Imaging of 3D Kerogen and Fracture Structure for Flow Path in Shales,” which she presented at the 58th Annual SPWLA Symposium. As a Distinguished Speaker Chiang will visit and speak at local chapters of SPWLA throughout the coming year. This honor represents an important achievement for NIST’s nSoft, as a definitive advance in the imaging of shale. Aramco North America Services (Houston, TX) has been a member of nSoft since 2015. This work was highlighted at the recent nSoft annual meeting.
NIST Standard Story: Matters of the Heart
September 20, 2017

NIST scientists have thoroughly measured and characterized more than 1,300 physical products, NIST Standard Reference Materials®, to help people in industry, academia, and government agencies calibrate instruments, verify their test methods, and develop new measurement methods. NIST reference materials, for example, help manufacturers make interoperable parts in far-flung facilities, medical labs check the accuracy of cholesterol and other clinical tests, and scientists monitor environmental threats.
What
Standard Reference Material® 2924 C-Reactive Protein (CRP) Solution
A unit of the SRM contains 3 vials, each with 1 milliliter of a solution of recombinant CRP. A certified concentration value for CRP is reported along with reference density, relative average mass and concentration values (expressed in terms of grams per liter). This SRM® is primarily intended for use in calibrating procedures and devices for the determination of C-reactive protein (CRP) in human serum. It can also be used for value-assignment of in-house calibrator solutions and control materials.
Why
C-reactive protein (CRP) is a biomarker for inflammation due to infections or other medical conditions (e.g. rheumatoid arthritis or lupus). Because persistent low levels of inflammation can also be associated with cardiovascular disease, high-sensitivity CRP (hs-CRP) assays, that can detect low levels of CRP



