Summary
NIST develops cancer measurements by working with our stakeholders to deliver improved measurement methods, data, test materials, interlaboratory testing projects, and reference materials for cancer biomarkers that can be used to improve measurements of assays used in basic research and in clinical applications. The projects are developed in consultation with cancer experts from industry, academic, and government laboratories.
Description
Collaborations and Cooperative Research Agreements
We welcome collaborations and can help with assay validation and development of reference materials. Below are some examples of our collaborations and ways we can develop test materials.
- Early Detection Research Network (EDRN): Interagency agreement to provide measurements to early detection of cancer biomarkers as a reference laboratory. Collaboration with EDRN biomarker discovery labs to help in method validation and test materials. Development and interlaboratory testing of reference materials for circulating tumor DNA (ctDNA), methylated DNA biomarkers in liquid biopsy, and extracellular vesicles.
- NIST has an ongoing collaboration with the Molecular Characterization Laboratory (MCL) at the Frederick National Laboratory for Cancer Research to provide them with RMs for validation of actionable targets. SRM 2373 genomic DNA for HER2 gene copy numbers was used by MCL scientists to validate the performance of digital PCR and Next Generation Sequencing (NGS) assays for evaluating gene variations associated with an increased risk of breast cancer. Reference: “Certified DNA Reference Materials to Compare HER2 Gene Amplification Measurements Using Next-Generation Sequencing Methods”
- Foundation NIH Cancer Biomarker Consortium, project: Development of circulating tumor DNA reference materials. Memorandum of Understanding to work to provide measurements of ctDNA for testing and developing reference materials for ctDNA. Partners NCI, Dana Farber Cancer Institute, and Astra Zeneca
- Collaboration with Dr. David Wong, UCLA School of Dentistry, Associate Dean of Research to investigate the mechanism of the electronic chip detection of cell-free DNA for cancer detection.
- Collaboration with Dr. Steve Bogen, Tufts Medical Center, quantitative measurements of the binding of PDL-1 antibodies for calibrations of immunohistochemistry measurements for cancer immunotherapies
- ATTC: Materials Transfer Agreement for extracellular vesicles (EVs) for materials for cancer detection.
- NIST has an ongoing cooperative research and development agreement (CRADA) with LGC Clinical Diagnostics to develop reference materials for DNA methylation measurements.

NIST Test Materials for Liquid Biopsy Cancer Biomarkers Measurements
The science of noninvasive disease detection has advanced greatly since circulating cell free DNA (cfDNA) was first reported in body fluids in 1948. Since then, the evolution of sensitive detection technologies and the findings of tumor-derived entities such as nucleic acids (cfDNA and cfRNA), extracellular vesicles (EVs), circulating tumor cells, and proteins have enabled the development of liquid biopsies with many clinical applications. Liquid biopsy has several advantages over the conventional tissue biopsies, including its minimally invasive nature, easy of sampling, a continuous monitoring of genetic alterations, and a better reflection of intertumoral and intertumoral heterogeneity. Liquid biopsy has great promise for early detection, treatment selection, and monitoring of cancer.
Reference materials are needed to help develop, validate, and ensure the quality of new cancer biomarker assays. The analysis of cfDNA is especially challenging because somatic variant alleles are typically present in low concentrations relative to germline DNA. cfDNA analyses are currently being studied for many applications, including disease detection, treatment monitoring, and assay development.
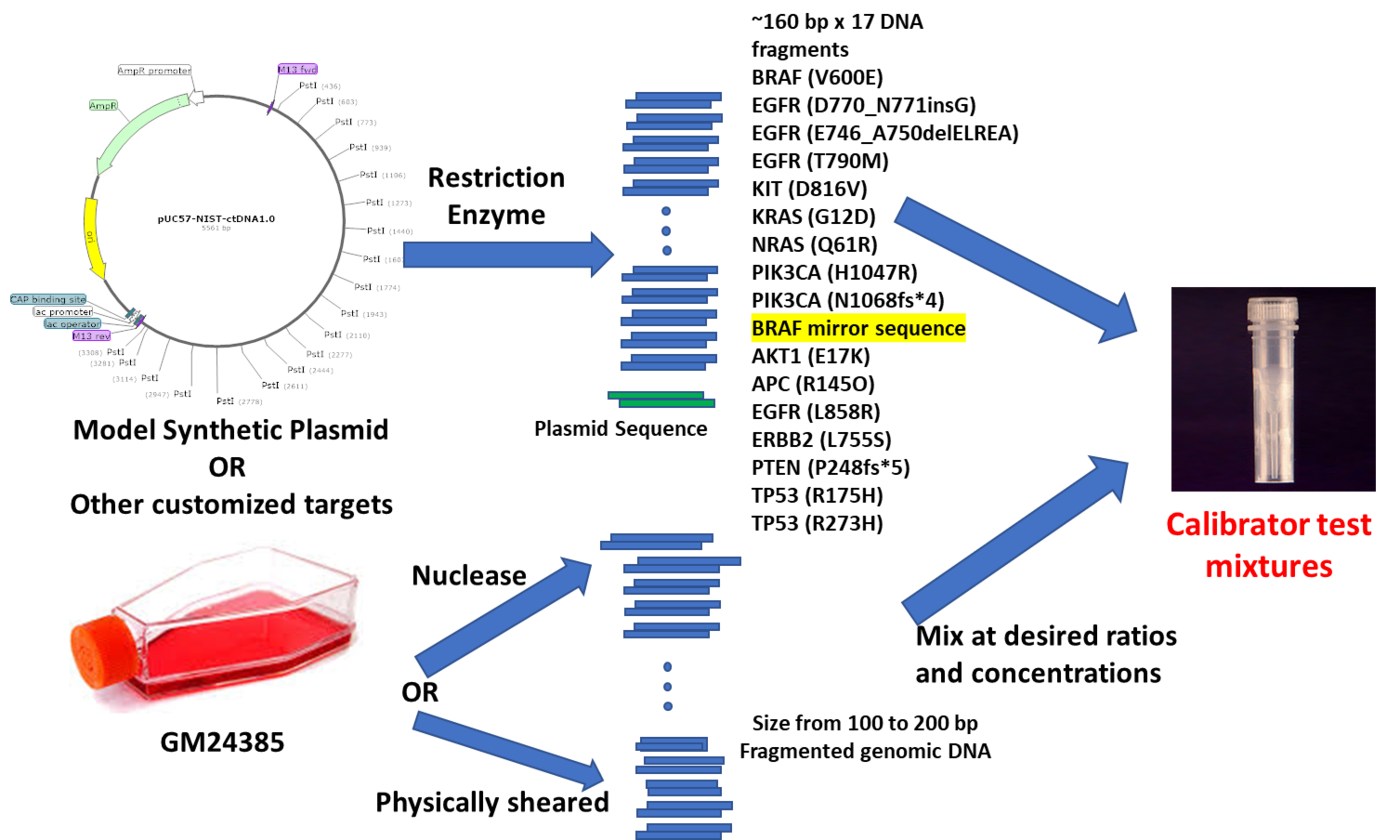
We have developed a platform to produce customized NIST ctDNA test materials (as shown in the figure). In brief, a plasmid was constructed in a way that the spike-ins with ~160 bp DNA fragments harboring the cancer biomarker variants were tandemly linked by a restriction enzyme site. Equal molar concentration of variants was achieved by restriction enzyme digestion. Background (normal) cfDNA was prepared from either by physically shearing or enzymatic digestion of genomic DNA from GM24385 cells, which has one of the most sequenced human genomes. NIST ctDNA test materials were produced by mixing the spike-ins and background (normal) cfDNAs at desired ratios and concentrations. These materials were then characterized by DNA concentration, DNA fragment size, and variant allele fractions (VAFs).
DNA methylation is one of the major epigenetic modifications that almost exclusively occur within the CpG dinucleotide in the human genome by substitution of a methyl group at position 5 of the cytosine base. Global hypomethylation and specific hypermethylation at gene promoters is one of the key hallmarks of many cancers. Methylation alterations usually happens at the early carcinogenesis and methylation patterns may indicates the tissue of origin. Therefore, cfDNA methylation has a great potential for cancer early detection biomarker.
Currently, bisulfite treatment is still the gold standard procedure for single-base resolution methylation assays. However, bisulfite treatment damages DNA, which results in fragmentation, DNA loss, and biased sequencing data. Furthermore, it could introduce DNA methylation false positives when the treatment is incomplete. Therefore, reference materials and reference/control assays are highly desired for DNA methylation measurements.
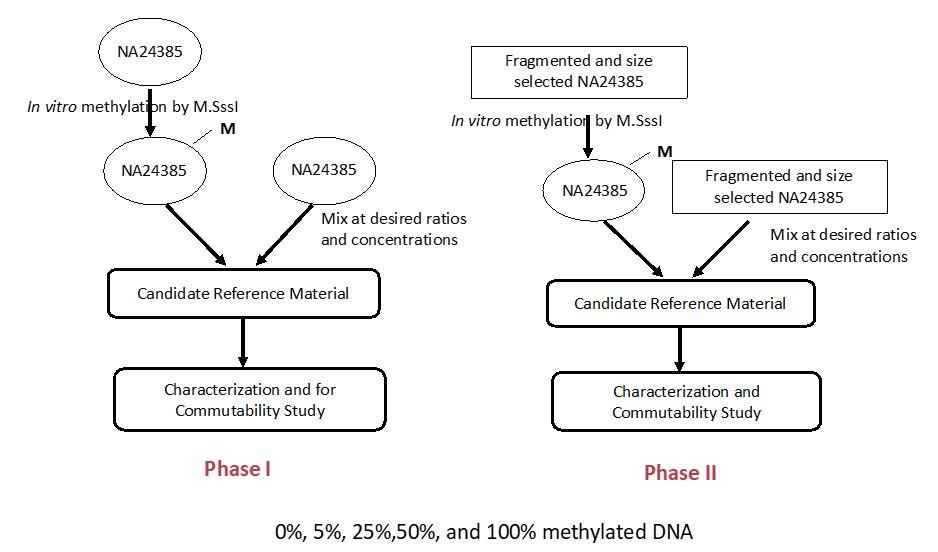
We have developed DNA methylation test materials for both genomic DNA (Phase I) and cfDNA (Phase II) formats. The below chart is the strategy for the preparation of NIST Phase I and Phase II materials:
NIST invites participants to an Interlaboratory Testing Program to evaluate the suitability and utility of two sets of methylated cell free DNA (cfDNA) test materials to serve as reference materials or controls for the measurements of cfDNA methylation. The study is sponsored by the Early Detection Research Network (NCI) and organized as a collaborative project developed by NIST and LGC Clinical Diagnostics (formerly known as SeraCare Life Sciences, Inc).
NIST Cancer Biomarker Reference Materials
STANDARD REFERENCE MATERIAL (SRM) 2373®: GENOMIC DNA STANDARDS FOR HER2 MEASUREMENTS
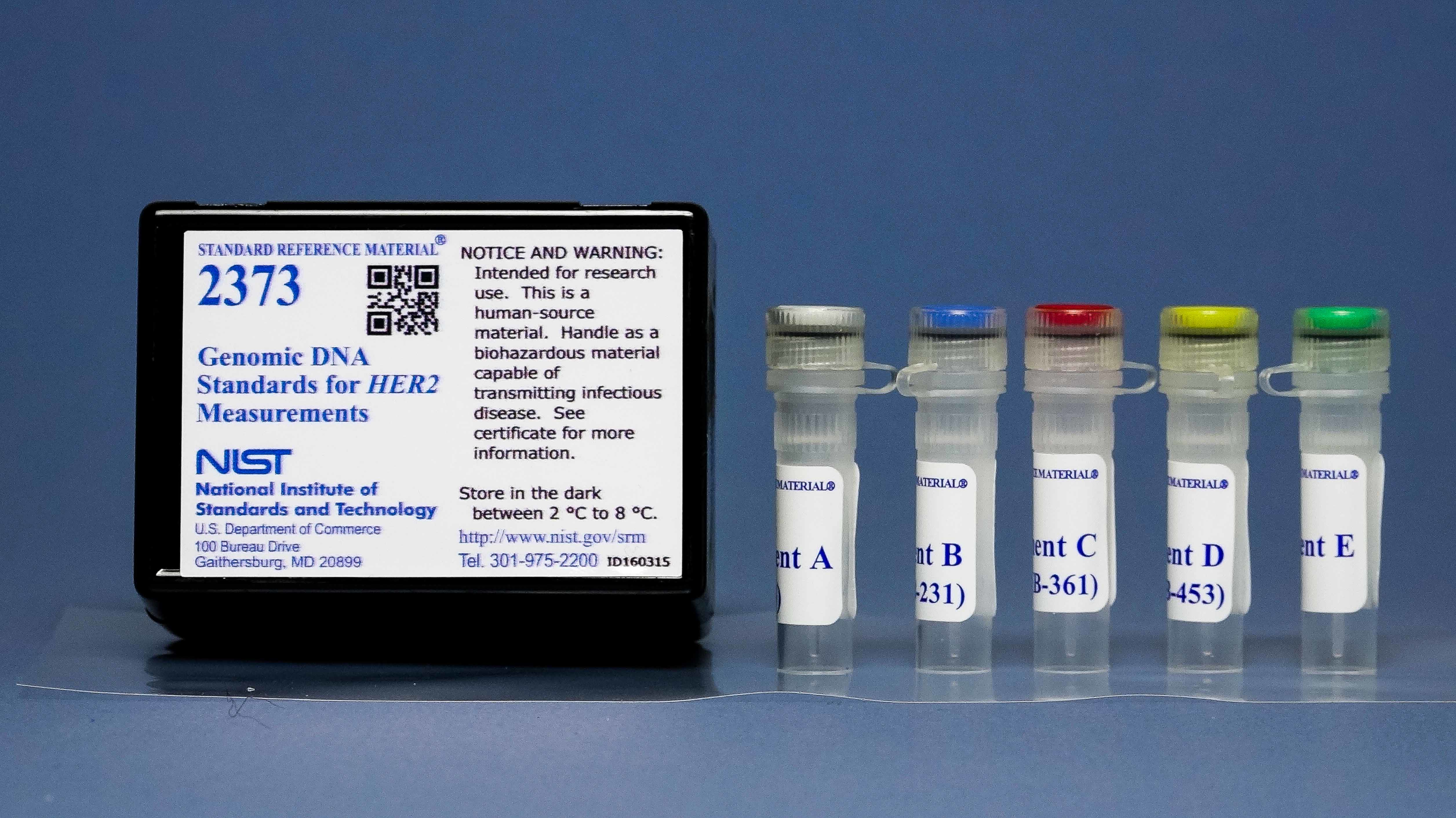
The amplification of the gene for Human Epidermal Growth Factor Receptor 2 (HER2) occurs in approximately 20 to 25% of breast cancers. The accurate measurement of this biomarker is important for the proper treatment with anti-HER2 therapeutics. Diagnostic tests like HER2 assays make it possible to select the best-personalized treatment for individual patients based on the genetic makeup of their tumors.
The copy numbers of the HER2 gene and 3 reference genes (not amplified) were measured using quantitative PCR and digital PCR assays. The certified values of the components are the ratios of the HER2 gene copy number to the reference gene copy numbers. The PCR assays were validated and calibrated using SRM®2372 component A (human genomic DNA). The stability of the components was shown by repeated measurements over several years. The DNA concentration determined from absorbance and the PCR assays was provided as informational value. This SRM is available from NIST as SRM 2373®.
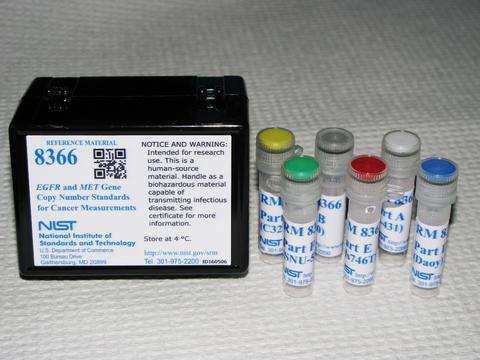
RM 8366: GENE COPY NUMBER STANDARDS FOR EGFR AND MET
We have developed genomic DNA RM for copy number measurements of the genes for MET and EGFR (Epidermal Growth Factor Receptor). MET and EGFR are both tyrosine kinase proteins found on the surfaces of cells that response to external growth factors. Mutations or amplification of the genes for MET and EGFR can cause increased cellular signaling for cell growth or mobilization resulting in cancer. The accurate measurements of these cancer biomarkers are important for the correct diagnostics to determine the treatment, since specific therapeutics are available for both of these targets.
RM 8366 consists of purified genomic DNA isolated from six different cancer cell lines with different amounts of amplification of the EGFR and MET genes (Fig.2). It is intended to harmonize the measurements of ratios of the human EGFR and MET genes to unamplified reference genes. This RM is available from NIST as RM 8366.