Patent of the Month: September 2021
Patent Description
The discovery of single-wall carbon nanotubes (SWCNTs) has unveiled the existence of not just one but a family of several hundred stable macromolecules. They are simple in composition and atomic structure—all made of carbon atoms locally bonded in the hexagonal geometry of graphene, but variations in the helical twist angle (θ) of the hexagons and in tube diameter (d) result in a diverse set of nanotube electronic structures. On the basis of theoretical analysis and experimental observation, SWCNTs can be ranked in an order according to the width of their electronic bandgap: armchair metallic tubes (θ=30°) with zero bandgap; non-armchair semimetallic tubes with small (<100 meV) but nonvanishing bandgaps that scale as cos(3θ)/d2; and semiconducting tubes with bandgaps that scale as 1/d.
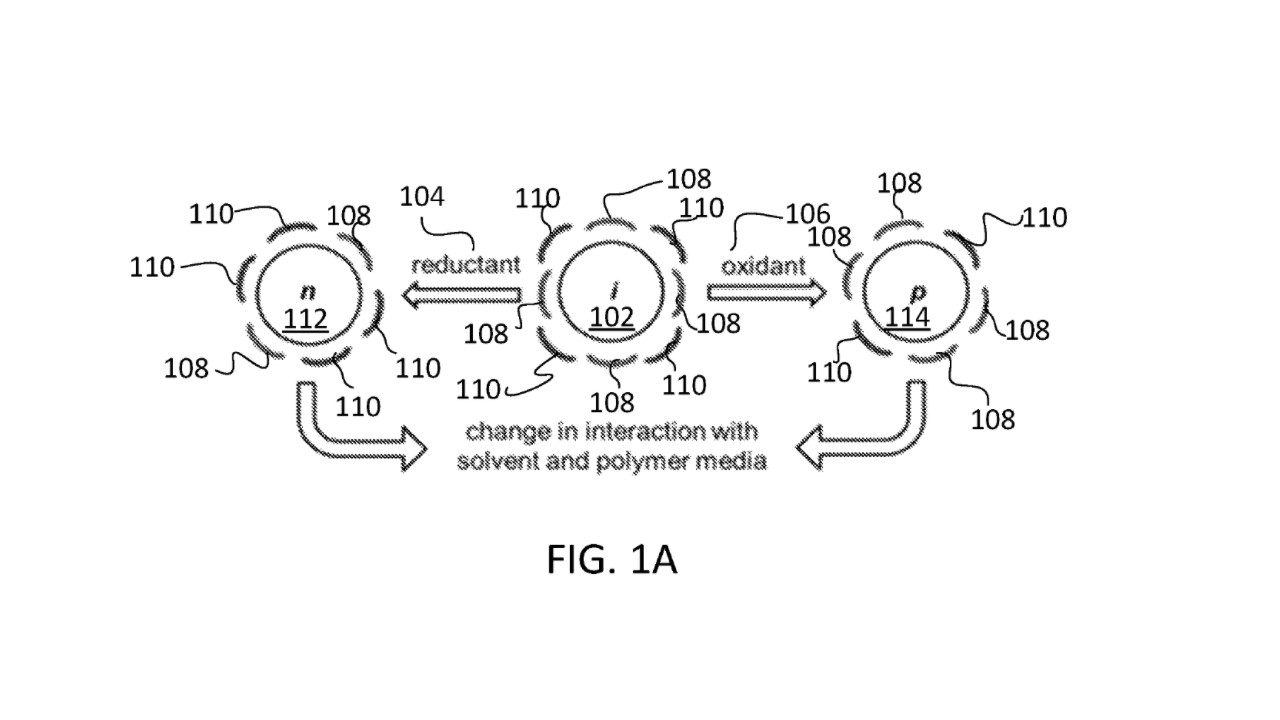
Refer to the FIGURE 1A (FIG. 1A) below. FIG. 1A shows a schematic diagram of the chemistry involved for this method. Black circles 102, 112, and 114 each represents an individual nanotube. A line 108 represents a first type of surfactant molecule and a line 110 represents a second type of surfactant molecule. The surfactant molecules 108 and 110 can form a surfactant coating layer around an individual nanotube. Exemplary surfactant molecules include sodium dodecyl sulfate (SDS) and sodium cholate (SC).
This method can cause electron transfer between SWCNTs and exogenous (i.e., externally added) redox molecules. The transfer of electrons induces surfactant coating layer reorganization, which in turn changes the interaction between dispersed SWCNTs and their solvent media, altering the outcome of a number of separation processes. Dispersed SWCNTs refers to SWCNTs that are stabilized by surfactant molecules, which help keep SWCTNs in solution. The oxidized, resting, and reduced states of nanotubes are denoted by p, i, and n, respectively. A resting nanotube 102, when exposed to an oxidant 106, becomes an oxidized nanotube 114. In contrast, when the resting nanotube 102 is exposed to a reductant 104, it becomes a reduced nanotube 112.
For illustration purposes, eight surfactant molecules are shown schematically arranged in an alternating pattern of surfactant molecule 108 and surfactant molecule 110 around the resting nanotube 102 in FIG. 1A. Different number of surfactant molecules (i.e., fewer than eight, or greater than eight) are possible.
After oxidation by the oxidant 106, the surfactant molecules rearrange such that only six surfactant molecules surround the oxidized carbon nanotube 114, with two surfactant molecules 110 being arranged directly opposite each other. Four surfactant molecules 108 occupy four remaining positions around the oxidized nanotube 114. The schematic diagrams are used to illustrate the reorganization of surfactant molecules, and the arrangement illustrated in FIG. 1A is simply an example of a possible arrangement.
After reduction by the reductant 104, the surfactants are rearranged such that only six surfactant molecules surround the reduced carbon nanotube 112, with two surfactant molecules 108 being arranged directly opposite each other. Four surfactant molecules 110 occupy four remaining positions around the reduced nanotube 112. In other words, the placement of the surfactant molecules 108 around the reduced nanotube 112 is the same as the placement of the surfactant molecules 110 around the oxidized nanotube 114. The arrangement of surfactant molecules depicted in FIG. 1A is only for illustrative purposes and other arrangements are possible. The changes in the arrangement of the surfactant molecules for nanotubes having different oxidation states (i.e., resting, oxidized or reduced) cause a change in the interaction of the respective carbon nanotubes with the solvent and polymer media. The polymer media can refer to, for example, the mixture of PEG, DX, surfactants and solvents. Water is an example of a solvent.
Inventors
Jason K. Streit
Angela R Hight Walker
Ming Zheng
University of Southern California
Technology Types
Analytical Chemistry and Carbon Nanotubes
Chemical Science
Benefits
This method makes the separation process much more robust than current practices.

