Summary
The National Institute of Standards and Technology (NIST) is working to advance measurement capabilities needed to study the world’s climates. Climate Science seeks to understand the relation between the physical and chemical properties of oceanic, atmospheric, and geographic masses with long term changes in climates. The Chemical Sciences Division is developing advanced metrology and certified reference materials to provide accurate and reproducible measurements of greenhouse gases (carbon dioxide, methane, nitrous oxide), halocarbons, aerosols, particulate matter, and chemical oceanographic parameters (seawater pH, total alkalinity, and dissolved inorganic carbon) – factors that influence and reflect climate change. Robust data are essential for the computational models used in assessing trends in climate change that can inform public policy decisions.
Description
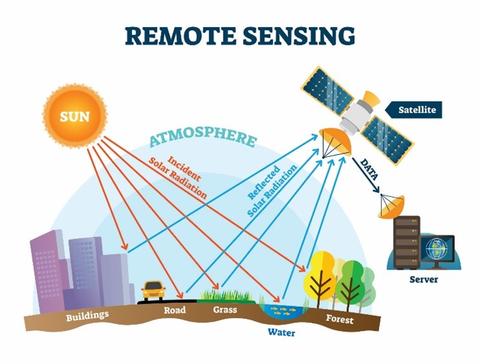
This program encompasses multifaceted efforts to advance the metrology of climate science. Projects include:
- development of ultra-high accuracy optical standards and technologies to determine greenhouse gases (GHGs) concentrations, atmospheric lifetimes, and aerosol radiative forcing
- development of a unified definition of seawater pH that is consistent with the definition of pH in solutions having much lower ionic strength and development of tools and standards to support SI-traceable measurements of seawater chemical parameters
- production of Standard Reference Materials (SRMs®) and other reference materials to provide measurement traceability to the SI (international system of units)
- production of Standard Reference Data (SRD®)
- production of Standard Reference Instrument (SRI)
- development of computational models
- calibration service for ozone analyzers
These activities provide technologies, instruments, standards, data, and informatics tools to support the Nation’s needs to identity and quantify the amount of GHGs and reactivity of aerosols in Earth’s atmosphere, as well as the state of the Earth’s oceans with respect to climate change. By advancing chemical metrology and data analysis tools, these activities provide critically needed aid to policy makers in areas such as environmental research, assessment and mitigation of climate change, and policy development.
Major Accomplishments
- Optical methods for remote sensing of GHG. NIST has developed and applied advanced cavity-enhanced laser spectroscopy methods yielding high-accuracy spectroscopic data for quantitative analysis of greenhouse gases such as carbon dioxide, water vapor, methane, and oxygen. These methods provide information on amount of substance and isotopic composition. This work has played a key role in enabling accuracy and continuity in space- and ground-based remote-sensing measurements of atmospheric carbon and other species that are required for robust, long-term investigations of climate change and air-quality monitoring.
- Reference Materials for GHG. Exceptionally accurate gas standards have been developed for the three most important atmospheric greenhouse gases (GHG) in air: carbon dioxide, methane, and nitrous oxide. Three sets of concentration standards linked to the kilogram were developed for dissemination to the entire climate science community: Northern Continental Air (SRM 1720), Southern Oceanic Air (SRM 1721), and Halocarbons in Continental Air (SRM 1722). These standard mixtures contain low, background amounts of target species in the Northern- and Southern-Hemispheres, respectively. This work harmonizes and extends previously existing gas standards maintained by participants in the World Meteorological Organization (WMO) Global Atmospheric Watch Program with standards needed by other science organizations that conduct research on the global carbon cycle and climate dynamics.
- Aerosol Metrology. Aerosols (i.e., airborne particles) represent the second largest contributor to atmospheric heating (or potentially cooling) after CO2. Aerosols are often heterogeneous in size, density, composition, and morphology. These species either heat or cool earth’s atmosphere depending on the specific aerosol’s absorption and scattering properties. Because the optical properties of aerosols are poorly understood, research has been undertaken to advance the metrology of aerosols to permit the development of improved climate models. A process has been developed that permits particle synthesis and innovative metrological techniques for characterization of aerosol optical properties of relevance to climate change. The key innovations include the ability to generate aerosols of well-defined optical properties, the separation of arbitrarily shaped particles by both size and mass, and measurement of their mass-specific optical absorption and scattering properties. This novel approach enables the establishment of new aerosol standards and overcomes limitations of speed and accuracy inherent in traditionally used filter-based sampling methods.
- pH metrology. Studies of global climate change rely on accurate characterization of carbon dioxide (CO2), the most prominent atmospheric greenhouse gas. Earth's oceans act as a repository for atmospheric CO2, and contain the single greatest quantity of actively cycled carbon in the world. Internationally recognized seawater pH standards are essential in providing an accepted benchmark for the uptake of CO2 by the oceans. NIST has developed, measured, and published results and detailed protocols for primary pH measurements of multiple artificial seawater pH standards that bracket the current and future pH ranges of natural seawater. This effort leverages the long-term experience of NIST with primary laboratory measurements of pH, the results of which are formally recognized by international comparisons with other National Metrology Institutes worldwide, to seawater pH metrology. Three aspects are recognized: (1) primary pH metrology (documented through pH SRMs and international pH Key Comparisons performed as part of the NIST involvement in CCQM and SIM, (2) new procedures developed at NIST for assessing the purity and homogeneity of indicator reagents, (3) published methodology for characterizing the pH response of indicator dyes, including rigorous uncertainty statements as a component required for achieving measurement traceability to the SI.
- Direct air capture (DAC). Direct air capture technologies extract carbon dioxide (CO2) directly from Earth’s atmosphere. The CO2 can be stored via mineralization in deep geological formations or used in applications such as in food processing or the production of synthetic fuels. DAC is therefore one potential component towards achieving net negative emissions and contributing to the mitigation of global warming and climate change.
Because of the low concentrations of CO2 in air (≈0.04%), for DAC to be successful, a new generation of sorbents, the materials that can capture and release CO2, will be required. These materials must have a high uptake capacity, have an energy-efficient uptake/release cycle, and operate over multiple uptake/release cycles at efficient rates over a range of temperatures and humidities. New and tailored instrumentation is required to make such measurements. Candidate DAC materials include metal-organic frameworks (MOFs), modified silicates, and zeolites.
To accelerate this technological innovation, the Division has a combined experimental and theoretical program. The experimental program includes measurements of the adsorption capacity of a material under static pressure and constant temperature conditions—known as sorption isotherm measurements. Another technique--known as breakthrough analysis—provides insights into a material’s performance under dynamic conditions. These measurements also provide information about how a material works with multicomponent gases and about the selectivity of a sorbent. Theoretical studies of these materials provide insights into the structure-property relationship of these sorbents, which This allows for a better understanding of the performance of these materials and enables the design of new materials. Of course, new instruments and theoretical methods are being developed as a part of this effort.
Ultimately, the goal is to provide reference sorption data on reference DAC materials. This will allow for the critical and impartial evaluation of new, emerging DAC materials and a more comprehensive understanding of their performance. - Climate models. In addition to carbon dioxide and methane, atmospheric release of other compounds often used by U.S. industry are thought to contribute to the global warming. The EPA has identified a set of nearly 1,200 compounds that are known or reasonable suspected to have a significant global warming potential (GWP). NIST has developed models that help to identify the GWP for individual compounds, based on quantum chemistry and artificial neural network calculations.
ASSOCIATED PUBLICATIONS
1. Orkin, V. L., Khamaganov, V. G., and Kurylo, M. J., "Experimental kinetic study of the reactions between OH radicals and three 2-butenes over the temperature range 220-370 K and pressure range 0.67-40 kPa (5-300 Torr)," International Journal of Chemical Kinetics, 55, 221-237 (2023).
2. Clegg, S. L., Humphreys, M. P., Waters, J. F., Turner, D. R., and Dickson, A. G., "Chemical speciation models based upon the Pitzer activity coefficient equations, including the propagation of uncertainties. II. Tris buffers in artificial seawater at 25C, and an assessment of the seawater Total pH scale," Marine Chemistry, 244, (2022).
3. Gordon, I. E., Rothman, L. S., Hargreaves, R. J., Hashemi, R., Karlovets, E. V., Skinner, F. M., Conway, E. K., Hill, C., Kochanov, R. V., Tan, Y., Wcislo, P., Finenko, A. A., Nelson, K., Bernath, P. F., Birk, M., Boudon, V., Campargue, A., Chance, K. V., Coustenis, A., Drouin, B. J., Flaud, J. M., Gamache, R. R., Hodges, J. T., Jacquemart, D., Mlawer, E. J., Nikitin, A. V., Perevalov, V. I., Rotger, M., Tennyson, J., Toon, G. C., Tran, H., Tyuterev, V. G., Adkins, E. M., Baker, A., Barbe, A., Cane, E., Csaszar, A. G., Dudaryonok, A., Egorov, O., Fleisher, A. J., Fleurbaey, H., Foltynowicz, A., Furtenbacher, T., Harrison, J. J., Hartmann, J. M., Horneman, V. M., Huang, X., Karman, T., Karns, J., Kassi, S., Kleiner, I., Kofman, V., Kwabia-Tchana, F., Lavrentieva, N. N., Lee, T. J., Long, D. A., Lukashevskaya, A. A., Lyulin, O. M., Makhnev, V. Y., Matt, W., Massie, S. T., Melosso, M., Mikhailenko, S. N., Mondelain, D., Muller, H. S. P., Naumenko, O. V., Perrin, A., Polyansky, O. L., Raddaoui, E., Raston, P. L., Reed, Z. D., Rey, M., Richard, C., Tobias, R., Sadiek, I., Schwenke, D. W., Starikova, E., Sung, K., Tamassia, F., Tashkun, S. A., Vander Auwera, J., Vasilenko, I. A., Vigasin, A. A., Villanueva, G. L., Vispoel, B., Wagner, G., Yachmenev, A., and Yurchenko, S. N., "The HITRAN2020 molecular spectroscopic database," Journal of Quantitative Spectroscopy & Radiative Transfer, 277, (2022).
4. Guallart, E. F., Fajar, N. M., Garcia-Ibanez, M. I., Castano-Carrera, M., Santiago-Domenech, R., Hassoun, A. E., Perez, F. F., Easley, R. A., and Alvarez, M., "Spectrophotometric Measurement of Carbonate Ion in Seawater over a Decade: Dealing with Inconsistencies," Environmental Science & Technology, 56, 7381-7395 (2022).
5. Humphreys, M. P., Waters, J. F., Turner, D. R., Dickson, A. G., and Clegg, S. L., "Chemical speciation models based upon the Pitzer activity coefficient equations, including the propagation of uncertainties: Artificial seawater from 0 to 45C," Marine Chemistry, 244, (2022).
6. Lisak, D., Charczun, D., Nishiyama, A., Voumard, T., Wildi, T., Kowzan, G., Brasch, V., Herr, T., Fleisher, A. J., Hodges, J. T., Ciurylo, R., Cygan, A., and Maslowski, P., "Dual-comb cavity ring-down spectroscopy," Scientific Reports, 12, (2022).
7. Long, D. A., Adkins, E. M., Mendonca, J., Roche, S., and Hodges, J. T., "The effects of advanced spectral line shapes on atmospheric carbon dioxide retrievals," Journal of Quantitative Spectroscopy & Radiative Transfer, 291, (2022).
8. Bailey, D. M., Zhao, G., and Fleisher, A. J., "Precision Spectroscopy of Nitrous Oxide Isotopocules with a Cross-Dispersed Spectrometer and a Mid-Infrared Frequency Comb," Analytical Chemistry, 92, 13759-13766 (2020).
9. Li, X. Y., Garcia-Ibanez, M. I., Carter, B. R., Chen, B. S., Li, Q., Easley, R. A., and Cai, W. J., "Purified meta-Cresol Purple dye perturbation: How it influences spectrophotometric pH measurements," Marine Chemistry, 225, (2020).
10. Yao, Q., Asa-Awuku, A., Zangmeister, C. D., and Radney, J. G., "Comparison of three essential sub-micrometer aerosol measurements: Mass, size and shape," Aerosol Science and Technology, 54, 1197-1209 (2020).
11. Brewer, P. J., Kim, J. S., Lee, S., Tarasova, O. A., Viallon, J., Flores, E., Wielgosz, R. I., Shimosaka, T., Assonov, S., Allison, C. E., van der Veen, A. M. H., Hall, B., Crotwell, A. M., Rhoderick, G. C., Hodges, J. T., Mahn, J., Zellweger, C., Moossen, H., Ebert, V., and Griffith, D. W. T., "Advances in reference materials and measurement techniques for greenhouse gas atmospheric observations," Metrologia, 56, (2019).
12. Plusquellic, D. F., Wagner, G. A., Briggman, K., Fleisher, A. J., Long, D. A., and Hodges, J. T., "Simultaneous DIAL, IPDA and point sensor measurements of the greenhouse gases, CO2 and H2O," 2019 Conference on Lasers and Electro-Optics (Cleo), (2019).
13. Zangmeister, C. D., Grimes, C. D., Dickerson, R. R., and Radney, J. G., "Characterization and demonstration of a black carbon aerosol mimic for instrument evaluation," Aerosol Science and Technology, 53, 1322-1333 (2019).
14. Rhoderick, G. C., Kelley, M. E., Miller, W. R., Norris, J. E., Carney, J., Gameson, L., Cecelsld, C. E., Harris, K. J., Goodman, C. A., Srivastava, A., and Hodges, J. T., "NIST Standards for Measurement, Instrument Calibration, and Quantification of Gaseous Atmospheric Compounds," Analytical Chemistry, 90, 4711-4718 (2018).
15. Zangmeister, C. D., You, R., Lunny, E. M., Jacobson, A. E., Okumura, M., Zachariah, M. R., and Radney, J. G., "Measured in-situ mass absorption spectra for nine forms of highly-absorbing carbonaceous aerosol," Carbon, 136, 85-93 (2018).
16. Zangmeister, C. D., Radney, J. G., "Absorption Spectroscopy of Black and Brown Carbon Aerosol,". In: Multiphase Environmental Chemistry in the Atmosphere. ACS Publications, pp. 275-297 (2018).
17. Bailey, D. M., Adkins, E. M., and Miller, J. H., "An open-path tunable diode laser absorption spectrometer for detection of carbon dioxide at the Bonanza Creek Long-Term Ecological Research Site near Fairbanks, Alaska," Applied Physics B, 123, 1-10 (2017).
18. Radney, J. G., You, R., Zachariah, M. R., and Zangmeister, C. D., "Direct In Situ Mass Specific Absorption Spectra of Biomass Burning Particles Generated from Smoldering Hard and Softwoods," Environmental Science & Technology, 51, 5622-5629 (2017).
19. Radney, J. G. and Zangmeister, C. D., "Light source effects on aerosol photoacoustic spectroscopy measurements," Journal of Quantitative Spectroscopy and Radiative Transfer, 187, 145-149 (2017).
20. Zangmeister, C., "Measured absorption spectra of aerosolized carbonaceous species and their influence on climate forcing," Abstracts of Papers of the American Chemical Society, 254, (2017).
21. Allison, T. C., "Application of an Artificial Neural Network to the Prediction of OH Radical Reaction Rate Constants for Evaluating Global Warming Potential," Journal of Physical Chemistry B, 120, 1854-1863 (2016).
22. Betowski, D., Bevington, C., and Allison, T. C., "Estimation of Radiative Efficiency of Chemicals with Potentially Significant Global Warming Potential," Environmental Science & Technology, 50, 790-797 (2016).
23. Radney, J. G. and Zangmeister, C. D., "Practical limitations of aerosol separation by a tandem differential mobility analyzer-aerosol particle mass analyzer," Aerosol Science and Technology, 50, 160-172 (2016).
24. Rhoderick, G. C., Kitzis, D. R., Kelley, M. E., Miller, W. R., Hall, B. D., Dlugokencky, E. J., Tans, P. P., Possolo, A., and Carney, J., "Development of a Northern Continental Air Standard Reference Material," Analytical Chemistry, 88, 3376-3385 (2016).
25. Lin, H., Reed, Z. D., Sironneau, V. T., and Hodges, J. T., "Cavity ring-down spectrometer for high-fidelity molecular absorption measurements," Journal of Quantitative Spectroscopy & Radiative Transfer, 161, 11-20 (2015).
26. Long, D. A., Wojtewicz, S., Miller, C. E., and Hodges, J. T., "Frequency-agile, rapid scanning cavity ring-down spectroscopy (FARS-CRDS) measurements of the (30012)<-(00001) near-infrared carbon dioxide band," Journal of Quantitative Spectroscopy & Radiative Transfer, 161, 35-40 (2015).
27. Orkin, V. L., Khamaganov, V. G., and Guschin, A. G., "Photochemical Properties of Hydrofluoroethers CH3OCHF2, CH3OCF3, and CHF2OCH2CF3: Reactivity toward OH, IR Absorption Cross Sections, Atmospheric Lifetimes, and Global Warming Potentials," Journal of Physical Chemistry A, 118, 10770-10777 (2014).
28. Orkin, V. L., Martynova, L. E., and Kurylo, M. J., "Photochemical Properties of trans-1-Chloro-3,3,3-trifluoropropene (trans-CHCl=CHCF3): OH Reaction Rate Constant, UV and IR Absorption Spectra, Global Warming Potential, and Ozone Depletion Potential," Journal of Physical Chemistry A, 118, 5263-5271 (2014).
29. Radney, J. G., You, R. A., Ma, X. F., Conny, J. M., Zachariah, M. R., Hodges, J. T., and Zangmeister, C. D., "Dependence of Soot Optical Properties on Particle Morphology: Measurements and Model Comparisons," Environmental Science & Technology, 48, 3169-3176 (2014).
30. Rhoderick, G. C., Duewer, D. L., Apel, E., Baldan, A., Hall, B., Harling, A., Helmig, D., Heo, G. S., Hueber, J., Kim, M. E., Kim, Y. D., Miller, B., Montzka, S., and Riemer, D., "International Comparison of a Hydrocarbon Gas Standard at the Picomol per Mol Level," Analytical Chemistry, 86, 2580-2589 (2014).
31. Long, D. A. and Hodges, J. T., "On spectroscopic models of the O-2 A-band and their impact upon atmospheric retrievals," Journal of Geophysical Research-Atmospheres, 117, (2012).
32. Long, D. A., Cygan, A., van Zee, R. D., Okumura, M., Miller, C. E., Lisak, D., and Hodges, J. T., "Frequency-stabilized cavity ring-down spectroscopy," Chemical Physics Letters, 536, 1-8 (2012).
33. Rhoderick, G. C., Carney, J., and Guenther, F. R., "NIST Gravimetrically Prepared Atmospheric Level Methane in Dry Air Standards Suite," Analytical Chemistry, 84, 3802-3810 (2012).
34. Patten, K. O., Khamaganov, V. G., Orkin, V. L., Baughcum, S. L., and Wuebbles, D. J., "OH reaction rate constant, IR absorption spectrum, ozone depletion potentials and global warming potentials of 2-bromo-3,3,3-trifluoropropene," Journal of Geophysical Research-Atmospheres, 116, (2011).
35. Rhoderick, G. C., Duewer, D. L., Ning, L., and DeSirant, K., "Hydrocarbon Gas Standards at the pmol/mol Level to Support Ambient Atmospheric Measurements," Analytical Chemistry, 82, 859-867 (2010).

