Taking Measure
Just a Standard Blog

From left: Carolyn Burdette (NIST), Laura Regalado (CENAM) and I discuss the analysis of vitamins in the lab at NIST.
Our modern world depends on global connections. For instance, how can you be sure that the nutrition labels are correct on imported food products such as that fancy pasta you like to buy? How do you know that those products are free of harmful substances like mercury or other heavy metals? Many authoritative organizations keep watch and play important roles in making sure that internationally traded products are safe and have an acceptable level of quality. And, you guessed it, many of the judgment calls come in the form of some sort of chemical measurement.
One of the organizations that works behind the scenes to do this is the International Committee for Weights and Measures (CIPM; from the French Comité International des Poids et Mesures). The CIPM’s job is to maintain the worldwide equivalence of “units of measure,” which in the International System of Units (SI) includes the much-beloved seven base units: the meter, mole, kilogram, kelvin, candela, ampere and second. Much of the work done to ensure the ability to compare measurements around the world happens in the CIPM’s Consultative Committees (or CCs for short).
For the state of the art of global chemical and biological measurements, there is the Consultative Committee for Amount of Substance: Metrology in Chemistry and Biology (CCQM; from the French Comité Consultatif pour la Quantité de Matière). With the redefinition of the SI scheduled to go into effect on May 20, and the CCQM celebrating its 25th anniversary, there’s no better time to tell you about how our committee serves the world through measurement.
Here’s the official party line, as excerpted from the 2017 CCQM Strategy Document:
The CCQM is responsible for developing, improving and documenting the equivalence of national standards (certified reference materials and reference methods) for chemical and biological measurements. The objectives of the CCQM are: 1) to progress the state of the art of chemical and biological measurement science, 2) to reach out to new and established stakeholders, and 3) to demonstrate the global comparability of chemical and biological measurements.
What this boils down to is that all the national metrology institutes (NMIs) regularly get together to design a series of interlaboratory tests to make sure that each NMI can measure the same thing to the same degree. When the results are in and everyone’s measurements have been shown to be comparable, then we can trust one another’s standards and thus one another’s measurements of commercial products. For the CCQM, this includes biological materials, gas mixtures, inorganic and organic chemical species in many types of industrial and food materials as well as healthcare applications, and most recently, we’ve begun to make stable isotope-ratio measurements, which are important for determining the origins of foods and environmental studies, among other things.
The National Institute of Standards and Technology (NIST) had a sizable contingent attending the CCQM working group meetings from April 6-12 at the International Bureau of Weights and Measures in Sèvres, France. Our committee’s work covers nearly all aspects of the CCQM. This was the CCQM’s 25th meeting, and former NIST Director Willie E. May is completing his term as president of the CCQM this month.

I have been part of NIST’s CCQM activities for the past decade, first as a laboratory metrologist and data analyst and now as a leader. As might be common for many of us at NIST, I started my research career as an expert in the physical sciences, or more specifically, a concoction of both environmental analytical and physical organic chemistry. Over time, my career has evolved, and I’ve taken on measurement science roles and started to attend CCQM meetings. I’m happy to say that I can now comfortably wear the hat of “chemical metrologist.”

And as an international expert in chemical metrology, it makes sense that my work at CCQM has centered on its activities in organic chemical analyses. Shortly after attending my first meeting, I began to realize NIST’s unique role as one of the primary NMIs on this international committee. This is no surprise, given that NIST has one of the oldest, largest and most diverse catalogs of certified reference materials in the world. Being the 800-lb gorilla in the (committee) room, NIST has been expected to coordinate and participate in many of the CC’s activities. Many other NMIs also claim SI traceability, an unbroken chain of measurements that link a product to a recognized international measurement standard, but their calibrations are often done using our gases, pure substances and solution standard reference materials (SRMs). This is—and remains—an immense responsibility for us and serves as the basis for all the chemical measurements that support the global economy.
Through my CCQM activities, it became clear that one of the missing links was an SI-traceable primary standard that would help labs around the world assess the purity of organic materials important for medical tests, food safety, drug production and more. The characterization of organic reference standards is performed using a now-common spectroscopic method called quantitative nuclear magnetic resonance (qNMR). This important need prompted us to develop NIST PS1, an ultra-purified benzoic acid standard for standards” in which we measure the number of organic molecules in samples by qNMR. NIST PS1 can, in turn, be linked to both the kilogram and the mole, the latter being the SI unit for amount of substance. It took us several years to complete, and we are proud of this accomplishment. This “first of its kind” primary standard provides a direct connection to the SI for the majority of NIST’s—and many other NMIs’—organic chemical measurements.
Another worldwide impact we’ve made has been the direct comparison of certified reference materials (CRMs) produced by most NMIs around the world. (CRMs are the generic name for the kinds of reference materials NIST produces in the form of SRMs, which is our trademarked brand name for them.) This endeavor is much further away from a direct realization of their SI traceability, but I would argue this is one of the best ways to determine just how comparable our measurements are in practice. Over the past decade, we’ve compared global suites of CRMs for hexane in methane gas (fuel components), ethanol in water (beverage alcohol levels) and creatinine (routine clinical biomarker) in human blood serum. I have literally cut my metrological teeth on these studies.
More recently, we have partnered with Singapore’s Health Sciences Authority to jointly coordinate a study of urea and uric acid (as routinely tested clinical metabolites) in human serum and plasma CRMs from five NMIs, including NIST. After painstaking, repeated measurements and a comprehensive data analysis, we can confidently state that our current catalogs contain metrologically equivalent CRMs.
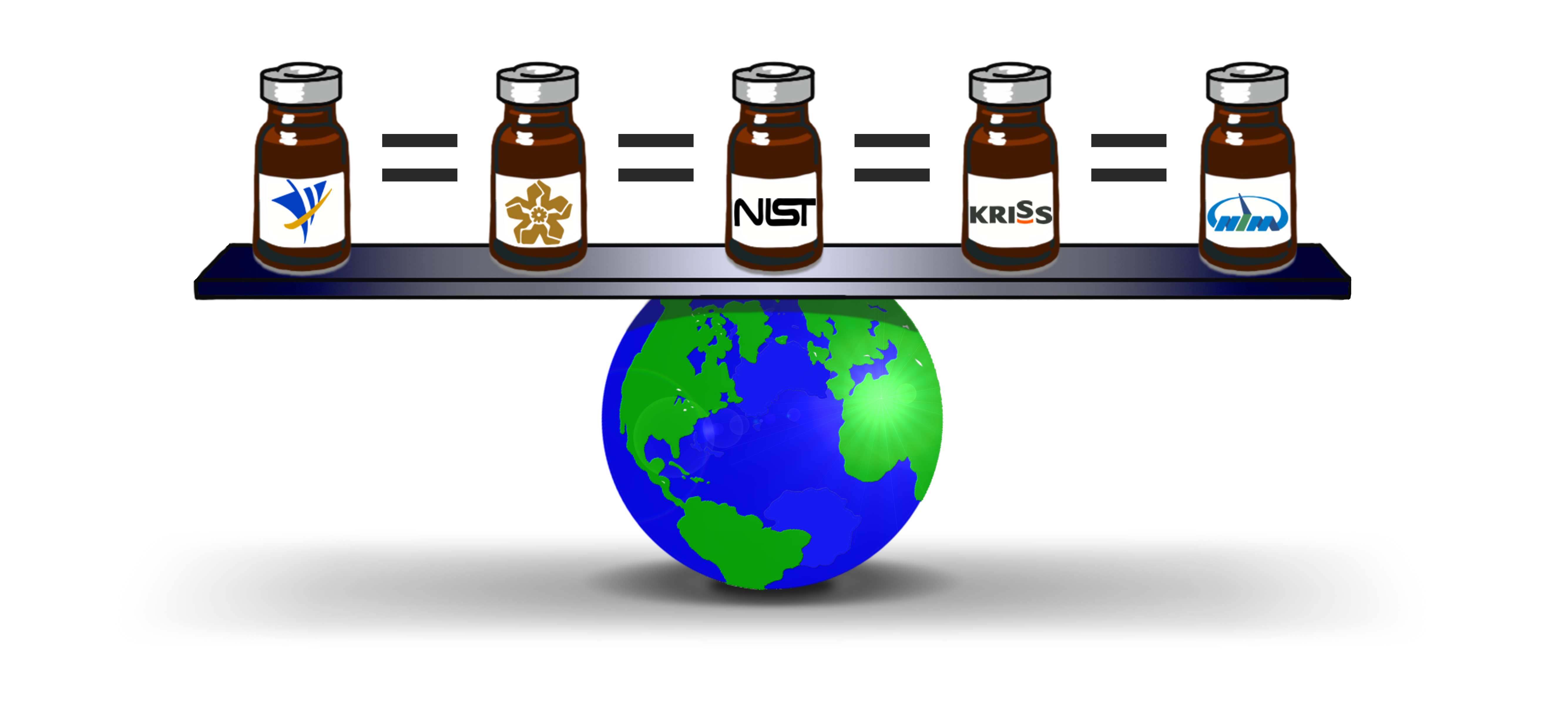
These studies ensure that the clinical measurement services provided as certified reference materials in Singapore, South Korea, China, Mexico and the U.S. are of the highest quality and equivalent to others around the world. This then trickles down to each country’s consumers through their measurement infrastructure.
Our many opportunities to make connections with our sister NMIs and the spinoff collaborations that result are the most personally rewarding experiences from my CCQM engagements. Our bilateral and multilateral arrangements include NMIs from Brazil, Argentina, Costa Rica, Canada, South Africa, Kenya, Australia, U.K., Germany, the Netherlands and many more. We are especially committed to bolstering these relationships through our own regional metrology organization, the Inter-American Metrology System (SIM).
As an example, we are collaborating with Mexico’s Centro Nacional de Metrología (CENAM) to develop higher-order vitamin measurement methods for foods and human serum and to further progress nutritional-content milk powder and infant formula-based CRMs for both institutes.

And, of course, we could not miss the opportunity to co-coordinate another CCQM study with CENAM for the comparison of vitamin B3 in milk powder and infant formula CRMs around the globe. The final results of this study were presented at the April 2019 CCQM meeting, and these materials have also been deemed equivalent!
The bottom line is that the work that the CCQM does quietly in the background ensures that the food, drink, medicine and countless other consumer products are safe no matter where in the world you travel. These kinds of connections are fundamental to our way of life. They draw us closer together and strengthen international trade and the worldwide economy. After all, the greater part of any economy is trust and confidence—trust and confidence that transactions are fair and that you are getting what you pay for regardless of the product’s country of origin. I’m proud to be a part of an institution that serves a vital function and that benefits literally everyone.
*edited April 25, 2019, to correct a caption
About the author
Related Posts
Comments
Hi, thank you for your interest! please feel free to send me an email to [email protected] with more detail on your food product query, and I will follow up.
this blog is very nice and information for Linking the World Through Chemical Measurements thanks for sharing us
thanks!






Greetings, I am planning to sell a packaged cake and I need to know what proven preservative better fit , can I have this information with ccqm? , My mail is [email protected] , thank you for this valuable information