Commercial-scale production of any high quality and consistent product relies on accurate measurements. Unlike static materials, biological systems and processes can be difficult to measure because they are always changing. NIST develops solutions for these measurement problems to help the U.S. maintain leadership of the global bioeconomy.
Engineering biology, also known as synthetic biology, enables the intentional design, manufacture and deployment of living cells, cell systems, and other biological processes to create materials and products that benefit us, like crops that resist drought and pests, renewable energy sources, advanced medicines, and more.
NIST’s measurement science and other technical activities support robust innovation and more sustainable manufacturing practices in distributed manufacturing centers, which help alleviate supply chain disruptions for synthetic biology’s most-needed products, like commodity chemicals and materials, and medicines.
NIST research promotes:
Innovation
We provide industry members and researchers with tools to automate the many complex processes required to intentionally design living cells to perform as factories.
- The Living Measurement Systems Foundry is a unique, flexible automation system for measuring the whole landscape of cells, from the genes that direct their growth to the substances they produce, and selecting cells for their desired functions.
- The NIST Prototype Cell Assay Measurement Platform (P-CAMP) is a model biofoundry for rapid generation of robust protocols for biological and cellular measurements. It provides unique end-to-end automation to advance measurement science for emerging biotechnologies.
- The Flow Cytometry Standards Consortium develops quantitative measurements and standards for the measurement of cell characteristics to support emerging biotechnologies and advanced biomanufacturing.
- NIST partners with the research community and contributes to the development of measurement solutions, shared protocols, and standards for innovation in engineering biology through the Global Biofoundry Alliance, the Build-A-Cell collaboration, and the Synthetic Biology Open Language standard.
Commercialization
NIST develops cell-based reference materials that provide benchmarks for industry developers and regulators to check the quality of their measurements and laboratory processes—helping to ensure rapid development, efficient manufacturing processes, and safe, marketable products.
- A living cell reference material will help researchers and product developers accurately count microbes as they are cultured. It can also be used as a safe material for training first responders to detect biothreats.
- The NIST Chinese Hamster Ovary cell line, or NISTCHO, will allow biopharmaceutical manufacturers and academic and government researchers to develop new measurements and methods for assessing the biological processes that occur in the CHO cell line, which is used around the world to produce monoclonal antibodies for medical therapies.
- Artificial intelligence and machine learning are powerful tools for large-scale measurements of complex biological systems. Regulators need visibility into the computer algorithms behind these tools in order to gain trust in them, while industry requires similar trust that algorithms are reliable from one production run to another. NIST develops “explainable” AI and machine learning for control of complex biological functions to meet industry’s product specifications.
American Leadership of Standards
NIST works closely across the federal research enterprise and industrial biomanufacturing groups to contribute to national initiatives and develop measurement solutions and standards that facilitate commerce in the U.S., and American manufacturers’ participation in international markets.

- We lead or contribute to standards development through Standards Development Organizations such as ISO and IEC to develop standards for the industry, thus promoting U.S. competitiveness.
- We coordinate with other government agencies on synthetic biology efforts, including participation in NSTC’s Interagency Synthetic Biology Working Group (SBWG).
- We participate in the EBRC, which, among other activities, develops research roadmaps to help overcome technical barriers in engineering biology.
- NIST collaborates with Manufacturing USA institutes, including BioMADE, NIIMBL, and BioFab USA, to advance biomanufacturing.
Safety and reliability
We host several consortia that provide insight into the needs of researchers, industry developers, and government regulators as they navigate the rapidly evolving biotechnology landscape.
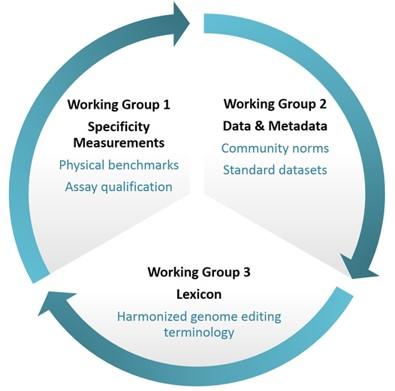
- The NIST Genome Editing Consortium develops tools to help ensure the accuracy of measurements for altered DNA, helping genetic engineers and regulators have confidence in new techniques. The consortium has completed a lexicon to facilitate common understanding for this rapidly evolving field.
- NIST is conducting an interlaboratory testing program for the measurement of DNA introduced into the genomes of target cells so that genetic engineers can quantify how well their techniques work.
- The Genome in a Bottle (GIAB) Consortium develops reference materials, methods, and data to increase confidence in DNA sequencing as a clinical tool. In addition, this consortium works to characterize the most challenging genome regions, long sequences with many repeats, to provide a reference for genes critical to human health and to advance understanding of the accuracy of genome engineering and synthetic biology.
Cyberbiosecurity
Data is the foundation for harnessing the genomes of organisms for making useful products, developing the complex manufacturing processes of the bioeconomy, and of new treatments for devastating diseases. Protecting that data is vital for guarding valuable intellectual property, preventing malicious harm to manufacturers and patients, and protecting citizens' privacy. NIST's researchers in cybersecurity and the biological sciences work together to assess and mitigate potential harms from the misappropriation and misuse of data.
Key Accomplishments
- NIST provides the most authoritative characterization of human genome references for use in benchmarking new methods of DNA analysis and technology development, optimization, and demonstration. These references have helped to advance global sequencing technology development and facilitated their use in emerging biotechnologies.
- NIST worked with more than 100 companies to characterize a monoclonal antibody reference material, known as the NISTmAb, that allows the biopharmaceutical industry to verify and improve their methods for quality control. Because its characteristics are so well known, the NISTmAb can also be used to evaluate new biomanufacturing methods and tools, accelerating innovation.
- NIST in collaboration with JCVI and MIT, developed a measurement approach to characterize genomically minimal cell models, a critical step toward the grand challenge of understanding the role of every gene in a cell, as well engineering synthetic cells that could function as “cell factories” to produce drugs, advanced materials, food, and fuels.
- NIST researchers engineered RNA circuits to act as biological computers to carry out a desired function. For example, RNA sensors embedded in cell factories offer the potential for nondestructive, real-time monitoring of cell function during the biomanufacturing process.