Summary
The global response to COVID-19 is leading to rapid advances in diagnostic, surveillance, and vaccine and therapeutic development. A critical measurement underpinning much of these efforts is a serological assay that helps to assess the complex patient responses to SARS-CoV-2. Specifically, reliable and rapid serological testing is needed to monitor the spread of the virus, patient immunity against the virus, and to determine the efficacy of vaccines and therapeutics. Yet, serology test results are highly variable for existing serologic tests, in part due to the lack of well characterized globally traceable reference materials needed for validation and assay control. Leveraging our Flow Cytometry Quantitation Consortium and state-of-the-art facilities, we have developed a rapid, multiplexed, and sensitive flow cytometry-based serological assay as well as a BSL-3 sparing neutralization assays (surrogate bead-based neutralization assay and pseudo-virus neutralization assays).
This work has supported the development of
- WHO/BS.2020.2403 WHO International Standard and Reference Panel for anti-SARS-CoV-2 antibody
- WHO/BS/2022.2427: 2nd WHO International Standard for anti-SARS-CoV-2 immunoglobulin and Reference Panel for antibodies to SARS-CoV-2 variants of concern
- Human SARS-CoV-2 Serology Standard developed by NIH/NCI Serology Science Network (SeroNet) and FNLCR.
These assays also enable the development of a robust measurement infrastructure for serological assays and associated critical reagents.
Description
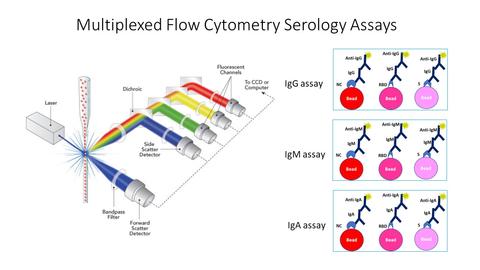
Multiplexed bead-based SARS-CoV-2 serological assay
We have developed a validated multiplexed bead-based SARS-CoV-2 serological assay that measures different antibodies produced by a patient (IgG, IgM, IgA). In collaboration with the NIST Applied and Computational Mathematics Division, we have developed a new strategy to classify more confidently the positive and negative samples. The validated assay was used to characterize the first and second WHO Serology Reference Panels through inter-laboratory studies as well as the study for the development of the US SARS-CoV-2 serology standard and a large-scale follow-up evaluation study led by NIH/NCI SeroNet and FNLCR.
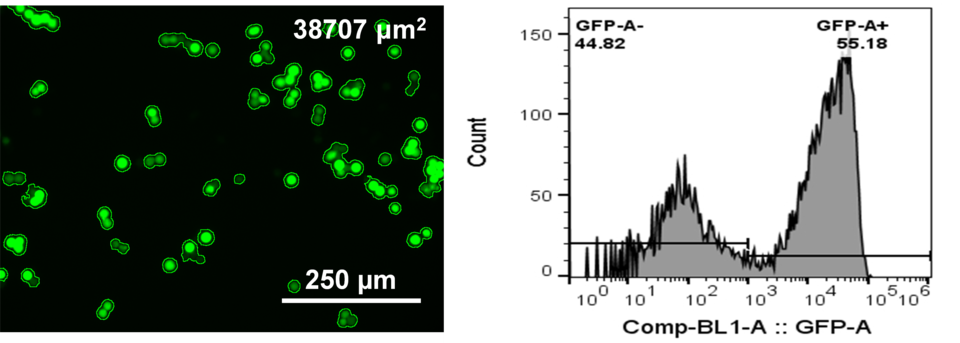
BSL-3 sparing neutralization assays
We have developed surrogate bead-based neutralization assay via flow cytometry and pseudo-virus neutralization assays via live cell imaging and flow cytometry to assist SARS-CoV-2 vaccine and therapeutic development. These assays will enable the functional assessment of the ability of serum antibodies to prevent cell binding, entry, or other effects of the virus on the cell in vitro. Data showed excellent correlation between our serology and bead-based neutralization assay results.
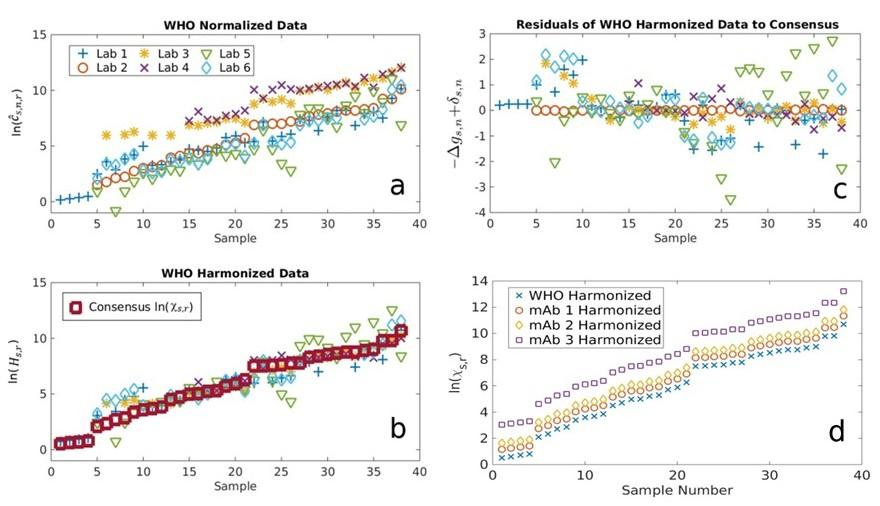
Interlaboratory study and reference materials to enable more quantitative and harmonized serology assays
NIST and CDC have led an effort collecting data through an interlaboratory study to address the following questions: 1) determine if a monoclonal antibody (mAb) can be used alone or with the WHO IS for assay harmonization and standardization, 2) understand the strengths and weaknesses of different neutralization assay formats, and 3) establish a more predictive method for correlates of protection. Using a new hierarchy of data analyses based on thermodynamics of antibody binding, we demonstrate that uncertainty reduction associated with harmonization can be achieved via a mAb. A key aspect of our new analysis method is a precise, mathematical definition of harmonization that separates uncertainty due to sample-assay and reference-assay effects, enabling the quantitation of, and thus the removal of, sources of uncertainty. Both the theory and experimental data confirmed that mAbs and WHO IS performed identically as a primary standard for establishing traceability and bridging different serology assay platforms using the new analysis method. A modeling framework is proposed that quantifies the probability of neutralization potential given a binding measurement. While approaches developed herein utilize COVID-19 as the use case, we expect the methods to be broadly applicable to other serology applications.