Summary
The COVID-19 pandemic highlighted the need for an agile and resilient pharmaceutical manufacturing and distribution framework. Advancements in manufacturing technologies can aid the move from few rigid centralized manufacturing facilities toward many agile distributed manufacturing and point-of-care (POC) manufacturing facilities to enable personalized and precision medicine. Recent NASEM reports (Innovations in Pharmaceutical Manufacturing on the Horizon) and FDA workshops for stakeholder engagement (FRAME) underline the emergence and criticality of this area. MMSD and NIST are developing the POC pharmaceutical manufacturing measurement science foundation, control strategies, and uncertainty analysis to support the production equipment and process analytical technology industries, as well as regulatory and other interested agencies.
Description

Demonstrative examples of manufactured pharmaceuticals at the point-of-care
We are employing quality by design (QbD) and risk assessment (cause-and-effect) approaches to investigate production technologies (e.g., drop on demand or inkjet printing platforms) and appropriate analytical control strategies for point-of-care manufacture of narrow therapeutic index drugs, medical countermeasure compounds, and biologics. The developed metrology for POC pharmaceutical manufacturing also aligns with efforts in nanomedicine to shift from stockpiling critical vaccines to a network of distributed vaccine manufacturing sites rapid response.
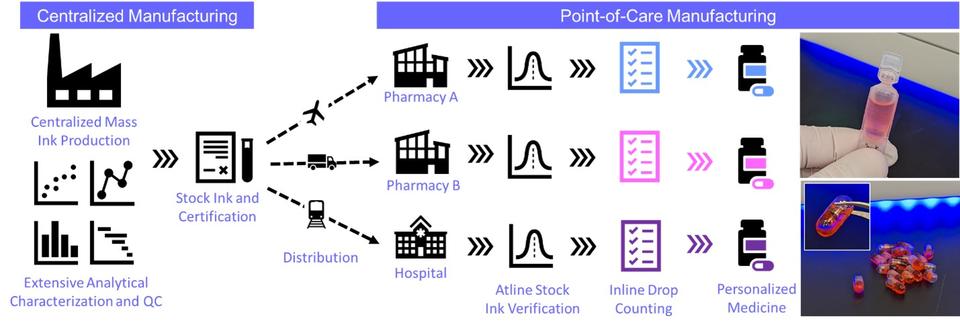
Point-of-Care Pharmaceutical Manufacturing Framework
Preliminary investigations consider drop on demand technologies and operate within a framework (Figure) that considers active pharmaceutical ingredient (API) 'ink' production at a traditional centralized manufacturing site with rigorous GMP standards and quality control. The certified API ink is distributed to POC production locations (e.g., hospitals, pharmacies, etc.) for personalized dose deposition and delivery (e.g., single-dose liquid vials, capsules, tablets, orodispersible films, etc.). Analytical capabilities across MMSD and NIST are employed to characterize dispensed mass/volume (gravimetry), amorphous and crystal polymorphs (SEM, THz Raman spectroscopy, and machine learning), chemical composition and quantification (mass spectrometry, UV-Vis and fluorescence spectroscopy), and 3D chemical distributions (ToF-SIMS depth profiling).
Quality by Design and Control Strategies
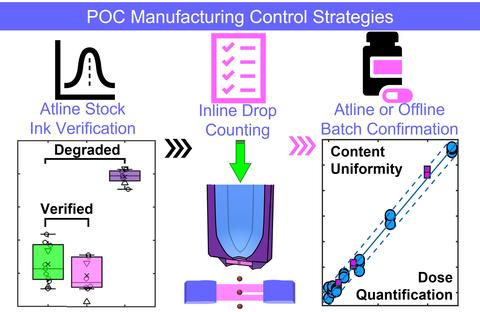
Quality by design principles provide a method to determine the main pharmaceutical unit operations for a given production and the critical production parameters and critical quality attributes. Risk assessment approaches, for example cause-and-effect analysis or Ishikawa diagrams, can aid in establishing the main areas of risk or sources of variability in the manufacturing process or end product. Critical process parameters and quality attributes exhibiting higher risk or variability require a higher level of built in control in the form of inline, atline, or offline control or verification strategies.
Control strategies for our initial framework includes atline ink verification by non-contact spectroscopic techniques, inline and in-process drop counting during the dispensing, and atline or offline batch quality and quantity confirmation by spectroscopy or spectrometry. Advances in the internet of things and machine learning approaches also introduce avenues for interconnectivity between POC facilities and quality control.
The POC Pharmaceutical Manufacturing and Precision Medicine Program also overlaps with and extends into related focus areas in MMSD such as POC diagnostics in cooperation with POC drug-delivery, public and health worker safety from toxic drug (e.g., chemotherapy agents) trace contamination, and visualization of drug delivery aerosols.
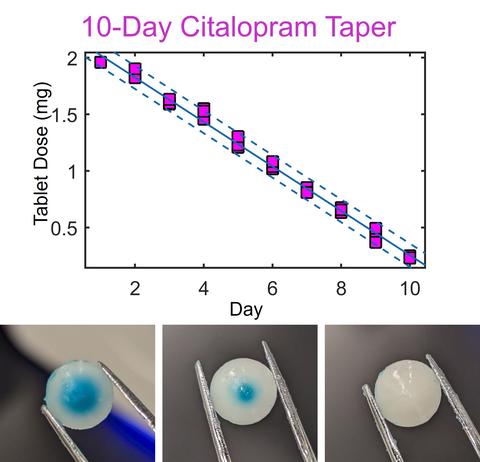
Personalized Tablet Dosing
POC pharmaceutical manufacturing also enables customizable and personalized dosages or dosage regimes. This adaptability provides an avenue for physicians to prescribe specific drug tapers when taking patients off antidepressants (e.g., selective serotonin reuptake inhibitors [SSRI]) or painkillers (e.g., opioids). By incorporating semisolid excipients with low melting points, produced tablets are liquid as active pharmaceutical ingredients are deposited and encapsulated by the material.
National Research Council (NRC) Postdoctoral Program Research Opportunities:
- Point-of-Care Pharmaceutical Manufacturing for Precision Medicine
- Mass Spectrometry Metrology for Trace Detection and Chemical Imaging
NIST Highlights:
Point-of-care medicine showcased by the NIST Taking Measure Blog: Your Future Medications Could Be Personalized for You on a 3D Printer and research by undergraduate SURF fellows: Spotlight: SURF Student Zainab Altamimi Spends Her Summer Researching the Capsules and Tablets in 3D Drug Printing

