Infrared studies for atmospheric atlases
The infrared studies at NIST have been dedicated since the 1950's to the study of atmospheric molecules both terrestrial and those found in the planets and their satellites. The data has been included in the HITRAN tables which is an atlas of the ro-vibrational spectra of these molecular species. The effort has been divided into two parts: high resolution studies, collision induced absorption and the water vapor continuum and performed with the aid of many collaborators.
High resolution studies:
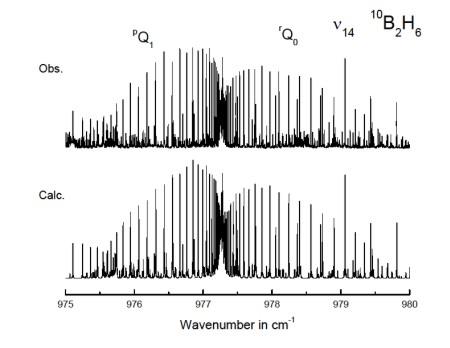
Recent studies include line frequencies and line intensities of ClONO2 for stratosphere, B2H6 for outer planets, SO2 for troposphere, propane & propene for troposphere and outer planet atmospheres, HOCl for stratosphere, near IR bands of O2 for heat balance data, and ClO2 for stratospheric monitoring. Most of these studies have a spectral resolution of 0.003 cm-1 or better.
Why? If frequencies and line intensities of a band are measured at a given temperature, the rotational constants and transition moments extracted can be used to calculate the band spectrum accurately over the range of atmospheric temperatures or the data can be used to estimate the temperature and pressures involved.
Line shift and pressure broadening coefficients of these molecules are still badly needed.
Continuum studies:
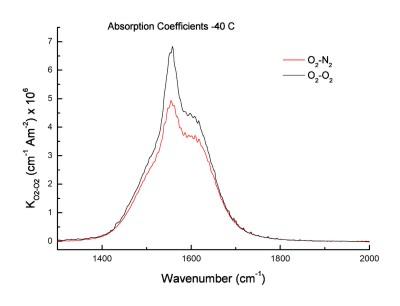
A small dipole moment change is generated during a molecular collision. This leads to very weak absorbance from homonuclear molecules such as the fundamental bands of N2, O2 and the gerade bands of CO2. This is called collision-induced absorption (CIA). Even though they are weak, the N2 and O2 absorptions play an important role in the radiative processes of the atmosphere. They can be studied in the lab with long paths 60 meters and moderately high pressures (5 atm).
The H2O continuum is one of the least understood and poorly measured phenomena in molecular spectroscopy and yet it plays an important role in the atmospheric energy balance. It has been attributed to underlying dimers or to line wing effects.
Room temperature collision-induced spectra (CIA) of O2 and N2 are somewhat better understood, but theoretical predictions of their temperature dependence are incorrect.
Related Links:
- Molecular spectroscopy
- Large-amplitude motion
- Matrix isolation infrared spectroscopy
- Vibrational and electronic energy levels of polyatomic transient molecules
- Spectroscopic identification of interstellar molecules
- High resolution infrared and Raman studies of atmospheric-related and highly strained molecules
Contacts
-
SSDWEB(301) 975-3727100 Bureau Drive
M/S 8440
Gaithersburg, MD 20899-8440

