High resolution infrared and Raman studies of atmospheric-related and highly strained molecules
The focus of these investigations is molecules of environmental importance and those that pose special problems of chemical bonding. The experimental approach is high-resolution non-linear Raman spectroscopy, especially in its CARS configuration, and high-resolution infrared Fourier transform spectroscopy. Ab initio quantum theoretical calculations are done along with the experimental investigations. The project is a joint undertaking with the Raman group at the Oregon State University in Corvallis, Oregon, and the infrared group at the Pacific Northwest National Laboratory in Richland, Washington.
Investigations of Sulfur Trioxide
Recent work has focused on the spectroscopy of sulfur trioxide molecule, a molecule that plays a significant role in the chemistry of the earth's upper atmosphere. High-resolution coherent anti-Stokes Raman spectra (CARS) of SO3 revealed significant perturbations of the totally-symmetric, fundamental S-O stretching mode of vibration. The successful analysis of these perturbations required the study of infrared transitions to vibration-rotational levels that lie close to those of the fundamental mode of the four symmetric top rotator isotopomers 32S16O3, 34S16O3, 32S18O3, and 34S18O3, as well as ab initio quantum theoretical calculations.
Investigations of Highly Strained Molecules
Propellane and bicyclopentane
In recent decades, organic chemists have been very successful in creating interesting strained ring compounds, many of which were thought previously not being capable of existence. Among these is the [1.1.1]propellane molecule with its novel "inverted" bond that joins the two carbon atoms at the apices of a triangular bipyramid. This "inversion" is in contrast to the expected tetrahedral bonding to the two hydrogen atoms attached to the apical carbons as shown for the bicyclo[1.1.1]pentane molecule. The height of the bipyramid of propellane formed by the carbon atoms is significantly less than the corresponding height in the bicyclopentane molecule, which is evidence that the bonding in propellane is highly strained. Investigations of the high resolution infrared and CARS spectra of propellane and bicyclopentane are ongoing, and are conducted in parallel with ab initio quantum theoretical calculations. Both molecules are symmetric top rotators and posses the symmetry of the D3h point group.
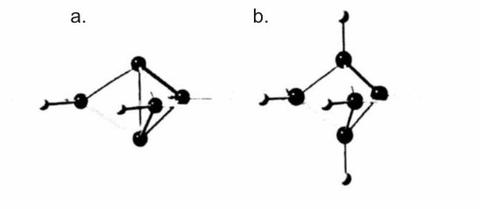
Spiropentane
Another molecule with strange bonding is spiropentane. Although originally synthesized around the turn of the 20th Century this molecule has only recently come into the purview of molecular spectroscopists. Spiropentane was recently successfully synthesized at Oregon State University and is currently the subject of high-resolution spectroscopic investigation.
Recent publications
"Analysis of High-Resolution Infrared and CARS Spectra of 32S18O3" T. Masiello, N. Vulpanovici, J. Barber, E.t.H. Chrysostom, J.W. Nibler, A. Maki, Th.A. Blake, R.L. Sams, and A. Weber J. Molec.Spectrosc. 227, 50-59 (2004).
"High-Resolution Infrared Spectroscopy of [1.1.1]propellane" R. Kirkpatrick. T. Masiello, N. Jariyasopit, A. Weber, J.W. Nibler, A. Maki, Th.A. Blake, and T. Hubler J. Molec. Spectrosc. 248, 153-150 (2008).
"High-Resolution Rovibrational Study of the Coriolis-coupled ν12 and ν15 Modes of [1.1.1]propellane" R. Kirkpatrick, T. Masiello, N. Jariyasopit,. J.W. Nibler, A. Maki, Th.A. Blake, and A. Weber J. Molec. Spectrosc. 253, 41-50 (2009).
A. Weber and D.R. Lide, Jr., "Molecular Spectroscopy," in Encyclopedia of Physics, 3rd ed., R.G. Lerner and G.L. Trigg, Eds., (Wiley-VCH, Weinheirn, 2005), vol. 2, pp. 1522-1600.

