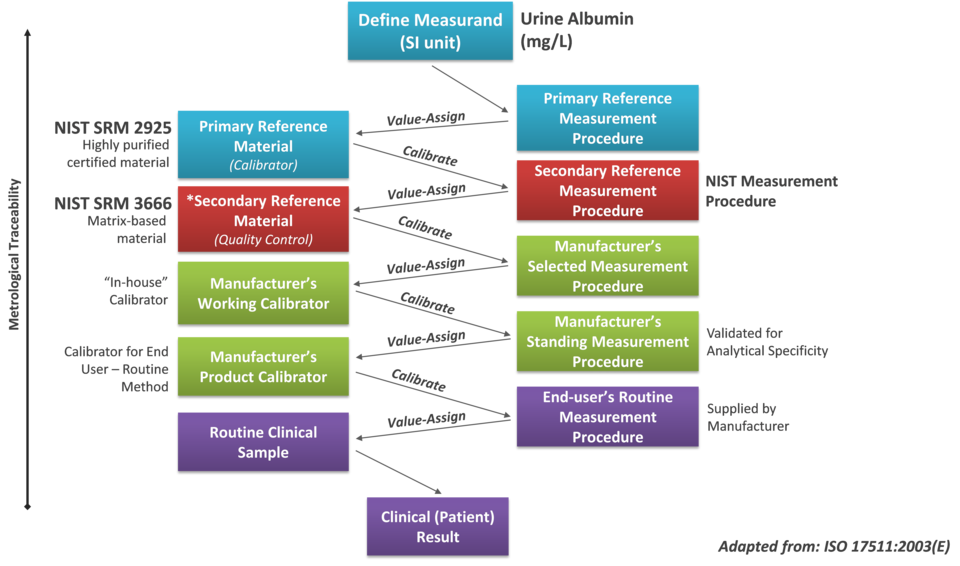
Summary
The NIST Urine Albumin Standardization Program focuses on developing higher-order reference materials and measurement procedures to establish a global traceability framework, ensuring that clinical results for kidney disease are accurate, consistent, and comparable.
Description
Kidney Disease – A Global Concern
Kidney disease is a significant global public health and economic issue, with Chronic Kidney Disease (CKD) ranking among the top 10 worldwide causes of death. The critical need for early and accurate diagnosis, effective treatment management, and ongoing patient care relies heavily on the reliable detection of key biomarkers. A major diagnostic and prognostic biomarker for kidney disease is urine albumin. The accuracy and consistency of clinical laboratory results for urine albumin are essential for healthcare practitioners to make reliable patient care decisions.
The Need for Standardization
Patient care decisions, made by healthcare practitioners for kidney disease, are influenced by the validity of clinical laboratory results. To ensure clinical results are accurate, precise, and globally comparable – independent of the specific testing method or laboratory – a robust system of standardization is required. This involves establishing a traceability framework of reference materials and measurement procedures that link clinical results to the fundamental units of measurement (SI units).
NIST's Standardization Initiative
To enhance the accuracy and global comparability of clinical urine albumin results, the National Institute of Standards and Technology (NIST) has partnered with The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) Laboratory Working Group (LWG) and The International Federation of Clinical Chemistry (IFCC) Working Group for the Standardization of Albumin Assays in Urine (WG-SAU) to establish a reference measurement system for urine albumin. NIST is leading the effort to develop the foundational components of the urine albumin reference measurement system: a series of higher-order certified reference materials and a reference measurement procedure. This work lays the foundation for building global confidence in clinical results for kidney disease diagnosis and management.
Major Accomplishments
Department of Commerce Gold Medal Award - Development of a groundbreaking standard reference material for kidney disease, SRM 3666 Albumin and Creatinine in Frozen Human Urine.
Publication - Commutability Assessment of NIST SRM 3666 Albumin and Creatinine in Frozen Human Urine Among Routine Clinical Laboratory Measurement Procedures (Link); Commutability assessment of SRM 3666 for use as a secondary measurement standard in the calibration hierarchy for urine albumin and creatinine for select in vitro diagnostic-measurement procedures (IVD-MPs).
NIST Reference Measurement Procedure - Listed in JCTLM Database for RMPs (NIST RMP - C21RMP9) - Multiplexed ID-LC-MS/MS method for the absolute quantification of urine albumin.
Release of NIST SRM 3666: Albumin and Creatinine in Frozen Human Urine (NIST SRM 3666; NIST Special Publication 260-238-upd4) - Human urine-based secondary reference material intended for use in a calibration hierarchy, in the validation of measurement procedures, or as quality control material for the determination of albumin, creatinine, or the albumin‑to‑creatinine ratio (ACR).
Publication - Estimation of Measurement Uncertainty for the Quantification of Protein by ID-LC-MS/MS (Link); First publication that provides a detailed framework on how to determine measurement uncertainty of a quantitative MS-based protein measurement procedure
NIST SRM 2925 - Listed in the Joint Committee for Traceability in Laboratory Medicine (JCTLM) Database for Reference Materials, an international database of higher-order RMs for clinical applications (SRM 2925 - C18RM1).
Release of NIST SRM 2925: Recombinant Human Serum Albumin Solution (Primary Reference Calibrator for Urine Albumin) (Frozen) (NIST SRM 2925; NIST Special Publication 260-199); Calibration tool for clinical laboratories and in vitro diagnostic manufacturers
Project Spotlights
- Medscape Medical News Article: New NIST Standard Will Improve Diagnosis of Kidney Disease (Link)
- NIST News: NIST Develops Human Urine Standard for More Accurate Diagnosis of Kidney Disease (Link)
- NIST Spotlight: Screening for Kidney Disease With Accurate Measurements (Link)
Publications
- Beasley-Green A, et al. (2025), "Commutability Assessment of NIST SRM 3666 Albumin and Creatinine in Frozen Human Urine Among Routine Clinical Laboratory Measurement Procedures". Clin Chim Acta. (Link)
- Beasley-Green A, Wood ESC, Camara JE, Heckert NA. (2025), "Extent-of-Equivalence Assessment of Certified Reference Materials for Albumin and Creatinine in Human Urine". NIST Special Publication 260-262. (Link)
- Beasley-Green A, Heckert NA. (2024), "Reference Measurement Procedure for the Absolute Quantification of Albumin in Urine Using Isotope Dilution-Liquid Chromatography-Tandem Mass Spectrometry (ID-LC-MS/MS)". NIST Special Publication 1200-31-upd1. (Link)
- Beasley-Green A, Camara J, Heckert NA. (2023), “Certification of Standard Reference Material 3666 Albumin and Creatinine in Frozen Human Urine”. NIST Special Publication 260-238-upd2. (Link)
- Beasley-Green A, Heckert NA. (2023), “Estimation of Measurement Uncertainty for the Quantification of Protein by LC-MS/MS”. Anal Bioanal Chem, 415:3265-3274. (Link)
- Beasley-Green A, Bunk DM, Alejo W, and Zhang NF. (2020), “Certification of Standard Reference Material 2925 Recombinant Human Serum Albumin Solution (Primary Reference Calibrator for Urine Albumin) (Frozen)”. NIST Special Publication 260-199. (Link)
- Beasley-Green A., Burris NM, Bunk DM, Phinney K. (2014), “Multiplexed LC-MS/MS Assay for Urine Albumin.” J Proteome Res, 13(9),earch, 13(9): 3930-3939. (Link)
Presentations
View the list of presentations.
National Research Council Post-Doctoral Research Opportunity: Qualitative and Quantitative Characterization of Clinical Analytes (Link)

