Summary
All inorganic materials contain point defects in the form of impurities, interstitials, or vacancies. Common imaging techniques fail to capture these defects because they typically substitute into a crystal lattice at parts per million concentrations, well below practical detection limits. It is important to be able to effectively characterize the concentration and nature (valence state, site of substitution, local structure, etc.) of point defects because they often play a primary role in determining the functional properties of a material. Traditional techniques that are used to measure point defects (electron diffraction and absorption) are best suited for high defect concentrations (typically 1 mol% or greater). Trace concentrations (less the 1 mol%) are always present in materials and can be introduced by intrinsic or extrinsic mechanisms. Electron paramagnetic resonance (EPR) spectroscopy is a characterization tool best suited for trace defect concentrations. In the NIST Materials Structure and Data Group, the measurement of point defect chemistry in complex oxides is being advanced by the development of new EPR characterization techniques, and important connections are made between these measurements and complementary advancements in detection of dilute point defects analyzed using electron microscopy and x-ray absorption.
Description
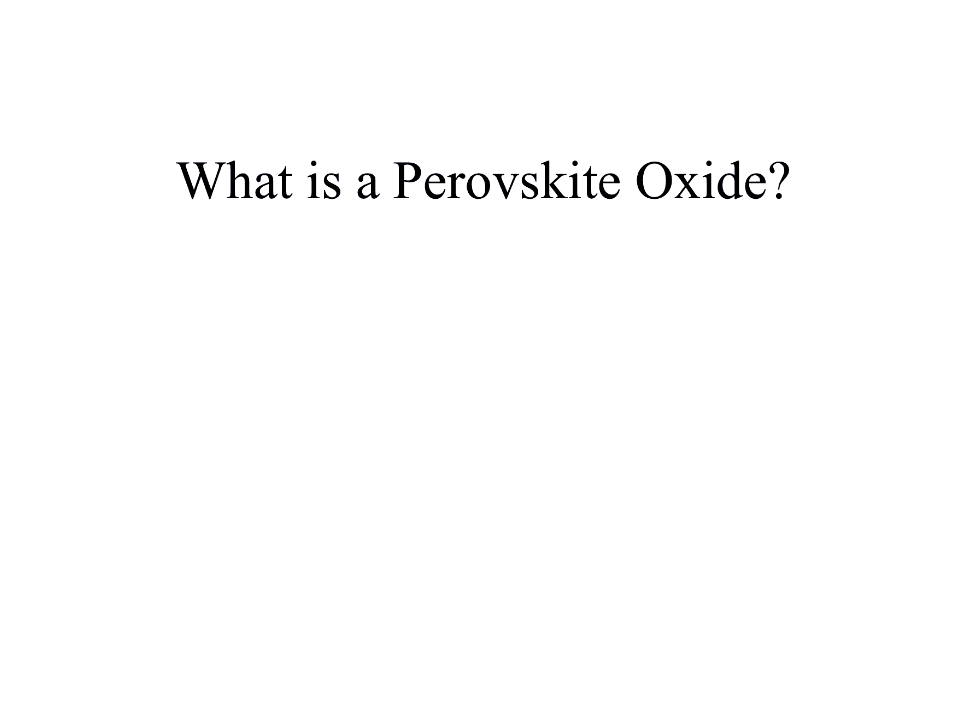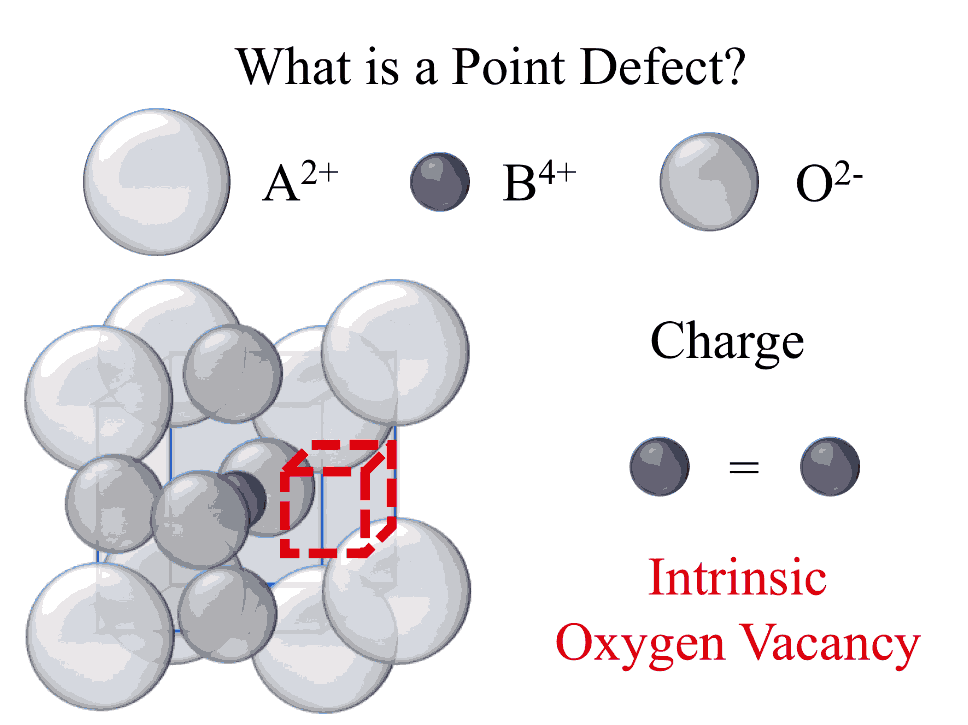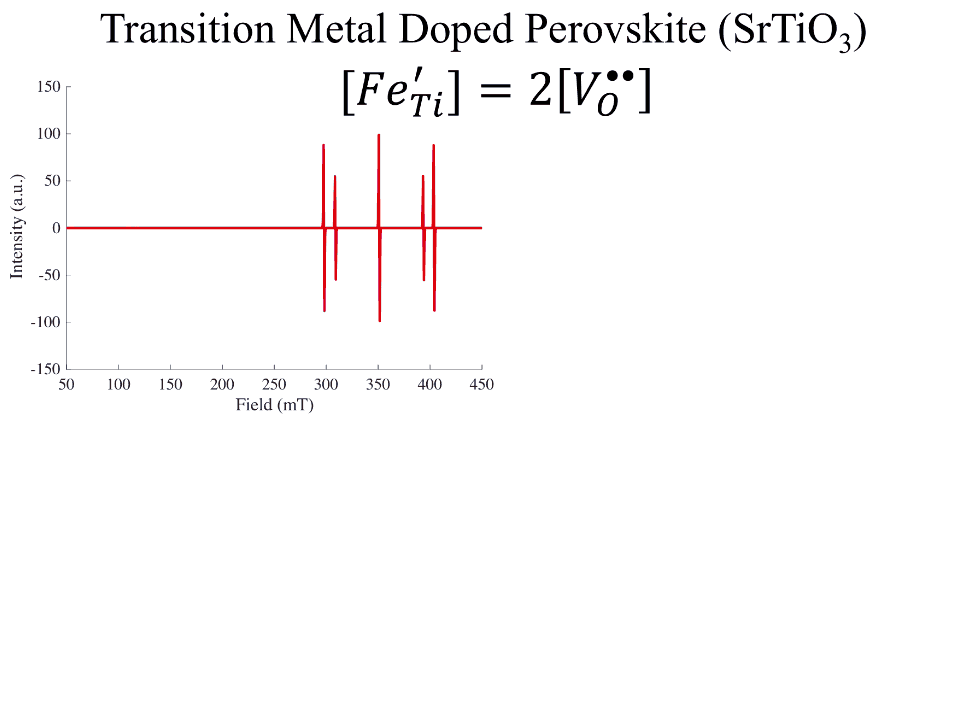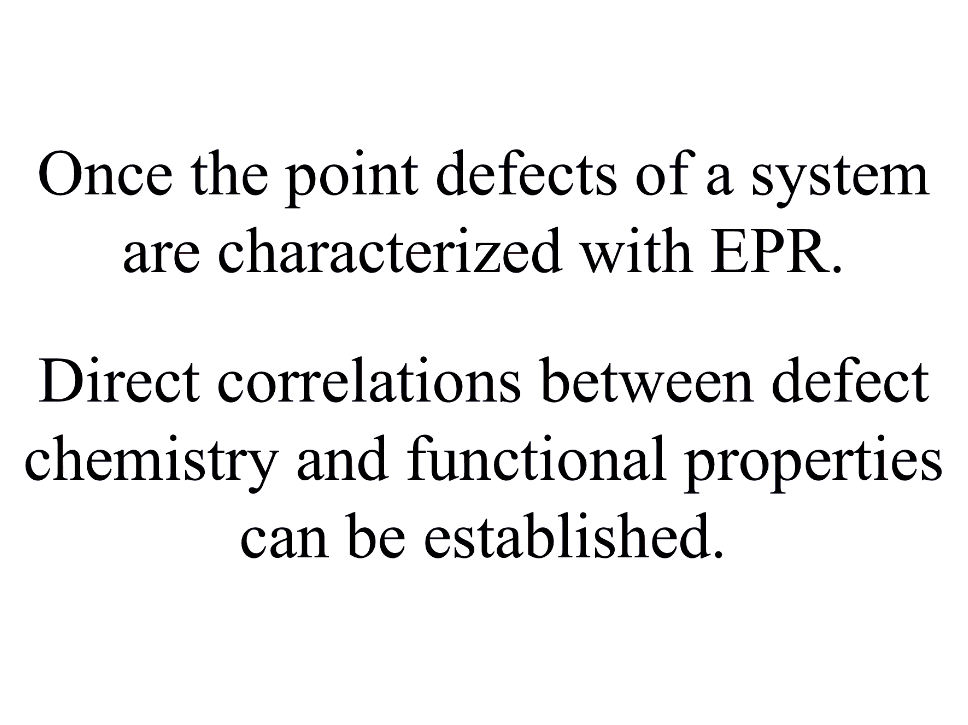
Project Goal: To develop magnetic resonance, x-ray absorption, electron diffraction, and electrical conductivity measurements to better characterize dilute concentrations of point defects in oxide materials and effectively correlate electro-mechanical properties to measured defect chemistry.
Oxide materials are used in diverse applications that include random access memory, solid oxide fuel cells, solid state refrigeration, and micro-actuators to name only a few. Because these applications require an electric bias or mechanical stimulus, ion diffusion is a prevalent process (typically in the form of vacancy diffusion). Fast-ion diffusion can be a desired or engineered phenomenon in the case of oxide fuel cells or oxygen sensors, or ion diffusion can result in local changes to composition resulting in a negative effect on performance and reliability over time. Accurate measurement of the mobility and local interaction of ionic charge carriers in a given system is dependent on effective analysis of the underlying defect chemistry. Better measurement of defect chemistry will facilitate advanced prediction of properties and help design improved materials for a given application.
Major Accomplishments
The Materials Measurement Laboratory (MML) and the Center for Nanoscale Science and Technology (CNST) staff published the first experimental evidence of the ability to control the valence and location of a transition metal ion, incorporated as a dopant in a perovskite oxide, by finely tuning the stoichiometry of the host material (10.1002/adfm.201602156). The electronic properties of wide-band gap perovskite oxides, which find applications as dielectrics, piezoelectrics, ionic conductors, etc., can be critically influenced by small concentrations of impurities. Common impurities include naturally-abundant transition-metal and alkali ions that are often found at the parts per million level in the raw materials used to synthesize functional oxides. The electrical neutrality condition dictates that the unbalanced charge, introduced by these ions, must be compensated by other charge carriers, such as electrons, holes, or charged vacancies. These extrinsic charge compensation mechanisms have a dominant impact on the functional properties of the material.
The behavior of transition-metal dopants in perovskites is particularly difficult to predict because these ions can assume multiple oxidation states (number of electrons gained by a metal atom chemically bonded to oxygen), which affects their radii and location in the structure. The type of lattice site occupied by the dopant and its oxidation state determine whether the material is a dielectric, an n- or p-type semi-conductor, an ionic conductor, or an electronically conductive material.
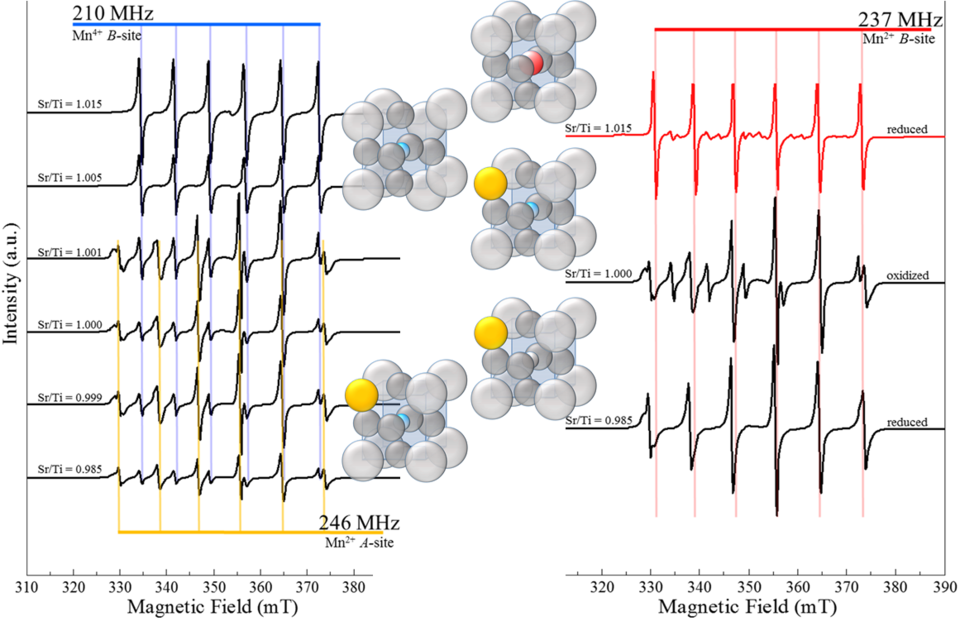
MML and CNST researchers synthesized a carefully designed set of prototypical perovskite samples. Experiment and theory were combined using electron paramagnetic resonance (EPR), x-ray absorption spectroscopy (XAS), and density functional theory (DFT) calculations to improve the field of point defect metrology (10.1021/acs.chemmater.0c01082) by demonstrating the factors that limit the efficacy of individual characterization techniques. Relationships between dopant substitution mechanisms and crystal chemistry/structure were studied (10.2139/ssrn.3606819) using these NIST developed techniques – providing a roadmap for future advancements in point defect characterization and material design.
(Magic Dopant) R.A. Maier, A-C. Johnston-Peck, and M.P. Donohue, Adv. Fun. Mat., DOI: 10.1002/adfm.201602156 (2016).
(Low Temperature Conductivity I) R.A. Maier and C.A. Randall, “Low-Temperature Ionic Conductivity of an Acceptor-Doped Perovskite: I. Impedance of Single-Crystal SrTiO3,” J. Am. Ceram. Soc., 99 [10] 3350–3359 (2016).
(Low Temperature Conductivity II) R.A. Maier and C.A. Randall, “Low Temperature Ionic Conductivity of an Acceptor-Doped Perovskite: II. Impedance of Single-Crystal BaTiO3,” J. Am. Ceram. Soc., 99 [10] 3360–3366 (2016).
(Defect Dipole) R.A. Maier, T.A. Pomorski, P.M. Lenahan, and C.A. Randall, “Acceptor-Oxygen Vacancy Defect Dipoles and Fully Coordinated Defect Centers in a Ferroelectric Perovskite Lattice : Electron Paramagnetic Resonance Analysis of Mn2+ in Single Crystal BaTiO3,” J. Appl. Phys., 118 164102 (2015).
R. A. Maier, E. Cockayne, M. Donohue, G. Cibin, and I. Levin, “Substitutional Mechanisms and Structural Relaxations for Manganese in SrTiO3: Bridging the Concentration Gap for Point-Defect Metrology,” Chem. Mater., 32, 11, 4651–4662 (2020).
C. Sims, R. A. Maier, A. C. Johnston-Peck, J. M. Gorham, V. A. Hackley, B. C. Nelson, “Approaches for the quantitative analysis of oxidation state in cerium oxide nanomaterials,” Nanotechnology 30 (8), 085703 (2019).
Russell A. Maier, K. F. Garrity, M. P. Donohue, A. Ozarowski, and I. Levin, “Effects of Octahedral Tilting on the Site of Substitution of Manganese in CaTiO3,” A. Mater., 10.2139/ssrn.3606819 (2020).

