Summary
Because of NIST's role as the National Metrology Institute (NMI) in the U.S., the Chemical Sciences Division's measurement capabilities must be benchmarked against the capabilities of NMIs worldwide as required by a Mutual Recognition Arrangement (MRA) established in 1999. The MRA provides a framework for obtaining reliable quantitative information on the comparability of measurement services provided by all NMIs, thereby providing governments and other parties with a technical foundation for wider agreements related to international trade, commerce, and regulatory affairs. The Chemical Sciences Division actively participates in the International Committee of Weights and Measures (CIPM) Consultative Committee for Amount of Substance – Metrology in Chemistry and Biology (CCQM) comparisons, which address chemical and biological measurement-related issues important for international trade, environmental, health, and safety-related decision making. We have also established a limited number of strategic bilateral collaborations and/or intercomparisons with NMIs around the world to demonstrate comparability of measurements and/or to cooperate in the development of reference materials.
Description
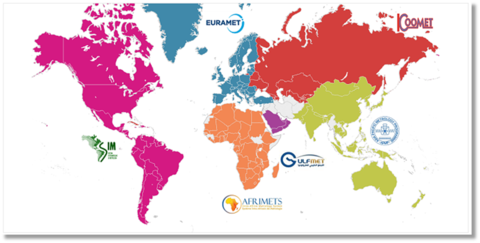
The CCQM is a committee of the International Bureau of Weights and Measures (BIPM). Six regional metrology organizations exist as part of this framework; the US belongs to the Inter-American Metrology System (SIM).
International agreements and decisions concerning trade and our social well-being increasingly require mutual recognition of measurements and tests among nations. The Mutual Recognition Arrangement (MRA) on national measurement standards and calibration and measurement certificates issued by NMIs rests upon the assumption of equivalence of national measurement standards in each country. In October 1999, the directors of NMIs for its original 38 member states of the Meter Convention and two international organizations signed the MRA; nearly 100 countries and associates now have signed the MRA. The MRA provides an open, transparent, and comprehensive framework for obtaining reliable quantitative information on the comparability of measurement services provided by the signatory NMIs.
Signatories to this MRA have the following responsibilities: (1) participate in relevant international comparisons, known as key comparisons, (2) participate in any relevant supplementary international comparisons of measurements, and (3) document the existence of a quality system and demonstrations of competence for its measurement services to customers. The expected outcome from these activities is to provide statements for each signatory of its calibration and measurement capabilities (CMCs) in a publically-available database. NIST has taken a leadership role in the CCQM and in the Chemical Metrology Working Group of the Interamerican System for Metrology (SIM) to assure the equitable and metrologically sound implementation of this MRA.
The CCQM currently has ten working groups: (1) Inorganic Analysis, (2) Electrochemical Analysis, (3) Gas Analysis, (4) Organic Analysis, (5) Surface Analysis (6) Protein Analysis, (7) Cell Analysis, (8) Nucleic acid Analysis, (9) Key Comparison and CMC Quality, and (10) Strategic Planning. These working groups are responsible for selecting and overseeing the operation of key comparisons, in which participating laboratories demonstrate their measurement or material preparation capabilities. Staff members of the Chemical Sciences Division (CSD) are involved in the various activities within several working groups. Since its inception over 20 years ago, over 150 key comparison studies and a similar number of pilot studies have been conducted under the auspices of the CCQM. CSD has participated in a large majority of these key comparisons, serving as the coordinating laboratory in nearly half of these key and pilot studies.
technical details
Participation in CCQM key comparisons is generally limited to NMIs with well-established chemical metrology programs. Within SIM, several NMIs have well-established programs; however, many NMIs are still developing their capabilities. To effectively address the unique needs of all 32 countries within SIM, whose capabilities in chemical metrology span a very broad range, the SIM program has focused on training and capability assessment rather than on participation in MRA-driven key and supplemental comparisons. The Chemical Sciences Division has served as host to numerous guest scientists from NMIs across SIM NMIs for periods of several months to a year. CSD has provided training in all aspects of chemical metrology, ranging from the preparation of primary gas mixtures to the development of trace-level elemental detection methods to the accurate measurement of vitamin metabolite levels in human blood samples. As a more recent example, CSD is providing SIM NMIs with in-depth classroom and hands-on laboratory training at NIST in the form of 1-week course for implementation of isotope dilution/mass spectrometry methods in the application of clinical marker measurements.
In addition to these regional activities, we have also established several strategic bilateral collaborations and intercomparisons with NMIs around the world. For example, since 1992 we have collaborated with the Netherlands Measurement Institute (NMi) to determine the equivalence of primary gas standards, resulting in a formal "Declaration of Equivalence" that is recognized by the U.S. EPA and European environmental regulatory bodies as documenting the equivalence between numerous NIST and NMi primary gas mixture suites. For nearly a decade, the CSD has advanced a partnership with the National Institute of Metrology, Standardization and Industrial Quality (INMETRO), the National Metrology Institute of Brazil. More recently, NIST and INMETRO signed a Cooperative Research Agreement to exchange technical information and capabilities related to chemical and physical measurement sciences, alternative energy sources, and measurement standards, including reference data and materials. In July 2013, a renewed agreement was also signed between NIST and the European Commission’s Joint Research Centre (JRC) Institute for Reference Materials and Measurements (IRMM) in Geel, Belgium to collaborate and coordinate with their research and development programs in metrology. This agreement includes cooperative research on modern measurements and their quality assurance and technical cooperation in the preparation and characterization of certified reference materials that span broad measurement areas, such as healthcare, food safety, environmental protection and nanotechnology.
Associated Publications
1. Wang, J., Chao, J., Wei, C., Li, H., Wang, Q., Song, P., Lu, H., Zhou, Y., Tang, Y., Wang, S., Yang, L., Nadeau, K., Pihillagawa, I. G., Johnson, M. E., Yu, L. L., Naykki, T., Sara-Aho, T., Zambra, R. P., Napoli, R., Rienitz, O., Noordmann, J., Pape, C., Towara, J., Tsz-Chun, C., Hei-Shing, C., Stakheev, A., Dobrovolskiy, V., Stolboushkina, T., Glinkova, A., Taebunpakul, S., Thiengmanee, U., Kaewkhomdee, N., Uribe, C., Carrasco, E., Botha, A., Fisicaro, P., Oster, C., Ahumada, D. A., Abella, J. P., Segura, C. S., Shin, R., Deborah, S. L. P., Dewi, F., Kiat, B. T. M., Zongrong, W. Y., Wah, L. H., Haraldsson, C., Merrick, J., Antin, L., White, I., Goenaga-Infante, H., Hill, S., Entwisle, J., Jacimovic, R., Zuliani, T., Fajon, V., Yim, Y. H., Heo, S. W., Lee, K. S., Lee, J. W., Lim, Y., Okumu, T. O., Ndege, M., Wangui, L., Can, S. Z., Coskun, F. G., Tunc, M., Giannikopoulou, P., Kakoulides, E., Inagaki, K., Miyashita, S., Klich, H., Jebali, R., Chaaban, N., Bergamaschi, L., Sobina, E., Tabatchikova, T., and Migal, P., "Final report of the CCQM-K145: toxic and essential elements in bovine liver," Metrologia, 57, (2020).
2. Waters, J. F., Mariassy, M., Bastkowski, F., Asakai, T., Prokunin, S. V., Gonzaga, F. B., Dimitrova, L., Amaya, R. C., Quezada, H. T., Vospelova, A., Rozikova, M., Snedden, A., Stoica, D., Sander, B., Jakusovszky, B., Maksimov, I., Onuma, S., Suzuki, T., Ruiz, J. M., Ortiz-Aparicio, J. L., Tincona, G., Vasquez, J., Pawlina, M., Kozlowski, W., Dobrovolskiy, V. I., Aprelev, A. V., Hankova, Z., Rodruangthum, P., Uysal, E., Liv, L., Nakiboglu, N., Gavrilkin, V., Petrenko, A., Manska, O., Easley, R. A., and Fajardo, S., "Key comparison CCQM-K18.2016: pH of carbonate buffer," Metrologia, 57, (2020).
3. Arzate, C. M. S., Burdette, C. Q., Aragon, H. G., Lippa, K. A., Osuna, M. A., Contreras, L. R., Lee, J., Lee, S., Baek, S. Y., Kim, B., Huang, T., Guo, Z., Li, H. M., Zhang, Q. H., Phillips, M. M., Toman, B., and Duewer, D. L., "CCQM-K147 Comparison of value-assigned CRMs for niacin (vitamin B-3) in milk powder and infant formula matrices," Metrologia, 56, (2019).
4. Duewer, D. L., Murray, J. A., Wood, L. J., Wise, S. A., Sander, L. C., Lippa, K., Hein, S., Koch, M., Philipp, R., Werneburg, M., Hackenberg, R., Polzer, J., Avila, M. A., Serrano, V., Kakoulides, E., Alexopoulos, C., Giannikopoulou, P., Gui, E. M., Lu, T., Teo, T. L., Hua, T., Chen, D. Z., Li, C. X., Ye, C. J., Li, H. M., Nammoonnoy, J., Quinn, L., Swiegelaar, C., Fernandes-Whaley, M., Goren, A. C., and Gokcen, T., "CCQM-K95.1 Low-polarity analytes in a botanical matrix: polycyclic aromatic hydrocarbons (PAHs) in tea," Metrologia, 56, (2019).
5. Duewer, D. L., Sander, L. C., Wise, S. A., Lippa, K., Philipp, R., Hein, S., Hackenberg, R., Polzer, J., Avila, M. A., Serrano, V., Kakoulides, E., Alexopoulos, C., Giannikopoulou, P., Chan, P. K., Lee, H. M., Tang, H. S., Tang, P. O., Yip, Y. C., Lu, T., Cheow, P. S., Teo, T. L., do Rego, E. C. P., Violante, F. G. M., de Carvalho, L. J., Leal, R. V. P., Sega, M., Rolle, F., Baek, S. Y., Kim, B., Lee, S., Cabillic, J., Fallot, C., Hua, T., Chen, D. Z., Ye, C. J., Li, C. X., Li, H. M., Itoh, N., Quinn, L., Prevoo-Franzsen, D., Fernandes-Whaley, M., Goren, A. C., Gokcen, T., Gunduz, S., Krylov, A., Mikheeva, A., Baldan, A., van der Hout, J. W., and van der Veen, A. M. H., "CCQM-K131 Low-polarity analytes in a multicomponent organic solution: polycyclic aromatic hydrocarbons (PAHs) in acetonitrile," Metrologia, 56, (2019).
6. Liandi, M. A., Bing, W. U., Ortiz-Aparicio, J. L., Manzano, J. V. L., Kozlowski, W., Dumanska, J., Pawlina, M., Pietrzak, A., Forero, A. M. C., Quesada, H. T., Sanchez, C. A. E., Amaya, R. C., Bohorquez, A. F. P., Sobral, S. P., Borges, P. P., Hatamleh, N., Puelles, M., Iglesias, A., Lozano, H., Jo, K., Hwang, E., Waters, J. F., Asakai, T., Mariassy, M., Shimolin, A., Sobina, A., Prokunin, S., Morozov, I. V., Dobrovolskiy, V. I., and Aprelev, A. V., "Assay of potassium hydrogen phthalate (CCQM-K34.2016)," Metrologia, 56, (2019).
7. Lippa, K. A., Duewer, D. L., Nelson, M. A., Davies, S. R., and Mackay, L. G., "The role of the CCQM OAWG in providing SI traceable calibrators for organic chemical measurements," Accreditation and Quality Assurance, 24, 407-415 (2019).
8. Liu, Q. D., Liu, H., Chen, Y. Z., Yong, S. R., Teo, H. L., Wong, L. K., Teo, T. L., Vamathevan, V., do Rego, E. C. P., Wollinger, W., Fernandes, J. L. N., Monteiro, T. M., Garrido, B. C., Violante, F. M., Shi, L. H., He, H. H., Quan, C., Xu, B., Li, H. M., Dai, X. H., He, Y. J., Lo, M. F., Yip, Y. C., Cabillic, J., Delatour, V., Ohlendorf, R., Henrion, A., Kawaguchi, M., Kang, D., Lee, H., Osuna, M. A., Serrano, V., Arzate, C. M. S., Krylov, A., Lopushanskaya, E., Nammoonnoy, J., Goren, A. C., Gunduz, S., Yilmaz, H., Mussell, C., Warren, J., Pritchett, J. S., Lippa, K., Nelson, M. A., Toman, B., Sniegoski, L. T., and Duewer, D., "High polarity analytes in biological matrix: determination of urea and uric acid in human serum (CCQM-K109)," Metrologia, 56, (2019).
9. Teo, T. L., Liu, Q. D., Cheow, P. S., Duewer, D., Lippa, K., Toman, B., Osuna, M. A., Serrano, V., Arzate, C. M. S., Wong, L. K., Chen, Y. Z., Yong, S., Teo, H. L., Liu, H., Tong, B., Kang, D., Lee, H., Dai, X. H., Quan, C., Shi, L. H., Li, H. M., and Pritchett, J. S., "Comparison of CRMs and value-assigned quality controls: urea and uric Acid in human serum or plasma (CCQM-K142)," Metrologia, 56, (2019).
10. Westwood, S., Josephs, R., Choteau, T., Daireaux, A., Wielgosz, R., Melanson, J., Thibeault, M. P., Meija, J., Quan, C., Hongmei, L., Huang, T., Zhang, W., Song, D., Martos, G., Delatour, V., Ohlendorf, R., Henrion, A., Kakoulides, E., Giannikopoulou, P., Alexopoulos, C., Chan, K., Lam, W. H., Fung, W. H., Yamazaki, T., Krylov, A., Lopushanskaya, E., Kharitonov, S., Belyakov, M., Sokolova, O., Liu, Q. D., Liu, H., Yong, S., Prevoo-Franzsen, D., Fernandes-Whaley, M., Kim, B., Jeong, J. S., Kwon, H. J., Yang, I., Nammoonnoy, J., Goren, A. C., Gunduz, S., Un, I., Bilsel, G., Biesenbruch, S., and Lowenthal, M., "Mass fraction assignment of Amino Acids in acidic aqueous solution (CCQM-K78.a)," Metrologia, 56, (2019).
11. Liaskos, C., Rhoderick, G., Hodges, J., Possolo, A., Goodman, C., Kim, Y. D., Kim, D. H., Lee, S., Allen, N., Corbel, M., Worton, D., Brown, R., and Brewer, P., "CCQM-K121-Monoterpenes in Nitrogen at 2.5 nmol mol(-1) Final Report," Metrologia, 55, (2018).
12. Shin, R., Dewi, F., Tong, B., Wah, L. H., Saxby, D., Armishaw, P., Ivanova, V., Feng, L. X., Wang, J., Busto, M. E. D., Fisicaro, P., Rienitz, O., Fung, W. H., Yau, M. H. P., Yim, Y. H., Buzoianu, M., Can, S. Z., Ari, B., Cankur, O., Infante, H. G., Perez-Zambra, R., Ferreira, E., Long, S., Davis, W. C., and Easley, R. A., "CCQM-K139 Elements in Human Serum," Metrologia, 55, (2018).
13. Vogl, J., Kipphardt, H., Richter, S., Bremser, W., Torres, M. D. A., Manzano, J. V. L., Buzoianu, M., Hill, S., Petrov, P., Goenaga-Infante, H., Sargent, M., Fisicaro, P., Labarraque, G., Zhou, T., Turk, G. C., Winchester, M., Miura, T., Methven, B., Sturgeon, R., Jahrling, R., Rienitz, O., Mariassy, M., Hankova, Z., Sobina, E., Krylov, A. I., Kustikov, Y. A., and Smirnov, V. V., "Establishing comparability and compatibility in the purity assessment of high purity zinc as demonstrated by the CCQM-P149 intercomparison," Metrologia, 55, (2018).
14. Wise, S. A., Phinney, K. W., Duewer, D. L., Sniegoski, L. T., Welch, M. J., Pritchett, J., Pabello, G., Calderon, M. A. A., Balderas, M., Liu, Q. D., Kooi, L. T., Rego, E., Garrido, B., Allegri, G., de La Cruz, M., Barrabin, J., Monteiro, T., Lee, H., Kim, B., Delatour, V., Peignaux, M., Kawaguchi, M., Bei, X., Can, Q., Nammoonnoy, J., Schild, K., Ohlendorf, R., Henrion, A., Goren, A. C., Yilmaz, H., Bilsel, M., Konopelko, L., Krylov, A., and Lopushanskaya, E., "CCQM-K11.2 Determination of Glucose in Human Serum and CCQM-K12.2 Determination of Creatinine in Human Serum," Metrologia, 55, (2018).
15. Wise, S. A., Phinney, K. W., Duewer, D. L., Sniegoski, L. T., Welch, M. J., Pabello, G., Caldero, M. A. A., Liu, Q. D., Kooi, L. T., Rego, E., Garrido, B., Allegri, G., de La Cruz, M., Barrabin, J., Puglisi, C., Lopez, E., Lee, H., Kim, B., Delatour, V., Heuillet, M., Nammoonnoy, J., Goren, A. C., Bilsel, G., Konopelko, L., Krylov, A., and Lopushanskaya, E., "CCQM-K6.2 Determination of Total Cholesterol in Human Serum," Metrologia, 55, (2018).
16. Kuroiwa, T., Zhu, Y., Inagaki, K., Long, S. E., Christopher, S. J., Puelles, M., Borinsky, M., Hatamleh, N., Murby, J., and Merrick, J., "Report of the CCQM-K123: trace elements in biodiesel fuel," Metrologia, 54, (2017).
17. Kuroiwa, T., Fung, W. H., Zhu, Y., Inagaki, K., Sin, D. W. M., Chu, H. S., Saxby, D., Merrick, J., White, I., Araujo, T., Almeida, M. D., Rodrigues, J., Yang, L., Pihillagawa, I. G., Mester, Z., Riquelme, S. S., Perez, L., Barriga, R., Nunez, C., Chao, J., Wang, J., Wang, Q., Shi, N., Lu, H., Song, P., Nuykki, T., Aho, T. S., Labarraque, G., Oster, C., Rienitz, O., Juhrling, R., Pape, C., Lampi, E., Kakoulides, E., Ketrin, R., Mardika, E., Komalasari, I., Okumu, T. O., Kang'iri, J. N., Yim, Y. H., Heo, S. W., Lee, K. S., Suh, J. K., Lim, Y., Manzano, J. V. L., Uribe, C., Carrasco, E., Tayag, E. D., Dablio, A. R. C., Encarnacion, E. K. P., Damian, R. L., Konopelko, L., Krylov, A., Vadim, S., Shin, R., Peng, S. L., Juan, W., Chang, X., Dewi, F., Horvat, M., Zuliani, T., Taebunpakul, S., Yafa, C., Kaewkhomdee, N., Thiengmanee, U., Klich, H., Can, S. Z., Ari, B., Cankur, O., Infante, H. G., Ferreira, E., Perez, R., Long, S. E., Kassim, B. L., Murphy, K. E., Molloy, J. L., and Butler, T. A., "Report of the CCQM-K124: trace elements and chromium speciation in drinking water-part A: trace elements in drinking water, part B: chromium speciation in drinking water," Metrologia, 54, (2017).
18. Wise, S. A., Tai, S. S. C., Duewer, D. L., Bedner, M., Camara, J. E., Lippa, K. A., Qinde, L., Kang, D., Kim, B., and Quan, C., "CCQM-K132: low-polarity analytes in a biological matrix: vitamin D metabolites in human serum," Metrologia, 54, (2017).
19. Bastkowski, F., Spitzer, P., Sander, B., Mariassy, M., Dimitrova, L., Reyes, A., Rodriguez, A., Manzano, V. L., Vospelova, A., Jakobsen, P. T., Pawlina, M., Korol, M., Kozlowski, W., Delgado, M., Canaza, G. T., Dias, J. C., Gonzaga, F. B., Szilagyi, Z. N., Jakusovszky, B., Vyskocil, L., Nongluck, T., Waters, J., Pratt, K. W., Asakai, T., Maksimov, I., Hankova, Z., Uysal, E., Gavrilkin, V., Prokunin, S. V., Ferreira, E., and Fajardo, S., "Key comparison on pH of an unknown phosphate buffer," Metrologia, 53, (2016).
20. Ma, L. D., Wu, B., Ortiz-Aparicio, J. L., Segoviano-Regalado, F., GarciaAlcantara, A. B., Valle-Moya, E., Rivera-Sanchez, G., Dumanska, J., Pawlina, M., Pietrzak, A., Kozlowski, W., Borges, P. P., Silva, W. B., Freitas, C. S., Araujo, T. O., Almeida, M. D., Lim, Y., Heo, S. W., Hwang, E. J., Wang, Q., Waters, J. F., Sieber, J. R., Pratt, K. W., Suzuki, T., Hioki, A., Mariassy, M., Hankova, Z., Shimolin, A., Sobina, A., Dobrovolsky, V., Prokunin, S., Aprelev, A., and Frolov, D., "CCQM-K48.2014: assay of potassium chloride," Metrologia, 53, (2016).
21. Hioki, A., Narukawa, T., Inagaki, K., Miyashita, S., Kotzeva, B., Kakoulides, E., Sxoina, V., Fung, W. H., Choi, Y. Y., Yau, H. P., Tsoi, Y. T., Lee, C. L., Kong, M. F., Shin, R., Juan, W., Yee, N. S., Uribe, C., Rodrigues, J. M., de Sena, R. C., Dutra, E. S., Bergamaschi, L., Giordani, L., D'Agostino, G., Valiente, L., Horvat, M., Jacimovic, R., Okumu, T. O., Kang'Iri, J., Orwa, T. O., Chao, W., Jingbo, C., Taebunpakul, S., Yafa, C., Kaewkhomdee, N., Chailap, B., Pharat, Y., Phukphattanachai, P., Turk, G. C., Long, S., Murphy, K. E., Davis, C., Ellisor, M., Merrick, J., White, I., Saxby, D., Linsky, S. M., Barzev, A., and Botha, A., "Report of the key comparison CCQM-K108 determination of arsenic species, total arsenic and cadmium in brown rice flour," Metrologia, 52, (2015).
22. Wise, S., "CCQM activities and impact in food safety and nutrition," Abstracts of Papers of the American Chemical Society, 250, (2015).
23. Rhoderick, G., Guenther, F., Duewer, D., Lee, J., Kim, J. S., Hall, B., Weiss, R., Harth, C., Reimann, S., and Vollmer, M., "CCQM-P151 Final Report Pilot Study CCQM P151 Halocarbons in Dry Whole Air," Metrologia, 51, (2014).
24. Rhoderick, G., Guenther, F., Duewer, D., Lee, J., Moon, D., Lee, J., Lim, J., and Kim, J. S., "CCQM-K83 Final Report International Comparison CCQM-K83 Halocarbons in Dry Whole Air," Metrologia, 51, (2014).
25. Vogl, J., Kipphardt, H., Torres, M. D. A., Manzano, J. V. L., Rodrigues, J. M., de Sena, R. C., Yim, Y. H., Heo, S. W., Zhou, T., Turk, G. C., Winchester, M., Yu, L. L., Miura, T., Methven, B., Sturgeon, R., Jahrling, R., Rienitz, O., Tunc, M., and Can, S. Z., "Final report of the key comparison CCQM-K72: Purity of zinc with respect to six defined metallic analytes," Metrologia, 51, (2014).
26. Westwood, S., Josephs, R., Choteau, T., Daireaux, A., Wielgosz, R., Davies, S., Moawad, M., Chan, B., Munoz, A., Conneely, P., Ricci, M., do Rego, E. C. P., Garrido, B. C., Violante, F. G. M., Windust, A., Dai, X. H., Huang, T., Zhang, W., Su, F. H., Quan, C., Wang, H. F., Lo, M. F., Wong, W. F., Gantois, F., Lalerle, B., Dorgerloh, U., Koch, M., Klyk-Seitz, U. A., Pfeifer, D., Philipp, R., Piechotta, C., Recknagel, S., Rothe, R., Yamazaki, T., Bin Zakaria, O., Castro, E., Balderas, M., Gonzalez, N., Salazar, C., Regalado, L., Valle, E., Rodriguez, L., Laguna, L. A., Ramirez, P., Avila, M., Ibarra, J., Valle, L., Perez, M., Arce, M., Mitani, Y., Konopelko, L., Krylov, A., Lopushanskaya, E., Lin, T. T., Liu, Q. D., Kooi, L. T., Fernandes-Whaley, M., Prevoo-Franzsen, D., Nhlapo, N., Visser, R., Kim, B., Lee, H., Kankaew, P., Pookrod, P., Sudsiri, N., Shearman, K., Goren, A. C., Bilsel, G., Yilmaz, H., Bilsel, M., Cergel, M., Coskun, F. G., Uysal, E., Gunduz, S., Un, I., Warren, J., Bearden, D. W., Bedner, M., Duewer, D. L., Lang, B. E., Lippa, K. A., Schantz, M. M., Sieber, J. R., and Westwood, S., "Final Report on Key Comparison CCQM-K55.c (L-(+)-Valine): Characterization of Organic Substances for Chemical Purity," Metrologia, 51, (2014).
27. Camara, J. E., Duewer, D. L., Gasca-Aragon, H., Lippa, K. A., and Toman, B., "CCQM-K80 Comparison of value-assigned CRMs and PT materials: creatinine in human serum Final Report," Metrologia, 50, (2013).
28. Hein, S., Philipp, R., Duewer, D. L., Gasca-Aragon, H., Lippa, K., and Toman, B., "CCQM-K79 Comparison of value-assigned CRMs and PT materials: Ethanol in Aqueous Matrix Final Report," Metrologia, 50, (2013).
29. Hioki, A., Nonose, N., Ma, L. D., Chao, J. B., Feng, L. X., Chao, W., Cho, K. H., Suh, J. K., Min, H. S., Lim, Y., Recknagel, S., Koenig, M., Vogl, J., de Sena, R. C., dos Reis, L. A., Borinsky, M., Puelles, M., Hatamleh, N., Acosta, O., Turk, G., Rabb, S., Sturgeon, R., Methven, B., Rienitz, O., Jaehrling, R., Konopelko, L. A., Kustikov, Y. A., Kozyreva, S. B., and Korzh, A. A., "Final report of the key comparison CCQM-K88: Determination of lead in lead-free solder containing silver and copper," Metrologia, 50, (2013).
30. Urquiza, M. P., Torres, M. M., Mitani, Y., Schantz, M. M., Duewer, D. L., May, W. E., Parris, R. M., Wise, S. A., Kaminski, K., Philipp, R., Win, T., Rosso, A., Kim, D. H., Ishikawa, K., Krylov, A. I., Kustikov, Y. A., and Baldan, A., "CCQM Key Comparison - Organic Solutions CCQM-K47 Volatile Organic Compounds in Methanol Final Report," Metrologia, 50, (2013).
31. Valiente, L., Saxby, D., Merrick, J., Kotzeva, B., Mester, Z., Yang, L., Willie, S., Feng, L., Wang, J., Labarraque, G., Rienitz, O., Sin, D. W., Mok, C., Ng, C., Fung, W., Yau, H., Wong, S., Zhu, Y. B., Yim, Y. H., Lee, K. S., Kim, I. J., Lim, Y. R., Cruz, P. R., Trejo, F. E. M., Moya, E. V., Castellanos, I. S., Torres, M. D. R. A., Manzano, J. V. L., Konopelko, L. A., Kustikov, Y. A., Shin, R., Horvat, M., Jacimovic, R., Milacic, R., Yafa, C., Taebunpakul, S., Kaewkhomdee, N., Phukphatthanachai, P., Cankur, O., Coskun, F. G., Turk, G. C., Davis, W. C., Wood, L. J., Murphy, K. E., and Entwisle, J., "CCQM-K89 Trace and Essential Elements in Herba Ecliptae Final Report," Metrologia, 50, (2013).
32. Camara, J. E., Lippa, K. A., Duewer, D. L., Gasca-Aragon, H., and Toman, B., "An international assessment of the metrological equivalence of higher-order measurement services for creatinine in serum," Analytical and Bioanalytical Chemistry, 403, 527-535 (2012).
33. Westwood, S., Josephs, R., Choteau, T., Daireaux, A., Mesquida, C., Wielgosz, R., Rosso, A., de Arechavaleta, M. R., Davies, S., Wang, H. J., do Rego, E. C. P., Rodrigues, J. M., Guimaraes, E. D., Sousa, M. V. B., Monteiro, T. M., Valente, L. A. D., Violante, F. G. M., Almeida, R. R. R., Quaresma, M. C. B., Nogueira, R., Windust, A., Dai, X. H., Li, X. M., Zhang, W., Li, M., Shao, M. W., Wei, C., Wong, S. K., Cabillic, J., Gantois, F., Philipp, R., Pfeifer, D., Hein, S., Klyk-Seitz, U. A., Ishikawa, K., Castro, E., Gonzalez, N., Krylov, A., Lin, T. T., Kooi, L. T., Fernandes-Whaley, M., Prevoo, D., Archer, M., Visser, R., Nlhapo, N., de Vos, B., Ahn, S., Pookrod, P., Wiangnon, K., Sudsiri, N., Muaksang, K., Cherdchu, C., Goren, A. C., Bilsel, M., LeGoff, T., Bearden, D., Bedner, M., Duewer, D., Hancock, D., Lang, B., Lippa, K., Schantz, M., and Sieber, J., "Final report on key comparison CCQM-K55.b (aldrin): An international comparison of mass fraction purity assignment of aldrin," Metrologia, 49, (2012).
34. Westwood, S., Josephs, R., Daireaux, A., Wielgosz, R., Davies, S., Wang, H. J., Rodrigues, J., Wollinger, W., Windust, A., Kang, M., Su, F. H., Philipp, R., Kuhlich, P., Wong, S. K., Shimizu, Y., Perez, M., Avila, M., Fernandes-Whaley, M., Prevoo, D., de Vos, J., Visser, R., Archer, M., LeGoff, T., Wood, S., Bearden, D., Bedner, M., Boroujerdi, A., Duewer, D., Hancock, D., Lang, B., Porter, B., Schantz, M., Sieber, J., White, E., and Wise, S. A., "CCQM-K55. a (Estradiol) Final Report: August 2012 CCQM-K55. a Key Comparison on the Characterization of Organic Substances for Chemical Purity," Metrologia, 49, (2012).
35. Westwood, S., Josephs, R., Choteau, T., Mesquida, C., Daireaux, A., Wielgosz, R., Davies, S., Windust, A., Kang, M., Ting, H., Kato, K., Frias, E., Perez, M., Apps, P., Fernandes-Whaley, M., Wiangnon, K., Ruangrittinon, N., Wood, S., LeGoff, T., Duewer, D., Schantz, M., Siekmann, L., and Esker, J., "An international comparison of mass fraction purity assignment of digoxin: The Comite Consultatif pour la Quantite de Matiere (CCQM) Pilot Study CCQM-P20.f (Digoxin)," Metrologia, 48, (2011).
36. Kipphardt, H., Matschat, R., Vogl, J., Gusarova, T., Czerwensky, M., Heinrich, H. J., Hioki, A., Konopelko, L. A., Methven, B., Miura, T., Petersen, O., Riebe, G., Sturgeon, R., Turk, G. C., and Yu, L. L., "Purity determination as needed for the realisation of primary standards for elemental determination: status of international comparability," Accreditation and Quality Assurance, 15, 29-37 (2010).
37. Ma, L. D., Feng, L. X., Hioki, A., Cho, K. H., Vogl, J., Berger, A., Turk, G., Macleod, S., Labarraque, G., Tong, W. F., Schiel, D., Yafa, C., Valiente, L., Konopelko, L. A., Quetel, C., Vermaercke, P., Manzano, J. V. L., Linsky, M., Cortes, E., Tangpitayakul, S., Plangsangmas, L., Bergamaschi, L., and Hearn, R., "International comparison of the determination of the mass fraction of cadmium, chromium, mercury and lead in polypropylene: the Comit, Consultatif pour la Quantit, de MatiSre pilot study CCQM-P106," Accreditation and Quality Assurance, 15, 39-44 (2010).
38. Sharpless, K. E., Rimmer, C. A., Phinney, K. W., Nelson, B. C., Duewer, D. L., Wise, S. A., Kim, B., Liu, J., Huang, T., and Zhang, W., "Final report on CCQM-K62: Nutrients in infant/adult formula-vitamins," Metrologia, 47, (2010).
39. Tai, S. S. C., Duewer, D. L., Urquiza, M. P., Lee, H., Mussell, C., Dai, X. H., Phinney, K. W., Welch, M. J., Wise, S. A., Murby, J., Kawaguchi, M., and Henrion, A., "Final report on CCQM-K63.a,b: Non-peptide hormones in serum: cortisol and progesterone," Metrologia, 47, (2010).
40. Wong, Y. C., Sin, D. W. M., Yip, Y. C., Valiente, L., Toervenyi, A., Wang, J., Labarraque, G., Gupta, P., Soni, D., Surmadi, Hwang, E., Yafa, C., Cankur, O., Uysal, E., Turk, G., Huertas, R., and Murphy, K. E., "International comparison of the determination of cadmium and lead in herb: the Comite Consultatif pour la Quantite de Matiere (CCQM) pilot study CCQM-P97 (vol 14, pg 151, 2009)," Accreditation and Quality Assurance, 15, 433 (2010).
41. Wong, Y., Sin, D., Yip, Y., Valiente, L., Toervenyi, A., Wang, J., Labarraque, G., Gupta, P., Soni, D., Surmadi, Hwang, E., Yafa, C., Cankur, O., Uysal, E., Turk, G., and Huertas, R., "International comparison of the determination of cadmium and lead in herb: the Comit, Consultatif pour la Quantit, de MatiSre (CCQM) pilot study CCQM-P97," Accreditation and Quality Assurance, 14, 151-158 (2009).
42. Goenaga-Infante, H., Sturgeon, R., Turner, J., Hearn, R., Sargent, M., Maxwell, P., Yang, L., Barzev, A., Pedrero, Z., Camara, C., Huerta, V. D., Sanchez, M. L. F., Sanz-Medel, A., Emese, K., Fodor, P., Wolf, W., Goldschmidt, R., Vacchina, V., Szpunar, J., Valiente, L., Huertas, R., Labarraque, G., Davis, C., Zeisler, R., Turk, G., Rizzio, E., Mackay, L. G., Myors, R. B., Saxby, D. L., Askew, S., Chao, W., and Jun, W., "Total selenium and selenomethionine in pharmaceutical yeast tablets: assessment of the state of the art of measurement capabilities through international intercomparison CCQM-P86," Analytical and Bioanalytical Chemistry, 390, 629-642 (2008).
43. Mariassy, M., Hioki, A., Konopelko, L. A., Dimchoglo, I., Winchester, M., Butler, T., Case, R., Hernandez, A., Galli, A., Alvarez, P., Lee, J. H., Royer, C., Shi, N. J., Chao, J. B., Wu, B., and Li, H. F., "Final report on key comparison CCQM-K59: Determination of nitrite and nitrate in calibration solutions," Metrologia, 45, (2008).
44. Hioki, A., Kurahashi, M., Turk, G., Matschat, R., and Recknagel, S., "CCQM-K33 Final Report: Determination of minor elements in steel," Metrologia, 43, (2006).
45. Schantz, M. and Wise, S., "CCQM-K25: Determination of PCB congeners in sediment," Metrologia, 41, (2004).

