Summary
Our goal is to develop methods and materials for imaging-based characterization of molecular signatures that occur during cellular proliferation, differentiation and internalization, in order to provide quantitative and reliable information regarding disease state from histopathology, and regarding cellular differentiation state in regenerative medicine.
Description
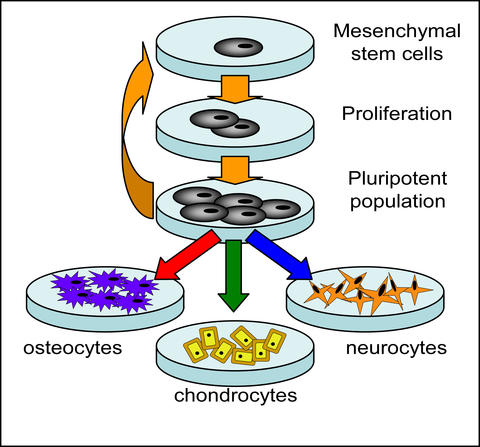
Proliferation and differentiation of mesenchymal stem cells (MSCs) to cell types such as osteocytes, chondrocytes and neurocytes.
Imaging and image analysis are used ubiquitously in biological and biomedical research. One of the great advantages of imaging is its intuitive information content; much can be learned from just a glance at the data. On the other hand, these data generally contain a very large amount of untapped information. For example, characterization of spatial phenotype heterogeneity in cells on a tissue scaffold is a critical question in tissue engineering, histopathology, and basic systems biology. However, there is currently no way to obtain such information short of using destructive and very labor intensive approaches.
We are developing imaging based methods to characterize cells and tissues in minimally or non- invasive formats which maintain high spatial and temporal resolution. This requires the development of high specificity probes capable of identifying specific signaling events and quantitative imaging methods which capture cell responses and response heterogeneity. We are also developing functional materials that induce specific cellular behavior, and that can be used as reference materials against which to calibrate our methods.
Major Accomplishments
We are a core member of the Integrated Technology Resource for Polymeric Biomaterials, an NIH-funded national research resource located within the New Jersey Center of Biomaterials.
The Bioimaging Project is collaborating with the University of Nebraska Medical Center to measure the molecular signatures and cell population heterogeneity in follicular thyroid cancer lesions using in situ hybridization of DNA-derivatized quantum dots and automated fluorescence microscopy. We anticipate the findings will lead to more accurate clinical diagnosis in biopsy samples.
Additional Technical Details
Label Free Imaging via CARS Microscopy
We were the first to demonstrate a broadband (3000 wavenumbers) CARS microscope, and have now designed and implemented a second generation broadband CARS microscope with significantly increased capabilities and sensitivity. This method uses two input light pulses to read out vibrational susceptibility of a sample, and possesses the inherent chemical sensitivity required to spatially map cell phenotype noninvasively. The presence of a nonresonant background (NRB) generally obscures the weak vibrational peaks found in the fingerprint region (500 to 1800) wavenumbers, and has made the fingerprint region unavailable to CARS for cell phenotyping. We have recently developed experimental and numerical approaches to overcome the NRB roadblock.
T.W. Kee and M.T. Cicerone* "Simple approach to one-laser, broadband coherent anti-Stokes Raman scattering microscopy" Optics Letters, 2004, 2701-2703.
Y.J. Lee and M.T. Cicerone* "Single-shot interferometric approach to background free broadband coherent anti-Stokes Raman scattering spectroscopy" Optics Express, 2009, 17, 1, 123-135.
Universal Gradient Substrates
Using "click" chemistry, we have designed a universal gradient substrate to which any azide-derivatized molecule can be attached. Using this method we have generated biomimetic RGD surfaces with 2 to 3 orders of magnitute variation in peptide concentration. The controlled variation of ligand density is critical to identifying specific receptors that guide cell-function, including adhesion.
ND Gallant, KA Lavery, EJ Amis, ML Becker* "Universal Gradient Substrates for Click Biofunctionalization" Advanced Materials, 2007, 19(7), 965-969.
Ligand Generation via Combinatorial Phage Display
Biological selection techniques (e.g., phage display, cell surface display, etc.) have recently been applied to material targets yielding "engineered peptides" that bind strongly and specifically to a range of different metal, semiconductor, oxide, and biomineral surfaces. We have been using this approach to bind hydroxyapatite, genetically engineered silks and tyrosine derived polycarbonates.
MD Roy, SJ Stanley, EJ Amis, ML Becker* "High Specificity Hydroxyapatite Binding Motif Identified via Phage Display" Advanced Materials, 2008, 20(10), 1830-1836.
DNA-derivatized Quantum Dots for In Situ Hybridization
Minimally invasive methods of identifying human disease states in situ and in vivo are of critical importance to early detection efforts, therapeutic intervention efficacy and increasing patient survival rates. These efforts however rely on robust and highly specific biomarkers which are both reliable and accessible to clinicians. We have developed a scalable, reproducible method for synthesizing high-quality optical probes thathave been derivatized with DNA-based aptamers targeted to mRNA signatures in cells via in situ hybridization methods.
MD Roy, A Herzing, SH Lacerda, ML Becker* "Emission-Tunable, Microwave Synthesis of Highly Luminescent Water Soluble Quantum Dots" Chemical Communications, 2008, 2106-2108.
Associated Product(s)
Project Summary (PDF)

