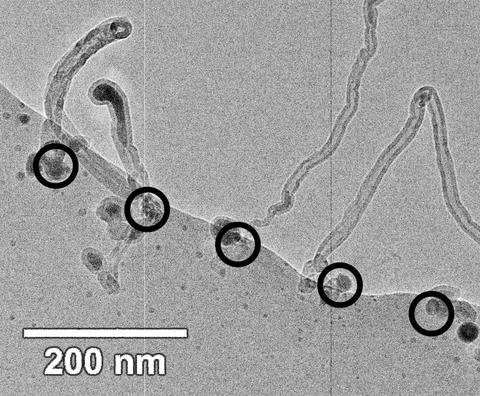
Researchers from the NIST Center for Nanoscale Science and Technology and Arizona State University have used an environmental scanning transmission electron microscope (ESTEM) to control the size and placement of iron nanoparticles in order to catalyze the growth of carbon nanotubes on a silicon oxide substrate.* Large scale synthesis of carbon nanotubes for low-cost field emission displays (FEDs) requires stringent control on nanotube length, diameter, and surface density. Using the ESTEM, the researchers were able to visualize the placement of the catalyst nanoparticles and the growth of the nanotubes in real-time. They tested the hypothesis that the diameter of carbon nanotubes is dependent on the size of the catalyst particles by depositing iron catalyst nanoparticles of different sizes and densities on a substrate using the microscope's electron beam to induce dissociation of the iron-containing catalyst precursors. They found that a number of factors control the size and catalytic activity of nanoparticles for nanotube growth, including the choice of the precursor (ferrocene or diiron nonacarbonyl), the substrate temperature, the residence time of the precursor on the substrate, and the electron beam energy. They were able to use the deposition time to control the particle size and the position of the electron beam to control the location of the catalyst particles on the surface of the substrate. They also found that the catalytic activity of the iron particles for tube growth depends upon the amount of carbon co-deposited with the iron during the electron beam-induced deposition process, because co-deposited carbon forms graphitic shells around the iron particles. These shells made the particles chemically inactive for inducing carbon nanotube growth. This problem was solved for the diiron nonacarbonyl precursor by increasing the substrate temperature to 100 °C, which reduced the amount of co-deposited carbon. Since heating the substrate did not affect co-deposited carbon levels in the ferrocene samples, diiron nonacarbonyl appears to be better suited as a catalyst precursor for controlled carbon nanotube growth. The researchers believe these results will help in creating substrates with carbon nanotubes at appropriate sizes and surface densities for use in FEDs.
*Controlling the size and the activity of Fe particles for synthesis of carbon nanotubes, S. W. Chee and R. Sharma, Micron 43, 1181–1187 (2012).
NIST Publication Database Journal Web Site

