They are hard to detect. They are extremely powerful. And they come in many forms.
This makes synthetic drugs like fentanyl particularly dangerous and hard to control.
But scientists are developing new methods to detect and identify these drugs while ensuring the safety of law enforcement officers, first responders and forensic chemists.
Safe, Efficient, Reliable: New Science in the Fight Against Killer Drugs
NIST researchers give law enforcement and public health experts new tools to combat fentanyl and other synthetic drugs.
(Updated April 12, 2023, to reflect the latest research.)
More than 107,000 people died of drug overdoses in the United States in 2021. That is the highest number ever recorded and a 15% increase from the year before.
This spike in deaths is being driven by fentanyl, a synthetic opioid that mimics the effects of heroin but is up to 50 times more potent. A graph of the overdose statistics from the Centers for Disease Control and Prevention shows the line for fentanyl turning sharply up starting in 2014. It’s been rising steeply ever since.
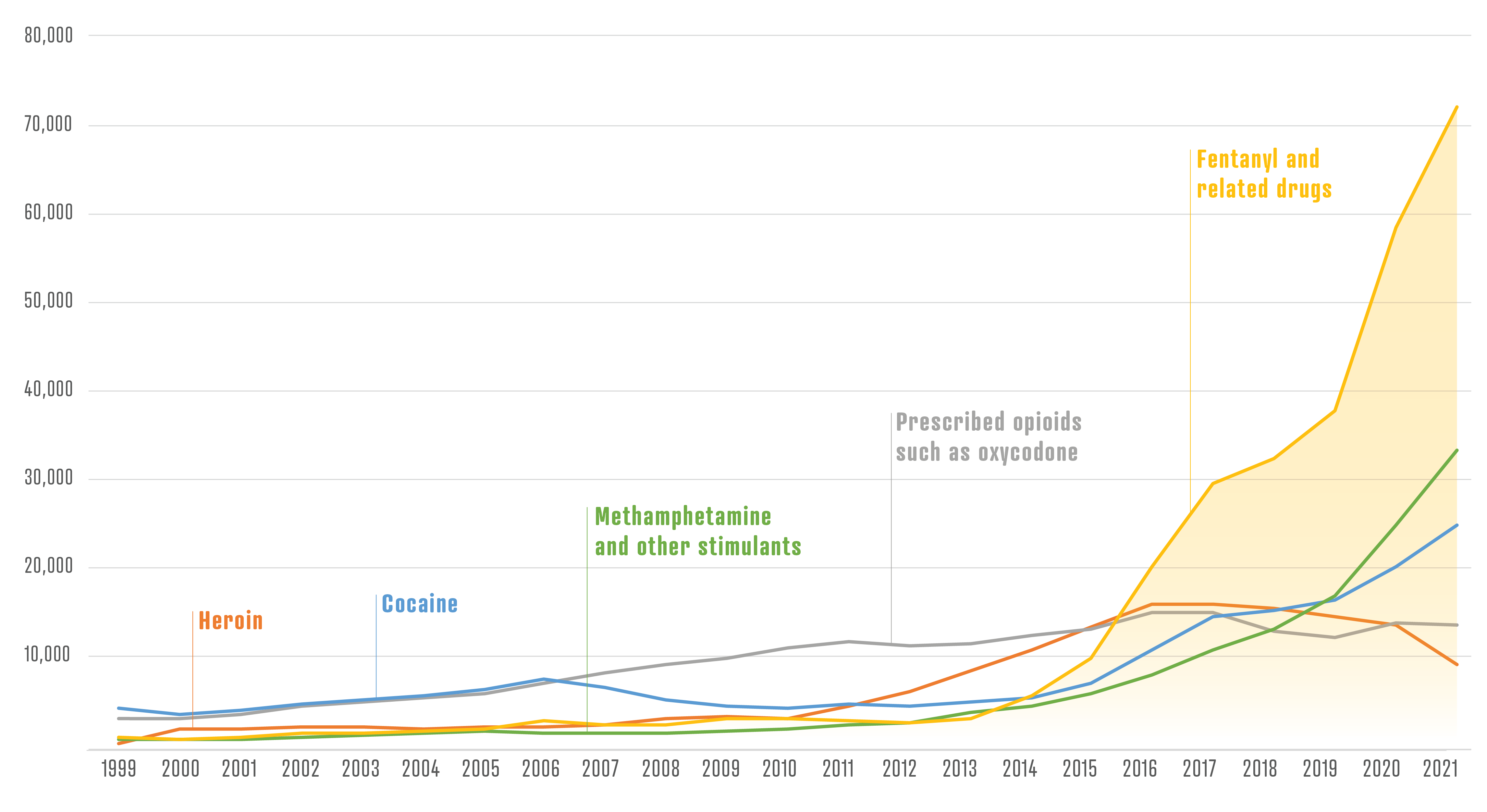
Fentanyl kills so many people in part because it is cheap, potent and easy to manufacture. In addition, clandestine chemists can easily cook up new varieties, or analogues, of fentanyl by tweaking its molecular structure. Dealers often spike heroin, cocaine and other drugs with fentanyl and its analogues. Users may not know what they’re getting, and they can more easily overdose as a result.
The high potency and changeability of fentanyl also present challenges to first responders, police and forensic chemists.
First, it is hazardous. During laboratory or field analysis, for example, particles can become airborne and even a small amount, if accidentally inhaled, can be dangerous.
Second, it hides behind other drugs. Analyzing drug mixtures that contain small but deadly amounts of fentanyl is complicated and requires the ability to detect substances at very low levels.
Third, it can fly under the radar. Traditional laboratory methods are not designed to detect and identify new drug analogues. This can hinder law enforcement and delay the public health response to newly emerging substances.

Drug abuse has plagued public health for decades, but the unprecedented toll of recent years reflects a new reality. Today’s illicit drugs are more dangerous and harder to control than the drugs of decades past.
“A new drug might appear, then three or six months later it’s gone, replaced by something new,” said NIST chemist and program manager Marcela Najarro. “It’s a totally different ballgame than 10 or 15 years ago.”
Limiting the harm from the opioid epidemic requires expertise from public health, social science, law enforcement and other domains. But one often overlooked need is expertise in the area of analytical chemistry.
As the nation’s chemical measurements laboratory, NIST is working on analytical methods to meet these challenges. Researchers with NIST’s Forensic Science Program are developing new tools that can speed up the public health response to newly emerging synthetic drugs. They are also improving existing technologies, developing new ones, and collaborating closely with law enforcement organizations and forensic labs to help them successfully implement solutions. Here is a rundown of what NIST is doing.
Part I: Monitoring the Illicit Drug Supply
New compounds constantly appear in the illegal drug supply, creating new and often unknown risks. NIST is working to monitor these changes, so that law enforcement and public health authorities can respond more quickly to emerging threats.
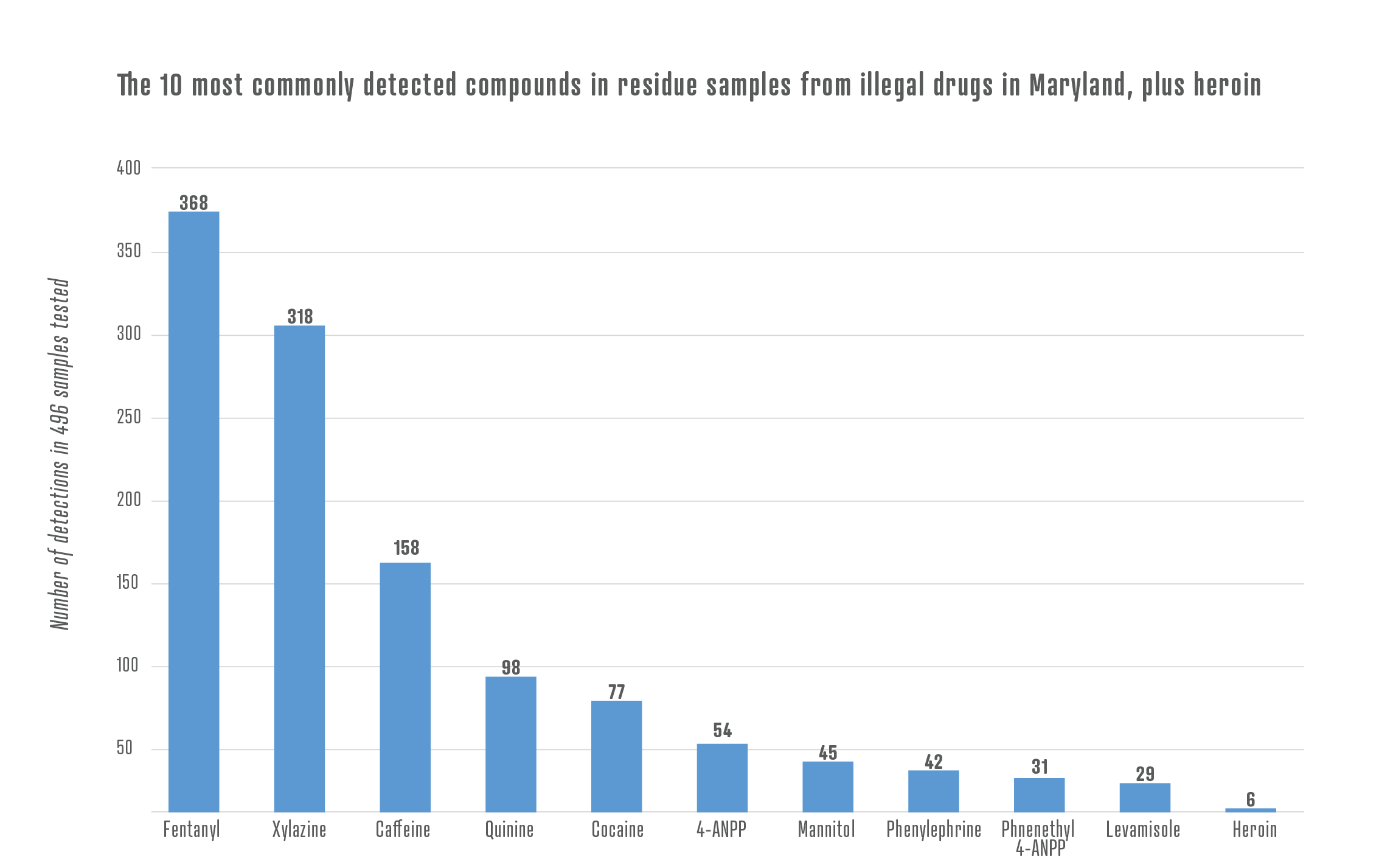
For example, the animal tranquilizer xylazine has flooded drug markets in the Northeast. When injected, xylazine causes wounds at the injection site, in some cases so severe that people need to have their arms or legs amputated.
As part of NIST’s Rapid Drug Analysis and Research, or RaDAR program, NIST is working with the Maryland Department of Health to analyze residues from illegally purchased drugs to determine the chemical compounds the drugs contain. NIST’s analysis found xylazine in 80% of samples that contained opioids.
The results from the analysis are shared at needle exchange centers so that people know what compounds are present in the drugs they are buying. NIST also shares the results more broadly with public health authorities, to provide an early warning when new drugs or highly potent drug analogues hit the streets.
“With this program, we can detect new substances more quickly than before,” said Erin Russell, who leads the Maryland Department of Health’s Center for Harm Reduction Services. “And we can mobilize resources where they’re needed based on what we’re seeing in the data.”
What NIST Is Doing
Part II:
Safe Handling in the Field and in the Lab
Police, first responders and forensic chemists face a new hazard: unknown substances of unknown potency. A bag of powder might be cocaine, or it might be fentanyl. Or it might be something worse.
Some synthetic drugs can be extremely dangerous. A fentanyl analogue called carfentanil, for instance, can be 100 times more potent than fentanyl — 5,000 times more potent than heroin. Drugs like this can be dangerous or even deadly if a small amount is accidentally inhaled.
To test seized drugs using conventional methods, police in the field or chemists in the lab have to open suspicious packages and handle the contents directly. “But with novel drugs, you have to assume they’re dangerous, because you don’t know how potent they are,” said Amber Burns, manager of the Maryland State Police forensic chemistry lab. “The best strategy is to minimize exposure.”
NIST scientists are working closely with Burns and other forensic chemists to develop new ways to test drug evidence that minimize exposure. One research collaboration recently showed that labs can get a reliable initial identification of seized material by swiping the outside of a package. Because this method does not require opening the package or handling its contents directly, it minimizes the risk of accidental exposure.
“With this method, labs can quickly identify hazardous samples and separate them for special handling,” said NIST research chemist Ed Sisco. “That speeds up workflow in the lab without compromising on safety.”
Accuracy also cannot be compromised. Forensic results in the hands of police and prosecutors can have a major impact on peoples’ lives. For this reason, NIST is helping labs ensure not only that these new methods are safe and efficient, but that they produce reliable results.
The researchers have published detailed accounts of how they implemented and validated these new methods so that other labs can follow suit. This helps ensure that these solutions can be implemented across the country and at scale.
The key to the success of these research projects is collaboration. In addition to running the Maryland State Police forensic chemistry lab, Burns is a visiting researcher at NIST. Similarly, Sisco is a guest researcher at the Maryland State Police forensics lab. This exchange of expertise helps ensure that the solutions NIST presents are realistic and address the real-world, complex problems that crime labs face.
This video describes one of many collaborative research projects aimed at protecting police and crime lab chemists from accidental exposure.
What NIST Is Doing
Part III:
Rapid Identification of New Substances
Fentanyl is not just a killer. It’s also a shape-shifter. Illicit chemists easily cook up new analogues, each with a slightly different molecular structure. These can be far more potent than the original. This makes them particularly deadly to users, who don’t always know what they are consuming.
The potential number of analogues is immense. This is what fentanyl and a few of its analogues look like:
There are hundreds of known fentanyl analogues. Here are just a few that have been identified in seized drugs in recent years.
Analogues often evade detection because of a scientific Catch-22. When forensic chemists analyze a suspicious-looking powder, they run it through an instrument that generates the molecular “fingerprints” of the chemical compounds present. They then run those fingerprints, called mass spectra, through a database to look for a match. However, they will only get a match if the compound is already listed in their database. New analogues, by definition, are not yet listed, and most forensic labs will be unable to identify them.
“The technology is not very good at identifying unknown unknowns,” Sisco said.
Most labs, if they detect something new, will have to send a sample to one of a small number of labs that have the sophisticated equipment and rare expertise needed to identify the compound and work out its chemical structure. This delay slows down the response by public health and law enforcement agencies.
“If people start overdosing and dying from a new drug analogue, authorities need to identify it as quickly as possible,“ said NIST research chemist Aaron Urbas in a news release. “If you want to focus your resources effectively, you need to know what you’re looking for.”
The challenge of identifying analogues is present with many types of drugs beyond fentanyl. Other synthetic opioids also spin off analogues, as do cannabinoids (aka synthetic marijuana), cathinones (aka bath salts), benzodiazepines and others. All can be deadly, and all appear in many forms.
To help labs identify novel substances more quickly, NIST is developing new software tools that partly solve the Catch-22 of drug identification by recognizing subtle patterns in molecular fingerprints. This software can indicate whether an unknown compound is likely to be a new fentanyl analogue, shortening the time it takes to work out its molecular structure and add its fingerprints to chemical identification databases. NIST scientists also regularly update chemical identification databases with new compounds and have developed online platforms where forensic and research chemists can share information on emerging substances more quickly.
What NIST Is Doing
Part IV:
The First Line of Defense
Most of the fentanyl used illegally in the United States originates in China. Stopping shipments of the drug before they enter the country is the first line of defense.
Fentanyl and its analogues enter the U.S. along two main routes. Bulk shipments arrive via Mexico, smuggled across the border at vehicle crossings. Smaller parcels arrive from China directly by airmail and commercial delivery services.
Consider the air shipping method first. At John F. Kennedy International Airport, roughly 1 million packages arrive from overseas every day. This presents a classic needle-in-a-haystack problem, but one that is made even more daunting by the fact that fentanyl is so potent. This means it can be profitably shipped in small quantities that are easy to conceal.
At postal inspection facilities, officers use chemical detectors, trained detection dogs and X-ray machines to find packages that might contain fentanyl. But each package has to be individually inspected — a huge, labor-intensive and potentially hazardous job.
NIST is developing technologies that can help identify suspicious packages efficiently, reliably and safely. One approach involves blasting packages with high-pressure jets of air, then directing the airflow to sensors that can detect traces of narcotics. If any traces are found, those packages can be separated for manual inspection.
NIST originally developed this method, called aerodynamic sampling, for high-throughput screening of airport luggage for explosives. But the same approach can be used to detect narcotics. In this video, NIST researcher Matt Staymates explains how he uses specialized video techniques to understand and improve aerodynamic sampling.
The challenge at the border is also immense. More than 6 million trucks and nearly 77 million personal vehicles entered the U.S. through ports of entry along the southern border in 2018. NIST researchers are also testing and improving existing technologies that can be deployed in that very different operational environment.
What NIST Is Doing
Part V:
Keeping It Real
Public health and law enforcement officials today face a growing and fast-changing challenge. To do their jobs — and to save lives — they need to be able to detect and identify a growing number of new and potent drugs.
Meeting this challenge requires new thinking in terms of analytical chemistry. NIST is developing new approaches in close collaboration with the experts, at the Maryland State Police Department and other agencies, who encounter these drugs every day. NIST program manager Marcela Najarro explains why this type of close collaboration is essential to move the science forward.
These collaborations are expanding to include other agencies facing similar challenges, and this page will be updated as new projects come online. Unfortunately, as the overdose epidemic intensifies, the research needs in forensic chemistry are growing. Today the main culprit is fentanyl. But many synthetic drugs are similarly dangerous and difficult to control. And new synthetic drugs, still unknown, will emerge in coming years.
The opioid crisis presents analytical chemistry challenges that are great and constantly changing. But NIST is working with forensic labs to help them meet those challenges — safely, efficiently and reliably.
Technical Contact
Marcela Najarro
marcela.najarro [at] nist.gov (marcela[dot]najarro[at]nist[dot]gov)

