Taking Measure
Just a Standard Blog
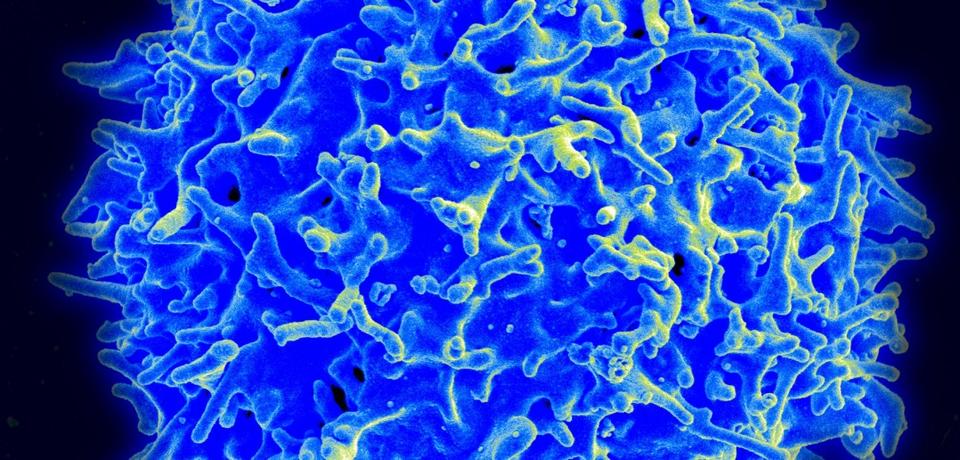
Scanning electron micrograph of a human T lymphocyte (also called a T-cell) from the immune system of a healthy donor.
I came to NIST in January 2015, not as a physicist, analytical chemist, or engineer, but as a biologist, a fit that surprised some of my friends from grad school. NIST … biology … whaaat? Most people think of measurements when they think of NIST, and the why and how for measuring biology is not always obvious.
Measurements for biological systems are usually complex and fuzzy because living things are complicated and behave unpredictably, despite our best efforts. This can be frustrating to people who measure things for a living (like the folks I work with), who prefer hard numbers and tight error bars. Yet, we use biotechnology to make many commercial products (medicines, beer, biofuels, and food, for example). As a program advisor, I spend a large chunk of my time figuring out what NIST can and should do for stakeholders across the wide landscape of commercial applications of biotechnology.
That can be hard to reduce to a talking point or elevator pitch, much less explain to my kids over dinner.
My “outputs” are often acronym-laden white paper summaries filled with technical jargon that only bureaucrats can love. But, occasionally I find myself in a room where everything we work for comes together in a big way that matters, and all the jargon and government-speak falls away. This is about one of those times, a couple of weeks ago, and it came courtesy of a small meeting we organized at NIST.
If this were a movie, we’d have to do a flashback here.
Like most people at NIST, I’m a shameless science junkie—always perking up at the mention of some new “way cool” discovery. But too often these new discoveries don’t quite live up to the hype. While this is a bummer when it comes to the latest cell phone, it’s completely heartbreaking in cancer research. In the 13 years since I lost my mom to cancer, there have been advances in chemotherapy agents and in our understanding of how tumor cells fight off treatment, but nothing really game-changing—until scientists and clinicians started showing data on something that seemed to work really well in small groups of patients who have certain treatment-resistant cancers and have run out of options.
A couple of months into this job, I heard Steven Rosenberg of the National Institutes of Health (NIH) speak about how researchers could now take specific immune cells (commonly called “T-cells”) from patients with types of melanoma, sarcoma, or lymphoma, genetically engineer them to target the cancer cells, and then put them back into the patient. I’d read about these treatments, but the “before-during-after” pictures he showed were stunning, as were the clinical response rates. These things didn’t just slow down the cancer, but seemed to be curing it.
Game-changer? Definitely.
Steven Rosenberg isn’t the only clinician with dramatic success stories using engineered immune cells to take out cancer. As of last month, more than 300 clinical trials worldwide were using these “adoptive T-cell” therapies. Large pharma, small biotechs, and academic clinical centers are partnering in droves to figure out how to get the therapies to patients who desperately need them. These are highly motivated scientists, doctors, and businesspeople working on treatments that could save literally millions of lives a year—and be worth billions of dollars—if they can get them to work in large trials for other types of cancers … so what’s stopping them?
It turns out a major stumbling block is a manufacturing problem rooted in measurements. While researchers can make these cell therapies in small batches, no one has yet perfected a full-scale manufacturing process. The Food and Drug Administration (FDA) is hesitant to approve these therapies for wider use because it’s not clear how to monitor and measure product quality.
After all, these therapies are not your grandmother’s white-powder cholesterol pill. They are complex therapies comprised of living cells that have been genetically engineered using scientific advances we only dreamed about 30 years ago. And whaddaya know—at NIST we are all about developing measurements to get new technologies over manufacturing hurdles and into the marketplace!
So, last month, just after President Obama introduced his plan for a cancer “moonshot” in his State of the Union Address, 130 researchers, regulators, manufacturers and clinicians came to NIST for a meeting on “Measurement Challenges for CAR-T Biomanufacturing,” which, believe it or not, is about as “sexy” a topic as science offers today.
(CAR-T stands for Chimeric Antigen Receptor T-Cells, a mouthful that basically means immune cells that have been manipulated to recognize a patient’s tumor cells. The genius in the acronym is that tweaked versions of these therapies get to have clever names like “Go-CARTs,” and “Armored CARs,” and “Cide-CARS,” which are more fun than your grandma’s cholesterol medication as well.)
The keynote speaker at our event, UPenn’s Bruce Levine, showed us over and over again why these therapies are worth our efforts. He presented data from small clinical trials showing the response for children and adults with leukemias and lymphoma with complete response rates of 90 percent for some trials, with periods of remission far outlasting the usual rate for these aggressive cancers.
(Once again, it’s tempting to whisper the word “cure” when talking about these cancer therapies, which would have been unthinkable just 10 years ago.)
Then, through the rest of the morning and afternoon, the FDA, the clinicians, the academics, the biotech and biopharma reps all asked NIST for help with measurements to ensure that these therapies can be manufactured quickly and safely, which is a real challenge, given that the starting material is different for each patient. Some of these measurement needs are head-scratchers (what assays in the lab predict effectiveness in the patient?), and some are simple in concept but really difficult in practice (how exactly do you count out a “dose” of living cells?).
Fortunately, our NIST folks have been on the job preparing for this challenge for several years now, figuring out what tools we need for measuring biology and the products made using biotechnology, with a shout out to my colleagues Mike Tarlov, Anne Plant, John Elliott, and Sheng Lin-Gibson, in particular.
This is no accident or lucky coincidence—it really does take vision, hard work, and nimble scientists to make an impact when and where it matters most. Right now, we are developing measurement tools for a host of biological applications—clinical diagnostics, biothreat detection, forensics, and food safety, just to name a few.
In the 30 years and counting since the first scientists began scratching out the possibility of using a patient’s own immune system to fight cancer, through all the teams of researchers working out how to engineer the cells and grow them in the lab, to the clinicians who went through the huge hassle of getting approval to try them in patients, to the patients who were trusting enough to let the clinicians try, to the regulators who have to set guidance for safety AND effectiveness, it now all comes down to manufacturing and measurements.
This where the rubber hits the road for us at NIST—helping industry overcome those last barriers to manufacturing these therapies for widespread clinical use through the development of better measurement tools. Our contribution isn’t likely to show up in the headlines, but we are problem-solvers with critical expertise in doing what seems like an impossible task—we find ways to measure living, breathing, messy, complicated and powerful biology.
That’s pretty cool.
About the author
Related Posts
Comments
- Reply