OSAC Newsletter August 2016

August 2016
Message from the Forensic Science Standards Board (FSSB) | OSAC Digital Evidence Subcommittee Survey on Accreditation, Standards and Certification | Upcoming Public Comment Period for Standards/Guidelines Under Consideration for the OSAC Registry | SWGDAM Guidelines Posted for Public Comment | Research & Development Needs | Recent OSAC Accomplishments: Standards and Guidelines Approved for the OSAC Working with an SDO Process | On the Horizon | Standards Developing Organization (SDO) News | OSAC Featured Unit: Wildlife Subcommittee
Message from the Forensic Science Standards Board (FSSB)
The positive progress during the first full year of OSAC has not been without some operational and communications challenges. While OSAC processes were developed to be transparent and thorough, we realized over the past year that further efficiencies and improvements could be gained.
With this focus in mind, the FSSB conceptualized and attended the recent OSAC Leadership Strategy Session on June 22, 2016. Three representatives from each OSAC main committee contributed, providing both written and verbal feedback related to the OSAC program. This exercise ultimately resulted in 25 programmatic recommendations that are now under further consideration and analysis for potential implementation later this year. Recommendations included:
• Refining OSAC infrastructure and processes to be more efficient without sacrificing quality. This will help ensure that OSAC members' time is used wisely, and will maximize their strategic input to scientifically sound standards and guidelines.
• Potentially refining the infrastructure to foster additional inter-committee efforts, so that collaboration between human factors, quality, legal, and statistician stakeholders can occur with forensic science practitioners as early and as often as practical.
• Developing an overarching strategy to best achieve timeliness, relevance, consensus, and quality in OSAC standards.

OSAC Leadership Strategy Session. John Paul Jones, Associate Director of OSAC Affairs at NIST, facilitated the OSAC Leadership Strategy Session. Attendees discussed a path forward for OSAC that will maximize collaboration between the various stakeholders.
One recommendation was to continue to hold these leadership discussions regularly, in order to ensure that these inter-committee and inter-stakeholder discussions continue at the committee chair level and beyond. For questions about this or other OSAC activities, contact forensics [at] nist.gov (forensics[at]nist[dot]gov).
OSAC Digital Evidence Subcommittee Survey on Accreditation, Standards and Certification
As the digital evidence forensic discipline progresses, there are questions regarding quality assurance policy that need to be addressed. The Scientific Working Group on Digital Evidence and the OSAC Digital Evidence Subcommittee created a survey to seek stakeholder opinions regarding laboratory accreditation, digital evidence standards, and examiner certification. Please take a few minutes and complete the survey; it will close on December 1st, 2016. Your participation is valued and will help those writing policy guides and standards from the perspective of the entire community.
https://www.surveymonkey.com/r/7RD9DTM
Upcoming Public Comment Period for Standards/Guidelines Under Consideration for the OSAC Registry

The Chemistry/Instrumental Analysis SAC has submitted the following standard (ASTM: E2451-13 Standard Practice for Preserving Ignitable Liquids and Ignitable Liquid Residue Extracts from Fire Debris Samples) to the OSAC Registry. The 30-day public comment period opens today, August 18, on the Kavi Public Workspace under "documents". The intent of the public comment period is to collect public opinion on inclusion of the documents on the relevant OSAC registry.
ASTM: E2451-13 Standard Practice for Preserving Ignitable Liquids and Ignitable Liquid Residue Extracts from Fire Debris Samples
This document describes procedures for preserving residues of ignitable liquids in extracts obtained from fire debris samples and questioned ignitable liquid samples.
SWGDAM Guidelines Posted for Public Comment
Three new SWGDAM Guideline documents have been posted for public comment on the SWGDAM Public Comment webpage. They are available for comment 30 days. Please forward this information to any appropriate personnel from your laboratory.
• Interpretation Guidelines for Autosomal STR Typing (Posted July 25, 2016)
• Contamination Prevention and Detection Guidelines (Posted July 25, 2016)
• Guidelines for the Processing of Sexual Assault Kits in a Laboratory (Posted July 25, 2016)

The Digital/Multimedia Scientific Area Committee has 11 Research and Development Needs to contribute. They are under various Digital/Multimedia subcommittees:
Under the Digital Evidence Subcommittee:
• Scientific Analysis of Hash Authentications
Under the Facial Identification Subcommittee:
• Assessment of the Accuracy of Getting Facial Images from DNA
• Human Factors in Facial Image Comparison
• Post Capture Imaging Processing
• Validation of the "Physical Stability of Facial Features of Adults" Document
Under the Speaker Recognition Subcommittee:
• DATASETS for Community Research Activities
• Forensic Speaker Recognition Evaluation
Under the Video/Imaging Technology & Analysis Subcommittee:
• Determination of the Size of the Smallest Detail Required for Tire and or Shoe Comparisons
• Factors Affecting Image Quality When Extracting a Still from Video
• Software Validation Repository
Research & Development Needs can be found in greater detail on the NIST OSAC Research and Development Needs webpage.
The Chemistry/Instrumental Analysis Scientific Area Committee (SAC) recently reviewed and approved the recommendation from various subcommittees to submit the following potential standards or guidelines to a Standards Developing Organization (SDO).
• ASTM E1388- Standard Practice for Sampling of Headspace Vapors from Fire Debris Samples (Submitted by Fire Debris and Explosives Subcommittee). The existing ASTM document has been reviewed by the OSAC. Suggested changes have been submitted to ASTM.
• ASTM E1412- Standard Practice for Separation of Ignitable Liquid Residues from Fire Debris Samples by Passive Headspace Concentration with Activated Charcoal (Submitted by Fire Debris and Explosives Subcommittee). The current ASTM E1412 has been reviewed by the committee and suggested changes have been submitted to ASTM.
• ASTM E1386- Standard Practice for Separation of Ignitable Liquid Residues from Fire Debris Samples by Solvent Extraction (Submitted by Fire Debris and Explosives Subcommittee). This has been approved by the subcommittee for movement to the SAC for consideration as a guide (the Registry request packet is currently being assembled).
• Standard Practices for Measurement Traceability in Forensic Toxicology (Submitted by Toxicology Subcommittee). The Chemistry SAC approved the final Traceability packet. The document was subsequently sent to an SDO to begin the next phase.
• Standard Practices for Method Validation in Forensic Toxicology (Submitted by Toxicology Subcommittee). The Chemistry SAC approved the final Method Validation packet. The Method Validation document was subsequently sent to an SDO to begin the next phase.
• Guidelines for Opinions and Testimony in Forensic Toxicology (Submitted by Toxicology Subcommittee). The Chemistry SAC approved the final Opinions and Testimony packet and the document was subsequently sent to a SDO to begin the next phase.The designated SDOs will subsequently announce when these standards and/or guidelines are available for public comment.

(OSAC Meetings for OSAC Members and Invited Guests) The next full OSAC meetings are scheduled for summer 2016 in Phoenix, AZ. Instead of meeting as one large group, the OSAC will meet separately in a series of smaller meetings.
o (OSAC Meeting Breakout #4) Biology/DNA SAC + 3 Subcommittees, and Crime Scene SAC + 7 Subcommittees, Aug 23-26, 2016
o (OSAC Meeting for OSAC Members and Invited Guests) The following full OSAC meeting is scheduled for April 3-7, 2017, tentatively in Leesburg, VA.
o OSAC public meeting date and times will be announced shortly.
Biology/DNA SAC News- The Biology/DNA Scientific Area Committee approved the Wildlife Forensics General Standard document in July 2016 and the document was submitted to the American Academy for Forensic Sciences (AAFS) Academy Standards Board (ASB) for consideration. The document will now move through the ASB process where it will hopefully be converted into an SDO approved standard which can then be considered by the OSAC for inclusion on the Registry of Standards. Kathy Moore, Chair of the Wildlife Forensics Subcommittee gave a presentation at the Green Mountain conference the last week of July. The Biology/DNA SAC, the Biological Methods Subcommittee, the Biological Data Interpretation & Reporting Subcommittee, and the Wildlife Forensics Subcommittee will be meeting August 23-26 in Phoenix, Arizona to continue work on several documents, including a terminology document for forensic biology, validation of probabilistic genotyping systems, internal validation of existing mixture interpretation procedures, and training/education requirements for serologists and DNA analysts. Dr. George Herrin, Chair of the Biology/DNA SAC is scheduled to give a presentation at the 27th ISHI meeting in Minneapolis, MN on September 29, 2016 covering recent SAC activities.
Standards Developing Organization (SDO) News
ASTM Committee E30 on Forensic Sciences Recent Activity
E30.01 Criminalistics
The following work items are underway:
Proposed Revision: WK55598 - Standard Guide for Forensic Paint Analysis and Comparison is a work item revision to existing standard E1610-14 (LINK EXPIRED)
Proposed Revision: WK55599 - Standard Guide for Using Infrared Spectroscopy in Forensic Paint Examinations is a work item revision to existing standard E2937-13 (LINK EXPIRED)
Proposed Revision: WK55604 - Standard Practice for Identification of Seized Drugs is a work item revision to existing standard E2329-14 (LINK EXPIRED)
Newly Approved ASTM Standard
E1588 - Standard Practice for Gunshot Residue Analysis by Scanning Electron Microscopy/Energy Dispersive X-Ray Spectrometry has been revised to E1588-16a developed by Committee E30.01, ASTM BOS Volume 14.02.
E30.12 Digital and Multimedia Evidence
The following work item is underway:
ASTM WK55615 Revision of E2825 - 12 Standard Guide for Forensic Digital Image Processing
------------------------------------------------------------------------------------------------------
The American Academy of Forensics Science (AAFS) Academy' Standards Board (ASB) announced their upcoming consensus body meetings.
All Consensus Body meetings are open to all interested parties, but only members may vote. For the full calendar, call in information and agenda, visit the ASB Upcoming Events Calendar.
August 18th 1:00 pm to 4:00 pm EDT - Disaster Victim Identification
September 1st 1:00 pm to 4:00 pm EDT - Patterned Injury
September 8th 2:00 pm to 5:00 pm EDT - DNA
September 15th 1:00 pm to 2:00 pm EDT - Firearms and Toolmarks
September 16th 1:00 pm to 4:00 pm EDT - Medicolegal
September 20th 2:00 pm to 5:00 pm EDT - Bloodstain Pattern Analysis
OSAC Featured Unit: Wildlife Subcommittee
By Kathy Moore, M.S., Chair of OSAC Wildlife Subcommittee
Wildlife forensics differs from human forensics in a variety of notable ways. In human forensics, the focus is on identifying the perpetrator of a crime. In wildlife forensics, the first step is often determining whether or not a crime has occurred, which depends on whether a protected animal or plant species is involved—and there are many, many species to consider. Identifying the species (such as black bear) is often what is required—rather than identifying the individual (Smokey Bear). Species-level identifications are based on class characteristics, diagnostic for particular species. These may be either physical, morphological characteristics or genetic sequences. Due to the great diversity of species that must be considered in wildlife forensics investigations, the development of databases used for individualization and geographic assignment is prioritized by different laboratory programs. In contrast to human DNA analyses, there are no standardized commercial kits to be used in DNA analysis of wildlife species, and loci are not standardized among laboratories that analyze the same species. Wildlife forensic laboratories also tend to be small, where an analyst is responsible for everything from sample login, through analysis, to generation of the report. Crimes investigated by wildlife forensic scientists include:
• Poaching (killing wild animals in violation of the law)
• Trafficking in protected animals/plants or products
• Seafood fraud
• Animal cruelty
• Animal attacks
• Human crimes if animal evidence is probative
In 2012, SWGWILD compiled several published sources to estimate that the transnational illicit wildlife trade, including wildlife, timber, and illegal fishing, is worth approximately $53 billion a year. A 2013 Scientific American article indicated that "illegal wildlife trafficking is an estimated $19-billion-a-year industry, which makes it the fourth most lucrative illicit activity in the world after the drug trade, counterfeiting and human trafficking." Wildlife forensic scientists are few in number, but analyze evidence related to this large and complex international trade, as well as suspected violations of domestic game and animal protection laws.
Examples of evidence items that could come across the lab bench of a wildlife forensic analyst include: thousands of pounds of suspected mislabeled fish, a feather cape, elk steaks from a hunter's freezer, exotic leather cowboy boots, dried shark fins, and intricate carvings made of ivory or bone. Most analysts specialize in certain techniques (such as DNA, morphology, or veterinary pathology) and taxonomic groups (mammals, reptiles, marine animals, etc.), as taxonomic groups may be governed by different laws, and considerations of species' biology and distribution can impact interpretation of analytical results.

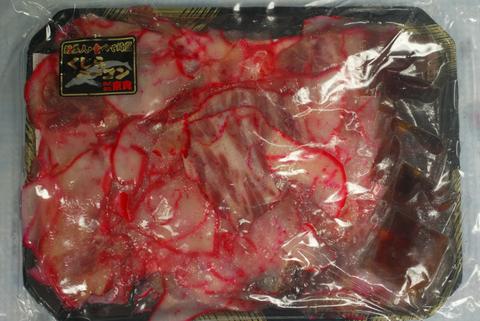
Two examples of species protected domestically by the Marine Mammal Protection Act and the Endangered Species Act, the scrimshawed sperm whale tooth, and a package of bacon made from Bryde's whale. Both are protected from international trade by the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES).
The Wildlife Forensics Subcommittee of OSAC focuses on standards and guidelines related to taxonomic identification, individualization, and geographic origin of non-human biological evidence based on morphological and genetic analyses. Kathy Moore, M.S., chair, works for NOAA Fisheries' Marine Forensic Laboratory, and has worked doing wildlife forensic methods development and/or casework for more than 20 years.
With input from members as well as guests representing state, national and international agencies, the subcommittee has completed work on its first document, "Wildlife General Standards", which has been approved by the SAC and sent to the AAFS's Academy Standards Board. Currently, the Subcommittee is working on the following projects:
• Wildlife Forensic Report Writing Standard
• DNA Standards
• Morphology Standards
• Education and Training Standards
• Biology SAC-wide terminology document
For additional information, please contact forensics [at] nist.gov (forensics[at]nist[dot]gov).

