Summary
Thermal desorption ambient mass spectrometry decouples analyte desorption and ionization. Here, analytes are thermally desorbed directly from a primary surface or a wipe collection using conductive, convective, and/or radiative modes of heating. This mode has been implemented extensively in the trace detection arena for rapid and reproducible analysis of wipe-collected samples. Similarly, recent efforts have demonstrated the utility of thermal desorption of wipe-collections for forensic screening.
Description
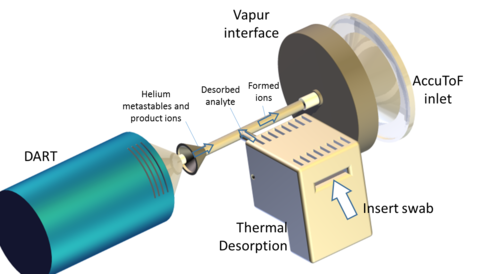
The Surface and Trace Chemical Analysis Group is involved in a number of research avenues for the rapid screening of wipe-collected samples for the security and forensic chemistry fields. Similar to the widely-deploy ion mobility spectrometry (IMS) systems for screening, this is accomplished through the thermal desorption of analytes from a wipe material. STCAG has research avenues aimed at developing platforms for coupling thermal desorption (TD) with direct analysis in real time mass spectrometry (DART-MS), as well as implementing alternative modes of wipe heating. Below are a few examples of research conducted in these areas.
The coupling of resistance-based heating and DART-MS has enabled rapid and reproducible detection of wipe-collected samples. This configuration has aided in reducing sample-to-sample variability seen with classical and off-axis DART configurations from approximately 45 % relative standard deviation (RSD) to less than 15 % RSD. The speed of analysis also poises the TD-DART-MS configuration to transform the current casework screening operating procedure. The reproducibility of this method has also enabled rapid quantification of samples. In addition, the confined geometry demonstrated considerable benefits for moving from the traditional DART source gas of helium to nitrogen. Recent work has focused on demonstrating the use of TD-DART-MS for a range of applications and on better understanding the fundamentals of the technique. The platform has been shown to successful detect a range of compounds of forensic interest from synthetic opioids to rodenticides to seed-based toxins and has been successfully applied to the analysis of drug residues from forensic cases. Using a combination of analytical responses and flow visualizations, a broader understanding of the principles of confined DART-MS configurations have been established, such as understanding how the role of mixing allows for superior performance of nitrogen as a source gas.
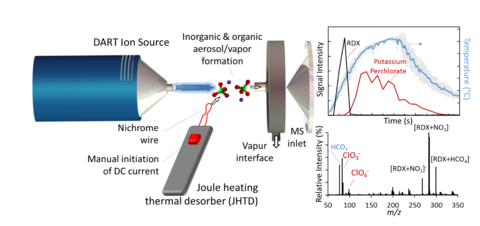
The trace detection of both traditional military grade explosives as well as homemade explosives (HMEs) and improvised explosive devices (IEDs) is a focus of many projects within STCAG. While much research has been conducted on military grade explosives, we aim to investigate the unique (and greatly variable) chemical properties of homemade explosives – ranging from high volatility peroxide-based to the low volatility fuel-oxidizer explosives. Explosive devices often contain a range of organic and inorganic species requiring robust detection schemes. The STCAG has a number of research avenues investigating methods for generating rapid heating ramps to elevated temperatures that enable the thermal desorption of both more volatile organic explosives/compounds and less volatile inorganic and oxidizer compounds. The traditional resistance-based thermal desorbers maintain a steady temperature aimed at being optimal for the majority of traditional explosives (~230°C to 250°C), however inorganic oxidizers have low vapor pressures at these temperatures. We are developing the use of infrared heating of the wipe to achieve elevated temperatures during a discrete emission interval. Under the mode of operation, the thermal desorber maintains an approximately steady temperature near 200°C to desorb organic materials, followed by a brief (5s to 15s) interval of infrared thermal desorption (IRTD) to achieve elevated temperatures (450 °C to 550 °C). The IRTD can be coupled with the DART-MS configuration as discussed above. This configuration has demonstrated the thermal desorption of both organic (nitroglycerin and pentaerythritol tetranitrate) and inorganic (potassium chlorate and potassium perchlorate) explosives.

In addition to IRTD-DART-MS, STCAG is also investigating the use of high temperature resistive Joule heating thermal desorption (JHTD) coupled with DART-MS for the detection and characterization of organic and inorganic explosive mixtures. JHTD is accomplished by passing current through a nichrome wire in discrete intervals to achieve rapid heating rates and elevated temperatures. Similar to IRTD-DART-MS, the implementation of discrete and rapid heating ramps enables the thermal desorption of both organic and inorganic compounds at their respective optimal temperatures. In this configuration, the intimate contact between compounds and the heating element also demonstrate the decomposition of inorganic oxidizers. Ongoing work seeks to implement this 1D wire configuration into a 2D metal mesh, advancing applicability of this platform.