Summary
This NIST-wide collaborative project aims to improve biotechnology and medical diagnostics by increasing the resolution and precision of measurements of cells through development of an optofluidic cytometer and associated measurement infrastructure. Critically, the work involves interplay of physical models, mathematical analysis, engineering, and biology expertise to achieve new measurement capabilities with unprecedented uncertainty.
Description
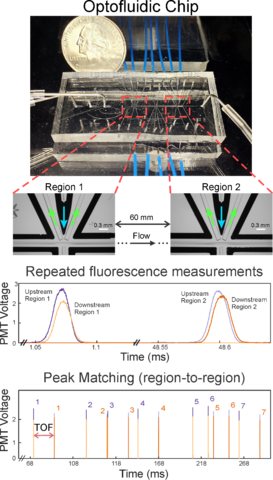
Flow cytometers are commercial instruments that provide rapid multiparameter measurements of single cells, which makes them indispensable in basic research and clinical studies of diseases and drug therapies. Measurements of cellular attributes can include indicators of cell size and shape and well as fluorescence measurements that are proportional to amount of a specific material to which the fluorophore is bound (e.g. a fluorescently labeled nucleotide or an antibody that links to a specific protein).Large data sets covering many cellular properties can be acquired, but the extent to which the data can be fully utilized is limited by an underdeveloped measurement uncertainty.
We are developing state-of-the-art optofluidic cytometers that incorporate optical waveguides to deliver light to and collect light from specific regions of microfluidic networks. The core of the project involves utilizing these integrated optical elements to create multiple measurement regions along a flow path. Thus, each cell is measured several times. A goal of the project is to create robust designs and measurement strategies that reduce the impact of variations due to system geometry and operating parameters.
To achieve these goals, we are working to solve technical challenges associated with fabrication, flow stability and light collection, while simultaneously collaborating on the development of mathematical concepts that not only improve fundamental measurement uncertainty but also use those metrics to control system operation in real time. By solving problems that become apparent when trying to repeat measurements, we are able to both improve our understanding of limits of traditional flow cytometers and achieve new capabilities for cell counting accuracy and tracking, analyze coincident events (doublets), and improve uncertainty and discrimination of small changes or rare objects within a complex sample.

