Summary
Broadband coherent anti-Stokes scattering (BCARS) microscopy is an imaging modality that probes the chemical content and abundance of samples without the addition of exogeneous fluorophores or labels which may perturb the intrinsic chemistry and function of cells and tissues. Underlying this technology is the use of ultrafast laser sources to drive molecular vibrations, with each molecule having a unique vibrational signature. In comparison to traditional [spontaneous] Raman spectroscopy and microscopy, BCARS is orders-of-magnitude faster. This project aims to develop the microscope system and analysis tools, revealing information about the chemical landscape in cells, tissues, and materials with unprecedented information breadth.
Description
BCARS image of a tissue sample
Impact
There is a need for label-free chemical microscopy in medicine, biology and materials science. Most of the current methods use chemical labels that often disturb the distribution and nature of chemical components being investigated. The method we are developing enables noninvasive and rapid collection of Raman spectra for imaging.
BCARS can be used to track cell signaling processes and can provide functional readouts of cell differentiation, allowing researchers to obtain cell responses to biomaterials in real time and on a cell-by-cell basis.
BCARS can be used to acquire high-resolution chemical maps of pharmaceutical tablets, including information on morphology of active ingredients, 10 to 100 times faster than spontaneous Raman scattering.
All researchers who use Raman imaging methods will benefit from our work, including biomedical researchers (and, potentially, clinicians), pharmaceutical industry scientists, geologists and others. Additionally, laser manufacturers have been influenced by our work; PolarOnyx, Toptica, Time-Bandwidth and Spectra Physics Laser have all developed and are marketing laser sources to enable BCARS.
Approach
CARS provides a signal that contains the Raman response of interest for performing label-free chemically sensitive microscopy. In CARS, a vibrational coherence is generated when a pair of photons (pump and Stokes) interact with the sample to excite a vibrationally resonant Raman mode at frequency ωvib = ωpump – ωStokes. A third (probe) photon is inelastically scattered off this coherent excitation, and anti-Stokes light (ωas = ωpump – ωStokes + ωprobe) is emitted from the sample.
The CARS signal has a frequency-independent non-resonant component and a frequency-dependent resonant component. The non-resonant component is entirely in phase with the driving field of the laser and gives us no information about the chemical nature of the sample. The resonant component contains the chemical information and has a frequency-dependent amplitude. It is out of phase with respect to the driving electric field of the laser. The resonant component contains elements with the same bandshape as the spontaneous Raman signal.
We obtain a broadband vibrational spectrum at each laser shot by using broadband Stokes light. The Stokes light contains 3000 cm-1 of bandwidth.
Major Accomplishments
We have had several "firsts" in this project. We were the first to demonstrate broadband CARS microscopy. With this system we obtain 3000 cm-1 spectrum at each laser shot, allowing us to rapidly image chemically complex systems, such as the polymer blend shown in the figure below. This image was obtained at 17 ms/pixel, about 50 times faster than can be obtained with spontaneous Raman spectroscopy.
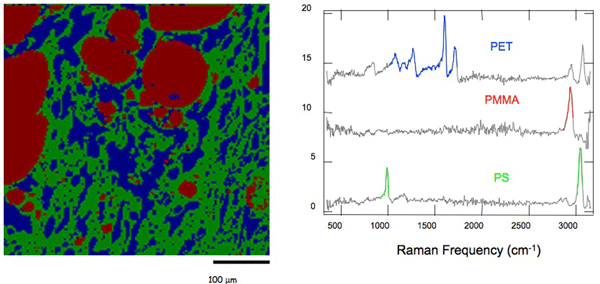
The spectra obtained from CARS includes a resonant and nonresonant component, which made it difficult to extract the Raman spectrum of interest. We have developed a deterministic mathematical approach to extracting the resonant (Raman) signal based on a time-domain Kramers-Kronig (TDKK) transform. The TDKK treatment makes the CARS signal linear in analyte concentration and quantitative. We show, based on imaging of a polymer blend below, that even minor components can be detected and quantitatively accounted for when the CARS signal is transformed in this way.
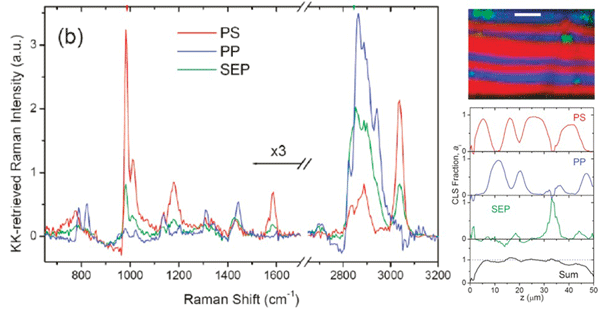
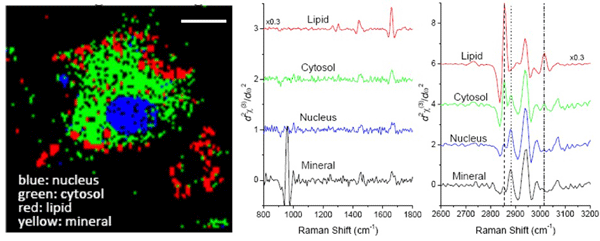
This TDKK transform allows us to take advantage of intrinsic heterodyne amplification of the weak resonant signal and allowed us to be the first group to obtain a full fingerprint and CH-stretch vibrational spectra simultaneously in biological cells. The chemical signatures contained in the combined CH and fingerprint spectral region allow us to discriminate differentiated cells from stem cells, as shown in the figure below.
Associated Product(s)
Download our project brief (PDF).

