Summary
The purpose of this project is to create rheological measurement methods and tools, focused on using minimal fluid volumes, to measure local flow dynamics and related structure and properties of polymer systems, including surfactant solutions, protein solutions, and suspensions, and of their self-association. Measurements combine rheometry, scattering, microscopy and/or particle imaging to quantify extensional behavior, viscosity at high shear rates, biomolecule self-association and aggregation, and viscoelastic instabilities.
Description
We aim to:
- Develop measurement capabilities to relate solution structure and viscosity through novel combinations of unique small volume, high shear rate rheometers with scattering and particle-imaging techniques. A major application of these capabilities is understanding self-association in fluids, such as of protein aggregation, including quantification of surface chemistry, surface adsorption, flow conditions, aggregate size, and surface kinetics, so that the formation of undesirable aggregates may be avoided. The following objective supports this broader objective.
- Develop 3D soft-matter flow metrology capabilities to resolve local flow, stress, and structure under high-shear conditions that lead to flow instabilities. One application involves organ-on-a-chip concepts and the measurement of organ mechanical properties.
Major Accomplishments
Microcapillary rheometers that use only 10 uL of solution have been developed
Tiny volume is a key requirement for the biopharmaceutical industry during drug formulation development, because hundreds of different combinations must be tested, and the amount of protein available for such tests is limited at that stage. MedImmune (now Astra-Zeneca) thus set up a CRADA with us to develop a rheometer and other diagnostics that needed less than 100 µL of solution.
As a result, a versatile and accurate microcapillary rheometer has been developed. This instrument uses less than 10 µL of fluid, is accurate and precise to a few percent, can measure a two-decade span of shear rate in approx. 10 minutes, and is temperature controlled ((0 – 80)°C). (Journal of Pharmaceutical Science 2015) Using this instrument, we demonstrate that various protein antibody solutions may exhibit Newtonian or non-Newtonian rheology, emphasizing the need to measure at a range of shear rate, temperature and other solution conditions.
The dynamics of concentrated self-associating protein solution was measured
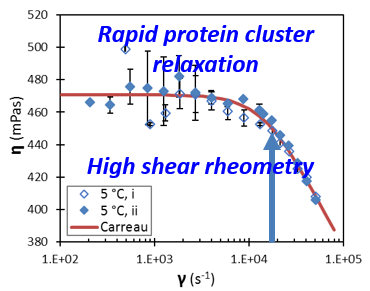
(Biomicrofluidics 2016) fse_sdh_lysozyme_shear_thinning
Improvements to this technology, including smaller form factor, continue through the NIST on a Chip initiative’s flow metrology development, published in Rheol Acta. Other development is controlled temperature of such rheometers.
While exploring the rheology of shear-banding wormlike micelles we discovered a surprisingly long entrance length before its capillary flow is fully developed, requiring L/d > 100. Particle tracking, as described below confirmed this behavior, and protocols for measuring the true viscosity were demonstrated. Other fluids (which don’t shear band) do not require such protocols. The flow behavior to very high shear rate and simultaneous measurements of structure and/or alignment was determined.
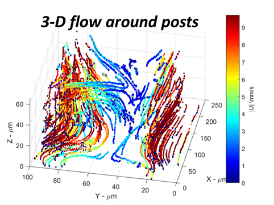
High-resolution particle-tracking flow metrology characterizes turbulent structures and other flow instabilities
Particle tracking is another essential aspect of our flow metrology research. Most importantly, this analysis determines when flow is suitable for measurement of fluid viscosity and steady-state structure, or when conversely flow instabilities occur at high shear rates encountered during manufacturing and use. Among the particle tracking methods used, holographic microscopy has exceptional temporal and spatial dynamic range. Our microscope can resolve particle position to 10s of nm over a depth of field of 200 um, and flow velocities (from 0.001 to 50) mm/s.

fig: fse_sdh_holog_scheme
Using these features, we have compared with flow calibration (Experiments in Fluids 2017) and tested viscoelastic flow. When fluids are viscoelastic (such as polymer solutions), normal stress differences can cause flow instability, and even turbulent flow, at negligible Reynolds number, especially when the flow has curved streamlines. We have reported studies of:
Spatial instabilities of flow in rectangular channels (Physics of Fluids 2017) and cross-slot geometry
(submitted to Soft Matter)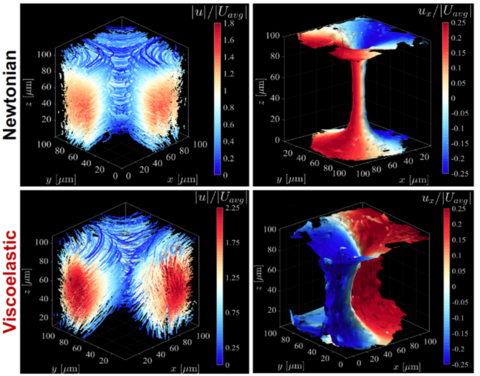
3D holographic particle-tracking velocimetry data and velocity isosurfaces for Newtonian (top panel) and viscoelastic (bottom panel) fluids in the cross-slot. - Temporal instabilities of flow through a constriction (Soft Matter 2018), around an obstacle (Journal of Fluid Mechanics 2019) or in cross-slot
Self-sustaining mechanisms of turbulence (Physical Review Letters 2019)
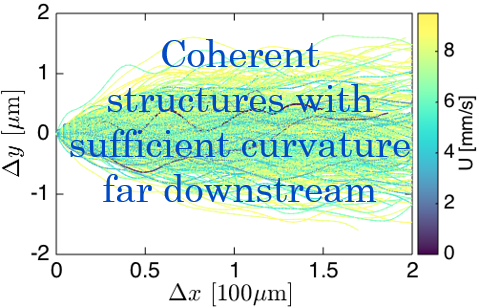
Particle tracks exhibit sufficient curvature to sustain turbulent flow. fse_sdh_turbulent_tracks
New high shear RheoSANS approached validated
Particle tracking and pressure gradient measurements demonstrated that some shear banding fluids surprisingly are slow to develop a rheometric flow for rheological measurements. (Journal of Rheology 2020) When rheometric flow is achieved, a depth sectioning technique, first demonstrated by Fernandez-Ballester, enables high shear rate rheoSANS measurements on a beamline.
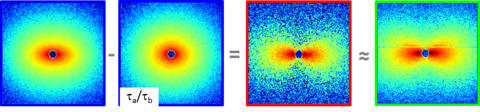
fse_sdh_urheoSANS_depth_section (Rheologica Acta 2018)
Flow-induced birefringence measures alignment
Birefringence measures alignment of the fluid and is a measure of the stress in the fluid. We combine 3D flow metrology (particle tracking) and 2D stress measurement (birefringence) to determine their relationship in steady and transient flow.
Protein adsorption and aggregation correlated
Globular proteins in solution adsorb to solid, liquid and air surfaces. These surfaces are sometimes a pathway to aggregation, and this instability must be prevented. Reversible and irreversible adsorption of NIST mAb and other proteins to various surfaces (stainless steel, alumina, and monolayer with different surface charge) has been measured by quartz crystal microbalance and neutron reflectometry and correlated to the amount of aggregated particles formed during flow. When high solution concentrations are in contact with stainless steel, for example, the molecules adjacent to the surface are conformationally altered, and in this condition most particle aggregates form. We are developing approaches to determine transport mechanisms leading to this surfaced-induced protein aggregation.
Rheological effects of particle shape and friction determined
Learn more by reading "A Crack in the Mystery of 'Oobleck'—Friction Thickens Fluids."
Measuring fluid properties and structure establishes the mechanisms that give rise to non-Newtonian rheology. One striking phenomenon is shear thickening, when the fluid viscosity rises sharply at high stress. To test interactions between particles, whether frictional or lubricating, we measured other stress components such as the first normal stress difference. Exploiting the strong temperature dependence of the viscosity of our glycerol based suspending fluid, we could readily access the stresses needed to initiate shear thickening while avoiding the flow instabilities that accompany high shear rates. This allowed us to explore shear thickening over an exceptionally wide range of volume fraction, and to measure normal forces too. Frictional force chain interactions between particles span across the fluid only at high stress. (Physical Review Letters 2016) Research elsewhere continues to test how frictional interactions arise and can be controlled.
Particle shape or directional interactions also affect fluid properties dramatically. Proteins and even mainly commercial particles are anisotropic. To help take colloidal rheology understanding beyond uniformly charged spheres, we have prepared and studied solutions of model cubelike particles.

(Soft Matter 2015)
Associated Product(s)
We use NIST RM-8671 in this work.

