Summary
Therapeutic protein solutions are highly concentrated during manufacturing purification processes and in finished dosage. At such concentrations, the fluid rheology may be complex, creating the need for a convenient accurate method to measure viscosity. Here we develop rheology tools to help evaluate protein stability across a range of solution conditions and protein concentrations that are relevant to manufacturing and product formulation. One tool, is a capillary viscometer that requires only a few micro-liters of solution to probe the range of flow rates and temperatures of interest. Another tool to be developed will expand the dynamic range of rheo SANS at the NIST neutron user facility (NCNR), i.e. an instrument for simultaneous rheology and neutron scattering. A third method probes concentrated protein films adsorbed to interfaces where aggregation may take place, and where properties are suspected to correlate with solution stability. Teaming with collaborators, we test industrially relevant materials, evaluate structure and aggregation kinetics, and contribute to the characterization of an antibody standard reference material SRM.
Description
Intended Impact
Why does the health-care industry care about rheology and scattering of protein solutions?
- Operations. Viscosity is a critical and fundamental constitutive input for drug delivery, device design and manufacturing. For example, high viscosity leads to poor syringeability due to high injection force required.
- Health. Aggregation of antibodies is a potential health risk, and viscosity and scattering measures are diagnostic of aggregation. Our collaborators from U. Delaware (see sidebar) note that the concern here, not yet verified, is whether aggregates may induce adverse immune responses.
This industry therefore needs a convenient method to measure viscosity that avoids potential problems with accuracy and excess sample volume, and which is conveniently part of their quality measurement system. It also needs techniques that can be used to predict protein solution stability, so as to inform molecular design and formulation development. This project is thus developing tools and analyses to measure properties of protein solutions and to identify measures that predict protein solution stability.
Objectives
- Develop and demonstrate rheology methods and tests that are most predictive and indicative of protein solution instability.
- Measure viscosity of an antibody SRM (standard reference material) under development.
- Couple techniques together: rheology/fluorescence; rheology/particle characterization; rheology/neutron scattering.
Technical Approach
Capillary viscometry is a well-established and robust measurement technique. It basically involves measuring a driving pressure and a corresponding flow rate through a capillary or flow channel. Here the challenge is to make testing very small volumes convenient. Specifically, to measure small flow rates ((0.1 to 100) nL/s), we developed a protocol to optimize the performance and statistics of a commercial microfluidic flow meter. And to quickly handle and load small volumes into the instrument, a micro-pipette simply dips into a standard sample vial. With minor modification, testing multi-well plate sample arrays would be feasible. To measure a wide range of viscosity (from (0.5 to 2000) mPa s), we use different pressure sources capable of delivering from 3 Pa to 100,000 Pa, and we employ micro-pipettes of different size.
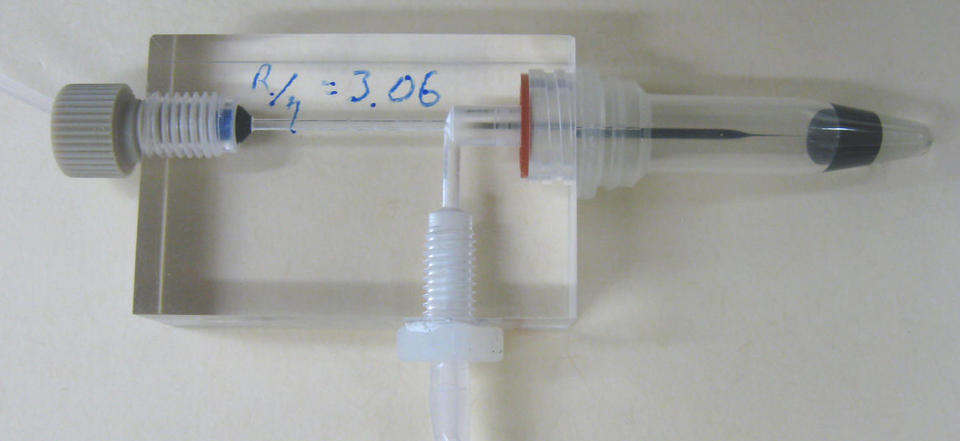
A photo of the micro capillary viscometer, which uses only microliters of sample.
This method (combined with scattering and other biophysical techniques) is now exploring correlations between viscosity, solution stability and cluster dynamics. Measuring the effects of pH, temperature, and salts is central to improving product yield during concentration of monoclonal antibodies (mAbs) in purification processes. Complex behavior is observed, and in some cases, flow may induce aggregation. The effects of pH and concentration are being measured over a much broader range than previously reported by others. The results demonstrate the failure of existing colloidal rheology models. This result strongly motivates theoretical modeling and model experiment studies. Specifically, two approaches are in progress: 1.) model colloids that possess simple directional interactions are being tested to find out the properties that deviate from existing models and 2.) a new theoretical model for protein solutions is being developed.
Air-water and oil-water interfaces are present throughout manufacturing and in final protein therapeutic products. The rheology of proteins at these interfaces is of special relevance to aggregation phenomena, because there proteins collect, may slightly denature and begin to aggregate. Interfaces therefore provide a natural model for instability. Unfortunately, interfacial rheology is not widely available, and data is thus sparse. To meet this need, a new method for dilatational interfacial rheology is being adapted for protein solutions, so that it will use smaller volumes and have wider dynamic range. These properties will be measured for a few proteins of interest under a wide range of solution conditions, and compared with bulk aggregation kinetics.
Major Accomplishments
- A versatile and accurate micro-viscometer has been developed at NIST and duplicated at MedImmune. This instrument uses less than 10 uL of fluid, is accurate and precise to a few percent, can measure a two-decade span of shear rate in approx. 10 minutes, and is temperature controlled ((0 – 80) °C). Using this instrument, we demonstrate that various protein antibody solutions may exhibit Newtonian or non-Newtonian rheology, emphasizing the need to measure at a range of shear rate, temperature and other solution conditions.
- Circular dichroism, rheology and neutron scattering demonstrate that a simple colloidal model of protein monomers must be adjusted to account for changes in the properties of the monomer with changing pH (as some anticipated) and monomer concentration (surprisingly).

