Summary
Far-infrared (or terahertz/THz, ca. 25 to 300 micron wavelength) femtosecond pulsed laser and Fourier-transform infrared methods are employed to measure biomolecular spectra in the condensed phase. This region of the spectrum is particularly sensitive to the detailed structural and environmental properties of these complex hydrogen-bonded species. Research interests in this area include: spectral measurement of amino acids and small peptides in reverse aqueous micelles, application of solid-state density functional theory to interpret measured terahertz spectra; time-resolved THz spectroscopy to directly monitor photochemical isomerization reactions and development of circular dichroism methods in this wavelength range.
Description
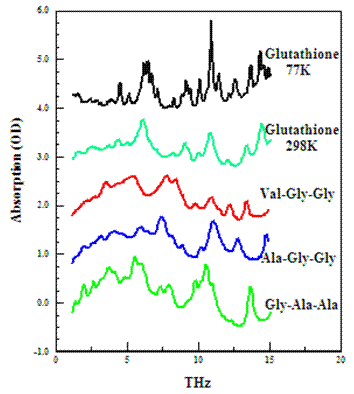
Far-infrared spectra for selected tripeptides in the solid state showing unique spectral differences arising from molecular and crystalline structures.
We employ novel complimentary measurement and theoretical techniques to explore the low frequency intramolecular dynamics of model biological molecules including amino acids, short peptides with constrained structures, proteins with well-defined tertiary structures and DNAs. Our current efforts focus on obtaining broadband THz spectra of protein constituents (e.g., amino acids serine, cysteine, proline, tryptophan and their substituted analogs and enantiomeric DL mixtures), synthetic peptides (e.g., dipeptides, tripeptides, poly(alanine) and sugars using pulsed laser and FTIR methods.
For biosamples examined as powders in transparent polyethylene matrix, solid-state periodic boundary calculations using Dmol3, CPMD, VASP and CHARMM are employed to determine the origin of internal and external vibrational modes. These measurements, in conjunction with theoretical modeling are envisioned as a means to extract important protein-folding rates and determine mechanisms responsible for DNA base pair hydrogen-bonding, surface interactions and helix dynamics.
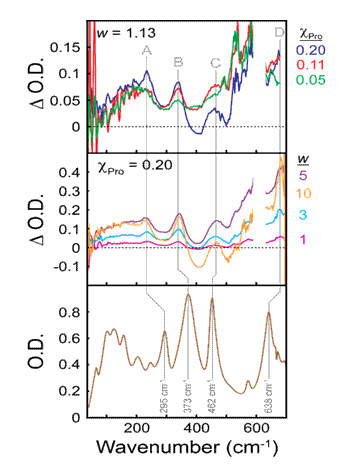
Studies of L-Proline embedded in nano-sized reverse aqueous micelles revealed concentration and size dependent behavior related to the surfactant charge and surface interactions. Detailed THz studies of H2O and D2O containing micelles were also performed. Comparisons between L- and DL-Proline measured THz spectra and periodic boundary calculations are underway.
These investigations use state-of-the-art, kilohertz repetition-rate amplified, approximately 45 femtosecond pulsed lasers for broadband (0.2-10) THz generation and detection including GaAs antennas as well as Lithoium Niobate, GaP and ZnTe nonlinear crystals for broadband spectroscopic determinations and imaging. A modified FTIR with a silicon-coated beamsplitter and room temperature DTGS detector is routinely used for survey transmission spectroscopy in the 40 cm-1 to 700 cm-1 spectral range. See facilities for further details.
Major Accomplishments
Terahertz materials spectral database (ca. 100 common items) in the NIST Chemistry WebBook.

