Summary
Discovery of novel materials for hydrogen gas storage has prompted new avenues of research towards developing hydrogen as an alternative fuel. However, optimizing candidate materials' properties for high hydrogen adsorption per unit weight, rapid desorption kinetics, and low hysteresis are still difficult to attain. To aid in this goal, we are focused on developing new measurement modalities to find metal alloy systems capable of meeting these stringent characteristics. In our group, infrared emission imaging and FTIR spectroscopy are applied towards measuring adsorption/desorption dynamics and identifying metal-hydrogenated species. Through collaboration with members of the Materials Measurement Laboratory, thin combinatorial films of various metal alloys and doped wedged metallic films have been investigated using these measurement tools. These measurements have helped identify prospective materials capable of meeting DoE and potential industrial end-user requirements. Studies include measurement of specifically designed Fe-doped magnesium nanofilms optimized for low-temperature, high reaction yields and adsorption/desorption rates.
Description
Infrared emission imaging and FTIR reflection spectroscopy is applied as a means to monitor thin-film hydrogenation reactions in temperature-controlled high pressure reaction chambers. Candidate alloyed and doped thin metallic films (<500 nm) are grown in-house on epi-alumina or silicon substrates and placed in these chambers to monitor adsorption/desorption kinetics or identify hydrogen-containing metallic species as a function of time. Specifically, wedged-shaped films permit detailed monitoring of reaction front migration along the film and this allows extraction of reaction rates, cycling hysteresis and underlying enthalpies of reaction (see Figure). Studies of the same samples under reflection FTIR conditions help identify creation of newly hydrogenated species within the metallic films.

Individual frames from time-resolved IR emission measurements for a Mg-4Fe 4 atomic % film hydrogenated at 0.1 MPa and 383 K. The boxed area at the bottom is a control for a 10 nm Pd overlayer on the alumina substrate. The time on each image denotes the elapsed hydrogenation time reaching the film end and reaction completion in 111 seconds.
Studies of lightly doped iron in magnesium (4 at. % Fe:Mg) have shown that these films adsorb up to 7 wt% hydrogen with reaction front mobility of ca. 37 nm/s at 423 K (150 C). Hysteresis for three adsorption/desorption cycles was found to be minimal after the first cycle (see below). These results suggest that properly grown Mg-Fe powders of 1-2 micron size can be fully charged with hydrogen within 1 min at temperatures near 150 C (423 K) enabling practical hydrogen storage applications.
Additional Technical Details:
Please see: http://www.nist.gov/mml/msed/thin_film_nanostructure/hydrogen-storage.cfm for a more detailed explanation of employed methods and measurements conducted with MML.
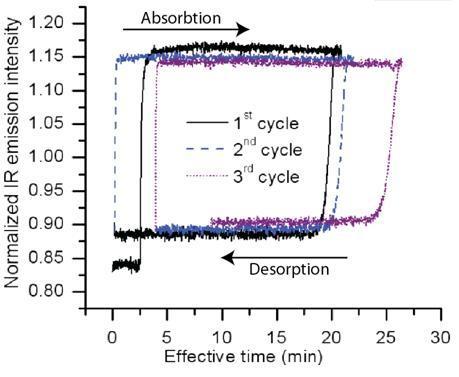
IR emission intensity hysteresis of hydrogen absorption under 0.1 MPa hydrogen pressure and desorption under dynamic vacuum of a wedge Mg-4Fe at.% film at 403 K. The IR emission intensity increases/decreases when hydrogen is absorbed/desorbed.
Major Accomplishments
- Demonstrated infrared emission imaging as efficient measurement method for monitoring reaction front migration in metal alloy films.
- Developed InSb camera imaging method to measure hydrogen reaction with nanolayer films of candidate hydrogen storage materials from 50 ms to hour timescales.
- Applied methods for measuring reaction rates, hysteresis and enthalpies for pure and (Mn, Ti, Y, Fe)-doped Mg films.
- Measured hydrogenated product film FTIR reflection spectra of various films (e.g., Y-Mg) to extract Mg-H and other product ratios. Enabled product species identification and comparison to TEM and X-ray compositional data of identical films.
- Participated in identifying doped 4 at% Fe:Mg as a strong candidate material for low-temperature adsorption-desorption hydrogen storage.
- US Patent #9,061,907 awarded on June 25, 2015 for a "Two-Component Structures Providing Fast Low-Temperature Charging of Mg with Hydrogen" for the Fe:Mg composite alloy for hydrogen storage material (with Leonid Bendersky and C. Y.Tan, MML) filed Sept. 19, 2012.

