Advanced electron paramagnetic resonance spectroscopy for structural characterization of bionanomaterials for bioscience and biotechnology
Summary
Recent advances in nanofabrication and nanobiotechnology rely on biomacromolecules. In biotechnology, biomacromolecules at nanometer length scales are either themselves therapies (monoclonal antibodies, for example) or are used in combination with nanoscale drug-delivery vectors. All these examples rely on nanoscale structural measurements to enable design and engineering of robust, reliable systems. Electron paramagnetic resonance (EPR) spectroscopy is a particularly powerful tool for nanoscale systems lacking long-range crystalline order — the regime into which many biomacromolecules fall. This project focuses on structural measurements of biomacromolecular systems important in biomedical research and biotechnology. Eventual extension to measurements in cellulo will be of interest to the biotechnology sector as a potential analytical tool for biologic structural characterization.
Description
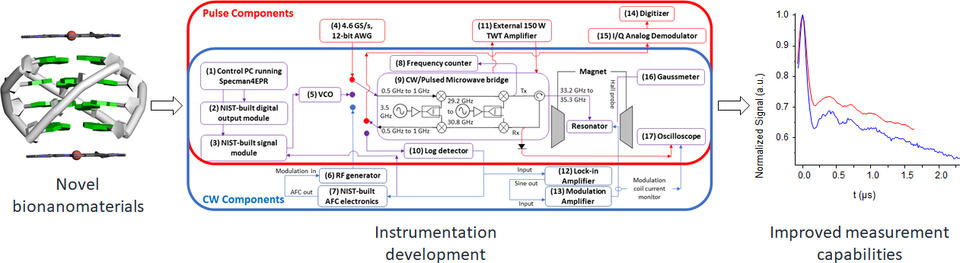
EPR spectroscopic methods to measure distances in the nanometer range, collectively referred to as pulsed dipolar spectroscopies, have been applied to biomacromolecular systems containing a pair of spin labels (nitroxide spin labels) to determine tertiary/quaternary structures and to follow conformational dynamics. The most common pulsed dipolar spectroscopy used in biomedical research is pulsed electron-electron double resonance (PELDOR). Because of its utility for biomacromolecular systems, PELDOR has become a measurement technique significant enough for focused development efforts from commercial vendors, in addition to its adoption by many academic research groups. It does, however, have challenges that must be addressed to broaden its application for problems important in biomedical research and biotechnology. Partnering with industry, we have built an electron paramagnetic resonance (EPR) spectrometer system that uses components unavailable on many other systems to improve resolution, signal-to-noise ratio, and decrease the sampling time. Advanced EPR spectroscopic measurements will enable hypothesis testing on the structure(s) of regions of biomacromolecules involved in eliciting therapeutic responses, for example. With our collaborators, we are involved in structural measurements on light-activated G-protein coupled receptors, ubiquitin-based cellular signaling pathways important in cancer, and iron-containing proteins involved in bacterial iron transport and eukaryotic protein degradation. Through such collaborations we aim to increase the adoption of structural measurements based on biological electron spin resonance and advance bioscience research.
Publications/Presentations:
The C-Terminus and Third Cytoplasmic Loop Cooperatively Activate Mouse Melanopsin Phototransduction by Juan C.Valdez-Lopez, Stephen T.Petr, Matthew P.Donohue, Robin J.Bailey, Meheret Gebreeziabher, Evan G.Cameron, Julia B.Wolf, Veronika A.Szalai, Phyllis R.Robinson, Biophysical Journal, Volume 119, Issue 2, 21 July 2020, Pages 389-401
Escherichia coli FeoC binds a redox-active, rapidly oxygen-sensitive [4Fe-4S] cluster, A.T. Smith, R.O. Linkous, N.J. Max, A.E. Sestok, V. A. Szalai, K.N. Chacón, Biochemistry 2019 58 4935-4949
Integration of a Versatile Bridge Concept in a 34 GHz Pulsed/CW EPR Spectrometer, A. Band, M. Donohue, B. Epel, S. Madhu, V. Szalai Journal of Magnetic Resonance, 2018 288 28-36
Instrumentation and Measurement Development for Electron Paramagnetic Resonance (EPR) Spectroscopy of Bionanomaterials, Szalai, V; Abhyankar, N., Donohue, M., Majikes, J., Band, A., Epel, B., Agrawal, A., and McMichael, R., University of Denver Department of Chemistry & Biochemistry, Denver, CO, October 31, 2019
EPR Spectroscopy Measurement Development for Nanobiomaterials, Szalai, V; Abhyankar, N., Donohue, M., Majikes, J., Band, A., Epel, B., Agrawal, A., and McMichael, R. Southeastern Magnetic Resonance Conference, Gainesville, FL, October 23, 2019
Instrumentation and Measurement Development for Electron Paramagnetic Resonance (EPR) Spectroscopy of Biomacromolecular Systems, Szalai, V; Abhyankar, N., Donohue, M., Majikes, J., Band, A., Epel, B., Agrawal, A., and McMichael, R., International Conference on Biological Inorganic Chemistry, Interlaken, Switzerland, August 16, 2019
Electron Paramagnetic Resonance (EPR) Spectroscopy for Bionanomaterial Measurements, Szalai, V., Abhyankar, N., Donohue, M., Majikes, J., Band, A., Epel, B., Agrawal, A., and McMichael, R., NIST, Applied Physics, Boulder, CO, July 26, 2019.

