Food Nutrition
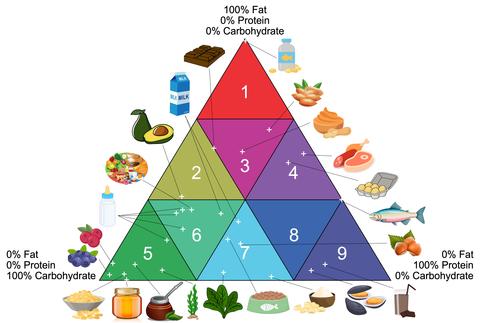
Well-characterized food-matrix reference materials are critical to facilitate compliance with nutritional labeling laws, provide traceability for food exports, improve the accuracy of label information for packaged foods, and contribute to studies of human nutritional status. Enactment of the Infant Formula Act of 1980 and the Nutrition Labeling and Education Act (NLEA) of 1990 resulted in requirements for labeling of processed and packaged foods in the US. Some nutrients (e.g., calories, fat, cholesterol) must be declared on the label, while information about other nutrients can be included at the manufacturer’s discretion. However, all nutrient declarations must be accurate and within thresholds set by the regulatory agency (i.e., U.S. Food and Drug Administration (FDA) or the U.S. Department of Agriculture (USDA)). In support of compliance with these regulations, the CSD has developed a series of food-matrix reference materials characterized for mass fractions of common nutrients. The selection of commodities for reference material development was strongly informed by the AOAC INTERNATIONAL triangle model for classification of foods into groups with similar composition [1]. The AOAC food triangle is based on the relative levels of fat, protein, and carbohydrate in the food, and built on the premise that foods with similar macronutrient profiles will offer similar analytical challenges for determination of micronutrients. Food-matrix reference materials are developed from animal, plant, and mixed-matrix sources to represent common food types with various macronutrient profiles [2, 3]. These reference materials are used by laboratories involved with the characterization of food composition (e.g., food manufacturers and processors, regulators, third-party testing laboratories) to develop and validate new analytical methods, to improve the quality of their analytical measurements, and to support compliance with a number of federal regulations enforced by the FDA and USDA [4].
Project Areas
Nutritional Elements
The FDA requires the content of sodium, calcium, iron, and potassium be provided on the nutritional label of any processed or packaged food sold in the US. In addition, other elements of nutritional interest, such as magnesium, phosphorus, or zinc, may be declared on nutritional labels to bolster or prove the food label or marketing claim. Prior to analytical measurement, the food sample is most often digested in concentrated acid using a microwave or open beaker technique. The NIST approach for elemental analysis includes the addition of internal standards and determination by inductively coupled plasma with optical emission spectrometry (ICP-OES) or with mass spectrometric detection (ICP-MS) [5]. In addition, NIST uses X-ray fluorescence spectrometry (XRF) and the use of multiple neutron activation analysis techniques, instrumental neutron activation analysis (INAA), radiochemical neutron activation analysis (RNAA), and prompt gamma neutron activation analysis (PGAA), for elemental measurements. To date, over 35 food-matrix reference materials have been issued with values assigned for nutritional elements. For additional technical information, contact Colleen Bryan Sallee (colleen.bryansallee [at] nist.gov (colleen[dot]bryansallee[at]nist[dot]gov)).
Water-Soluble Vitamins
Declaration of water-soluble vitamin content is not required on nutritional labels, but many are included as optional values to bolster or prove food label or marketing claims. Water-soluble vitamins may be present in foods in a variety of forms, with varying levels of individual vitamers depending on the type of food product. Prior to analytical measurement, vitamins are extracted from the food sample, which may include weak hydrolysis to release protein- or lipid-bound forms. The NIST approach for water-soluble vitamin measurement includes the addition of labeled vitamins as internal standards and determination by liquid chromatography with tandem mass spectrometric detection (LC-MS/MS) [6, 7, 8]. To date, over 15 food-matrix reference materials have been issued with values assigned for water-soluble vitamins. For additional technical information, contact Melissa Phillips (melissa.phillips [at] nist.gov (melissa[dot]phillips[at]nist[dot]gov)).
Fat-Soluble Vitamins and Carotenoids
The FDA requires the content of vitamin D be provided on the nutritional label of any processed or packaged food sold in the US. Declaration of other fat-soluble vitamin content is not required on nutritional labels, but many are included as optional values to bolster or prove food label or marketing claims. Fat-soluble vitamins may be present in foods in a variety of forms, with varying levels of individual vitamers depending on the type of food product. Prior to analytical measurement, vitamins are extracted from the food sample, which may include saponification to release lipid-bound forms and/or esters. The NIST approach for fat-soluble vitamin measurement includes the addition of labeled vitamins or related compounds as internal standards and determination by liquid chromatography with absorbance detection (LC-absorbance), with fluorescence detection (LC-fluorescence), or with tandem mass spectrometric detection (LC-MS/MS). To date, over 15 food-matrix reference materials have been issued with values assigned for fat-soluble vitamins. For additional technical information, contact Kate Rimmer (kate.rimmer [at] nist.gov (kate[dot]rimmer[at]nist[dot]gov)).
Fatty Acids
The FDA requires the content of total fat, calories due to fat, and total saturated and trans fats be provided on the nutritional label of any processed or packaged food sold in the US. In addition, other fatty acids of nutritional interest, such as docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA), often are declared on nutritional labels to bolster or prove the food label or marketing claim. Fatty acids are typically present in plant and animal tissues as triglycerides and/or phospholipids, with varying levels of individual fatty acids depending on the type of food product. Prior to analytical measurement, fat is extracted from the food sample, triglycerides and phospholipids are saponified (hydrolyzed) into their free fatty acid components, then all free fatty acids are converted to fatty acid methyl esters (FAMEs). The NIST approach for fatty acid measurement includes the addition of deuterated fatty acids as internal standards and determination by gas chromatography with flame ionization detection (GC-FID) or with mass spectrometric detection (GC‑MS) [9, 10]. To date, over 20 food-matrix reference materials have been issued with values assigned for fatty acids. For additional technical information, contact Ben Place (benjamin.place [at] nist.gov (benjamin[dot]place[at]nist[dot]gov)).
Cholesterol
The FDA requires the cholesterol content be provided on the nutritional label of any processed or packaged food sold in the US. Cholesterol is present in animal tissues as free cholesterol and also bound in triglycerides. Prior to analytical measurement, cholesterol is extracted from the food sample, cholesterol esters are saponified (hydrolyzed) into free cholesterol, then derivatized. The NIST approach for cholesterol measurement includes the addition of labeled cholesterol as an internal standard and determination by gas chromatography with mass spectrometric detection (GC‑MS) [11, 12]. Currently, 5 food-matrix reference materials have been issued with values assigned for cholesterol. For additional technical information, contact Melissa Phillips (melissa.phillips [at] nist.gov (melissa[dot]phillips[at]nist[dot]gov)).
Measurement Services
Reference Materials for Food Nutrition
SRM 1546a Meat Homogenate
SRM 1548b Typical Diet
SRM 1549a Whole Milk Powder
SRM 1845a Whole Egg Powder
SRM 1849b Infant/Adult Nutritional Formula I
SRM 1869 Infant/Adult Nutritional Formula II
SRM 2383a Baby Food Composite
SRM 2384 Baking Chocolate
SRM 2386 Avocado Powder
SRM 2387 Peanut Butter
SRM 3233 Fortified Breakfast Cereal
other Food-matrix RMs
SRM 1515 Apple Leaves
SRM 1566b Oyster Tissue
SRM 1567b Wheat Flour
SRM 1568c Rice Flour
SRM 1570a Trace Elements in Spinach Leaves
SRM 1573a Tomato Leaves
SRM 1577c Bovine Liver
SRM 1945 Organics in Whale Blubber
SRM 1947 Lake Michigan Fish Tissue
SRM 1974c Organics in Mussel Tissue (Mytilus edulis)
SRM 2385 Slurried Spinach
SRM 2974a Organics in Freeze-Dried Mussel Tissue (Mytilus edulis)
SRM 3232 Kelp Powder (Thallus laminariae)
SRM 3250 Saw Palmetto (Serenoa repens) Fruit
SRM 3251 Saw Palmetto (Serenoa repens) Extract
SRM 3253 Yerba Mate Leaves
SRM 3254 Green Tea (Camellia sinensis) Leaves
SRM 3255 Green Tea (Camellia sinensis) Extract
SRM 3275 Omega-3 and Omega-6 Fatty Acids in Fish Oil
SRM 3278 Tocopherols in Edible Oils
SRM 3281 Cranberry (Fruit)
SRM 3282 Low-Calorie Cranberry Juice Cocktail
SRM 3299 Ground Turmeric (Curcuma longa L.) Rhizome
SRM 3300 Curcumin Extract of Turmeric (Curcuma longa L.) Rhizome
SRM 3530 Iodized Table Salt (Iodide)
RM 8037 Krill Oil
RM 8183 Botanical Oils Containing Omega-3 and Omega-6 Fatty Acids
RM 8403 Cocoa Flavanol Extract
RM 8404 Almond Flour for Allergen Detection
RM 8405 Hazelnut Flour for Allergen Detection
RM 8256 Wild-caught Coho Salmon
RM 8257 Aquacultured Coho Salmon
RM 8258 Wild-caught Shrimp
RM 8259 Aquacultured Shrimp
QUALITY ASSURANCE PROGRAMS (QAPs)
NIST Quality Assurance Programs (QAPs), such as the Food Nutrition and Safety Laboratory QAP (FNSQAP) and the Dietary Supplements Laboratory QAP (DSQAP), provide opportunities for participants to identify and understand community-wide measurement challenges, improve the accuracy of their measurements, and demonstrate an effort to comply with various regulations. These programs offer the opportunity for laboratories to assess their in-house measurements, and participation in a quality assurance program in conjunction with the use of RMs has been shown to improve the comparability and precision of data over time. For additional information, contact the QAP Teams at QAPHUB [at] nist.gov (QAPHUB[at]nist[dot]gov).
Key Collaborators
NIST’s mission is to promote U.S. innovation and industrial competitiveness by advancing measurement science, standards, and technology in ways that enhance economic security and improve our quality of life. Stakeholder engagement is critical for development of impactful measurement services, and some long-standing partnerships are described below. To share your measurement services needs or suggest new or upgraded solutions, please contact Melissa Phillips (melissa.phillips [at] nist.gov).
AOAC INTERNATIONAL

AOAC INTERNATIONAL is a 501©(3), independent, third party, not-for-profit association and voluntary consensus standards developing organization that brings together government, industry, and academia to establish standard methods of analysis that ensure the safety and integrity of foods and other products that impact public health around the world. NIST staff are actively involved in AOAC standards development activities and engaged with AOAC stakeholders to inform development of measurement services to meet food nutrition and labeling needs.
Consumer Brands Association (formerly Grocery Manufacturers Association)

The Consumer Brands Association champions the industry providing the consumer packaged goods industry, including product categories such as household and personal care to food and beverage products. The Consumer Brands Association has a rich history of shaping the food industry and members include some of the largest and most well-known brands in the US. Beginning in the early 2000s, NIST and the Consumer Brands Association (formerly the National Food Products Association and Grocery Manufacturers Association) began a strategic partnership to improve measurements of member laboratories and contribute industry data to reference material value assignments. Many NIST Certificates of Analysis contain reference to this partnership.
National Institutes of Health Office of Dietary Supplements (NIH ODS)

The NIH ODS was created in 1995 to promote scientific research in the area of dietary supplements, and the Analytical Methods and Reference Materials (AMRM) program was established in 2002 to support and accelerate the development, validation, and dissemination of analytical methods and reference materials for dietary supplements and their ingredients. NIST has partnered with the AMRM program since its inception, primarily on natural products research and reference materials. Additionally, determination of vitamins and minerals in foods and supplements has been supported and enhanced through the longstanding collaboration with NIH ODS.
US Food and Drug Administration (FDA)

At the US FDA, the Center for Food Safety and Applied Nutrition (CFSAN) and the Office of Regulatory Affairs (ORA) are national leaders in protecting and promoting public health by ensuring proper labeling of foods for sale in the US. In initial meetings with the FDA in the 1990s, needs for reference materials to establish accuracy in food labeling helped shape the beginnings of the NIST food nutrition reference material program. For the last 30 years, NIST and FDA have maintained open communications that facilitates development and proper use of measurement services.
US Department of Agriculture (USDA)

The USDA provides leadership on food, agriculture, natural resources, rural development, nutrition, and related issues based on public policy, the best available science, and effective management. Various laboratories at USDA have directly collaborated with NIST on value assignment of nutrients in food reference materials, and continue to be involved in discussions about emerging needs for new measurement services.
Opportunities
- NIST supports the efforts of small and medium-sized manufacturers through the Manufacturing Extension Partnership and MEP-Assisted Technology and Technical Resource (MATTR) Program
- Food Safety Postdoctoral Opportunities at NIST are available in cooperation with the National Academies/National Research Council.
References
[1] Wolf, W.R., Andrews, K.W. Fresenius J Anal Chem 352, 73–76 (1995). https://doi.org/10.1007/BF00322300
[2] Phillips, M.M., Sharpless, K.E., Wise, S.A. Anal Bioanal Chem 405, 4325–4335 (2013). https://doi.org/10.1007/s00216-013-6890-5
[3] Wise, S.A., Phillips, M.M. Anal Bioanal Chem 411, 97–127 (2019). https://doi.org/10.1007/s00216-018-1473-0
[4] Sharpless, K.E., Duewer, D.L., Lippa, K.A., Rukhin, A.L. NIST Special Publication 260-181 Revision 1 (2015). https://doi.org/10.6028/NIST.SP.260-181r1
[5] Winchester, M.R., Butler, T.A., Turk, G.C. Analytical Chemistry 82 (18), 7675-7683 (2010). https://pubs.acs.org/doi/10.1021/ac101471a
[6] Phillips, M.M. Anal Bioanal Chem 407, 2965–2974 (2015). https://doi.org/10.1007/s00216-014-8354-y
[7] Camara, J.E., Lowenthal, M.S., Phinney, K.W. Anal Bioanal Chem 405, 4561–4568 (2013). https://doi.org/10.1007/s00216-013-6733-4
[8] Thomas, J.B., Yen, J.H., Sharpless, K.E. Anal Bioanal Chem 405, 4539–4548 (2013). https://doi.org/10.1007/s00216-013-6891-4
[9] Place, B.J. J AOAC INT 100, 814–819 (2017). https://doi.org/10.5740/jaoacint.16-0356
[10] Sander, L.C., Schantz, M.M., Sharpless, K.E., Wise, S.A. Lipid Technology 21, 7-12 (2009). https://doi.org/10.1002/lite.200800080
[11] AOAC Official Method 996.06; Official Methods of Analysis, 18th ed.; AOAC International, Gaithersburg, MD (2000). http://eoma.aoac.org
[12] Ellerbe, P., Meiselman, S., Sniegoski, L.T., Welch, M.J., White, V.E. Anal Chem 614, 1718–1723 (1989). https://doi.org/10.1021/ac00190a025
Contact
To share your measurement services needs or suggest new or upgraded solutions, please contact Melissa Phillips (melissa.phillips [at] nist.gov (melissa[dot]phillips[at]nist[dot]gov)).

