Summary
The Micronutrients Measurement Quality Assurance Program (MMQAP) was initiated in 1984 by the National Cancer Institute Division of Cancer Prevention and Control to ensure the long-term reliability of the measurements made while studying the possible cancer chemoprevention roles of these compounds. Coordinated by the Chemical Sciences Division, MMQAP supported measurement technology for selected fat- and water-soluble vitamins and carotenoids in human serum and provided participants with measurement comparability assessment through use of interlaboratory comparison studies, Standard Reference Materials and control materials, and methods development and validation. The MMQAP concluded in 2017, and parts of the MMQAP community will now be served through the ClinQAP.
Description
The MMQAP supported the long-term (months to years) reliability of selected fat- or water-soluble vitamin measurements in human serum and plasma. Results from the comparison studies helped participants to make accurate clinical and health-care decisions as well as to maintain and improve their measurement comparability.
Major Accomplishments
The MMQAP administered over 90 interlaboratory comparison exercises for the measurement of selected fat- and water-soluble vitamins in human serum and plasma. The comparability of laboratory measurements for target analytes improved substantially over time through the development and promulgation of robust measurement technologies, identification and production of suitable reference materials, isolation and identification of measurement system biases, and support and encouragement of long-term within-laboratory measurement quality control efforts. The average estimated coefficient of variation for retinol and α-tocopherol has been approximately 5% and about 10% for β-carotene since 2000.
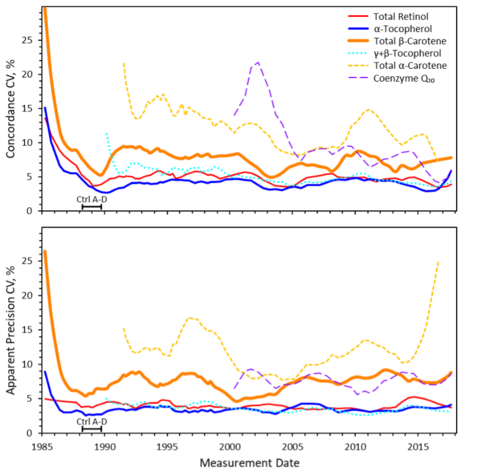
Each of the various curves represents the concordance or apparent precision metric transformed into a coefficient of variation (CV) expressed as percentage. The interval marked “Ctrl A-D” represents the period during which control samples A through D were provided to participants to enable validation of their measurement processes before they analyzed the unknown samples.
Study Reports
Reports for intercomparisons from 1988 onward are available as NIST Interagency/Internal Reports (IRs):
| Year | NIST IR | Year | NIST IR | |
| 1986 | 7880-38 | 2002 | 7880-21 and 7880-20 | |
| 1987 | 7880-37 | 2003 | 7880-19 and 7880-18 | |
| 1988 | 7880-36 | 2004 | 7880-17 and 7880-16 | |
| 1989 | 7880-35 | 2005 | 7880-15 and 7880-14 | |
| 1990 | 7880-34 | 2006 | 7880-13 and 7880-12 | |
| 1991 | 7880-33 | 2007 | 7880-11 and 7880-10 | |
| 1992 | 7880-32 | 2008 | 7880-9 and 7880-8 | |
| 1993 | 7880-31 | 2009 | 7880-7 and 7880-6 | |
| 1994 | 7880-30 | 2010 | 7880-5 and 7880-4 | |
| 1995 | 7880-29 | 2011 | 7880-3 and 7880-2 | |
| 1996 | 7880-28 | 2012 | 7880 and 7880-1 | |
| 1997 | 7880-27 and 7880-40 | 2013 | 7880-41 and 7880-42 | |
| 1998 | 7880-26 | 2014 | 7880-43 and 7880-44 | |
| 1999 | 7880-25 | 2015 | 7880-45 and 7880-46 | |
| 2000 | 7880-24 | 2016 | 7880-40 and 7880-47 | |
| 2001 | 7880-23 and 7880-22 | 2017 | 7880-48 |
Related Standard Reference Materials

SRM 968f Fat-Soluble Vitamins in Frozen Human Serum is intended for use in validating methods for determining fat-soluble vitamins in human serum and plasma and qualifying control materials produced in-house and analyzed using those methods. This SRM may be purchased from the Office of Reference Materials at NIST.
Additional Technical Details
Serum-based samples with assigned values for target analytes (e.g., retinol, tocopherols, and βcarotene) and performance-evaluation standards were distributed by NIST to participants for analysis. NIST staff provided participants with technical feedback concerning their measurement performance and suggestions for methods development and refinement. A certificate of participation in the QAP was issued at the end of each calendar year.
ASSOCIATED PUBLICATIONS
If you or your institution subscribe to a journal cited below, clicking on the link should take you to a page from which you can access the reference.
- Duewer, D.L., Brown Thomas, J.M., Kline, M.C., May, W.E., Sharpless, K.E. (2022), NIST Micronutrients Measurement Quality Assurance Program: Performance History of the Fat-Soluble Vitamin-Related Studies, NIST Interagency/Internal Report (NISTIR) 7880-50, National Institute of Standards and Technology, Gaithersburg, MD.
- Duewer, D.L., Brown Thomas, J.M., Young, S. (2022) NIST Micronutrients Measurement Quality Assurance Program Sample Integrity Confirmation, NIST Interagency/Internal Report (NISTIR) 7880-49, National Institute of Standards and Technology, Gaithersburg, MD.
- Duewer, D.L., Brown Thomas, J.M., Sharpless, K.E., Margolis, S.A. (2021), Lessons from the NIST Micronutrients Quality Assurance Program for Vitamin C, 1993 to 2015: Sample Stability, Assay Reproducibility, and Use of Controls to Improve Comparability, Anal. Bioanal. Chem., 413:289-298.
- Brown Thomas, J.M., Duewer, D.L., Burdette, C.Q., Sniegoski, L.T., Yen, J.H. (2017) Certification of Standard Reference Material® 968f Fat-Soluble Vitamins in Frozen Human Serum, NIST Special Publication 260-188, National Institute of Standards and Technology, Gaithersburg, MD.
- Thomas, J.B., Duewer, D.L., Mugenya, I.O., Phinney, K.W., Sander, L.C., Sharpless, K.E., Sniegoski, L.T., Tai, S.S., Welch, M.J., Yen. J.H. (2012) Preparation and Value Assignment of Standard Reference Material 968e Fat-Soluble Vitamins, Carotenoids, and Cholesterol in Human Serum, Anal. Bioanal. Chem., 402:749-762.
- Brown Thomas, J., Sharpless, K.E., eds. (1995) Methods for Analysis of Cancer Chemopreventive Agents in Human Serum, NIST Special Publication 874, National Institute of Standards and Technology, Gaithersburg, MD.
- Sharpless, K.E., Duewer, D.L. (1995) Population Distributions and Intralaboratory Variance for Fat-Soluble Vitamin-Related Compounds in Human Serum, Anal. Chem., 67:4416-4422.
- Brown Thomas, J., Kline, M.C., Schiller, S.B., Ellerbe, P.M., Sniegoski, L.T., Duewer, D.L., Sharpless, K.E. (1996) Certification of Fat-Soluble Vitamins, Carotenoids, and Cholesterol in Human Serum: Standard Reference Material 968b, Fresenius' J. Anal. Chem., 356:1-9.
- Margolis, S.A.; Duewer, D.L. (1996) Measurement of Ascorbic Acid in Human Plasma and Serum: Intralaboratory Repeatability, Interlaboratory Repeatability, Interlaboratory Reproducibility, and Stability in Lyophilized Plasma and Metaphosphoric Acid Preserved Plasma and Serum, Clin. Chem., 42:1257-1262.
- Duewer, D.L., Brown Thomas, J., Kline, M.C., MacCrehan, W.A., Schaffer, R., Sharpless, K.E., May, W.E. (1997) NIST/NCI Micronutrients Measurement Quality Assurance Program: Measurement Reproducibility, Repeatability, Stability, and Relative Accuracy for Fat-Soluble Vitamin-Related Compounds in Human Sera, Anal. Chem., 69:1406-1413.
- Margolis, S.A., Schapira, R.M. (1997) Liquid Chromatographic Measurement of L-Ascorbic Acid and D-Ascorbic Acid in Biological Samples, J. Chromatogr. B, 690:25-33.
- Sharpless, K.S., Brown Thomas, J., Turley, C.P., Brewster, M.A. (1997) "Vitamin A," In: Laboratory Medicine: A Scientific and Managerial Infobase, Version 2.6, A.J. Pesce and L.A. Kaplan, (eds.) Pesce Kaplan Publisher, Cincinnati, OH, CD-Rom.
- Sharpless, K.S., Brown Thomas, J., Turley, C.P., Brewster, M.A. (1997) "Vitamin E," In: Laboratory Medicine: A Scientific and Managerial Infobase, Version 2.6, A.J. Pesce and L.A. Kaplan, (eds.) Pesce Kaplan Publisher, Cincinnati, OH, CD-Rom.
- Brown Thomas, J., Kline, M.C., Duewer, D.L., Sharpless, K.E. (1998) The Stability of Retinol, α-Tocopherol, Trans-Lycopene, and Trans- β-Carotene in Liquid-frozen and Lyophilized Serum in Human Serum, Clin. Chim. Acta, 276:75-87.
- Duewer, D. L., Kline, M.C., Sharpless, K.E., Brown Thomas, J.M., Gary, K.T., Sowell, A.L. (1999) Micronutrients Measurement Quality Assurance Program: Helping Participants Use Inter-laboratory Comparison Exercise Results to Improve their Long-term Measurement Performance, Anal. Chem., 71:1870-1878.
- Duewer. D.L., Kline, M.C., Sharpless, K.E., Brown Thomas, J., Stacewicz-Sapuntzakis, M., Sowell, A.L. (2000) NIST Micronutrients Measurement Quality Assurance Program: Characterizing Individual Participant Measurement Performance Over Time, Anal. Chem., 72:3611-3619.
- Duewer. D.L., Kline, M.C., Sharpless, K.E., Brown Thomas, J. (2000) NIST Micronutrients Measurement Quality Assurance Program: Characterizing the Measurement Community's Performance Over Time, Anal. Chem., 72:4163-4170.
- Brown Thomas, J., Kline, M.C., Gill, L.M., Yen, J.H., Duewer, D.L., Sniegoski, L.T., Sharpless, K.E. (2001) Preparation and Value Assignment of Constituents in Standard Reference Material 968c: Fat-Soluble Vitamins, Carotenoids, and Cholesterol in Human Serum, Clin. Chim. Acta, 305:141-155.

