Summary
Liquid scintillators have been at the heart of radiation measurements at NIST for decades. NIST scientists have pushed the limits of liquid scintillation-based techniques for radionuclide metrology, neutron detection, and, most recently, neutrino detection. This diversity of applications demands a range of scintillators, variously formulated for optimal light yield, salt tolerance, or timing characteristics. To meet these demands, we investigate the surfactant chemistry, micellar dynamics, and fluorescence characteristics of complex scintillation “cocktails”.
Description
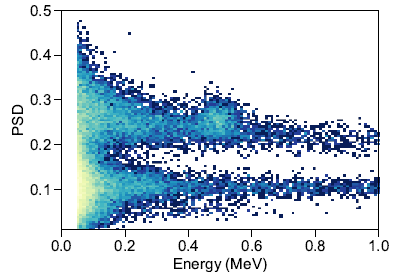
Pulse shape discrimination (PSD) in a Li-loaded scintillator allows tagging of neutron capture events.
It is now common to use surfactants to entrain aqueous metal salts in organic scintillators. This is crucial for radioactivity measurements because most radionuclides of interest are metals. More, non-metal radionuclides encountered in the environment or in medicine are nearly always in aqueous form. This is also crucial for building large neutron/neutrino detectors loaded with neutron-capturing Li or Gd. Adding aqueous material to an organic scintillator matrix is a delicate operation. Desirable optical properties are achieved with a microemulsion phase where the aqueous material is accommodated in nanometer-scale reverse micelles. Adding too much or too little aqueous material risks entering a potentially unstable and/or turbid phase with properties unsuitable for most applications.
We have developed approaches to identify scintillators with application-appropriate properties and continue to study how compositional changes can improve long-term stability and light yield while incorporating high aqueous fractions.
Our research on reverse micelles in scintillators has changed how NIST and other national metrology institutes prepare samples and calculate efficiencies for the CIEMAT-NIST efficiency tracing (CNET) method and the triple-to-double coincidence ratio (TDCR) method. (See, e.g., Applied Radiation and Isotopes 70, 2164 (2012), doi: 10.1016/j.apradiso.2012.02.089; Journal of Physical Chemistry A 118, 8563 (2014), doi: 10.1021/jp502540n)
Our ongoing research has been key to scintillator and detector design considerations for the Precision Reactor Oscillation and Spectrum Experiment (PROSPECT), which has investigated anomalies in neutrino spectra and oscillations at a reactor. (See, e.g., Journal of Instrumentation 14 P03026 (2019), doi: 10.1088/1748-0221/14/03/P03026)
Increasingly, it is apparent that different applications require optimization of different scintillator properties. For example, radionuclide metrology applications benefit from a reduced “delayed fluorescence” component, while applications like PROSPECT that rely on good pulse-shape discrimination rely on easily distinguishable fast and slow components. We continue to seek approaches to tailor scintillator properties to specific applications.
Notice of Online Archive: This project ended in 2023 and thus this page is no longer being updated and remains online for informational and historical purposes only. The information is accurate as of July 2024. For questions about page contents, please contact Denis Bergeron.

