Summary
Accurate measurement of microbial viability is crucial in infectious disease diagnosis and treatment; for development of microbial therapeutics, probiotics and antibiotics; in food and drinking water safety; and for ensuring product sterility during biomanufacturing. Viability determination traditionally relies on plate counting, which can take days to weeks to achieve results. Other techniques, such as colorimetric or fluorescent metabolic assays, DNA-based methods, and membrane integrity assays, all have limitations in sensitivity and specificity. We are pursuing method and technology development to improve confidence in detecting and quantifying viable microbes, including those that are not culturable. These methods will be applied to quantify microbial whole cell reference materials.
Description
Validating DNA-based methods to quantify viable bacteria (S. Da Silva, G. Pinheiro)
Microbial detection technologies based on nucleic acid measurements, such as polymerase chain reaction (PCR), are powerful detection tools that can provide relatively rapid results as compared to traditional culture- and phenotype-based approaches. However, their application is limited because typical protocols cannot differentiate live and dead bacteria. Modified PCR protocols have promise to detect only viable cells but have not been fully validated.
Our goal is to establish and evaluate sources of uncertainty for a measurement capability to quantify the absolute number of viable microbial cells using droplet digital PCR (ddPCR), as a collaborative project with INMETRO, the national metrology institute of Brazil. This approach uses cell membrane integrity to approximate viability, as viable cells typically have intact cell membranes. Prior to ddPCR, cells are treated to prevent amplification of DNA from leaky (approximating dead) cells, so only DNA from cells with intact membranes should be detected. To aid in method validation, culture conditions are being established to minimize variability in the number of genomes per cell. Results are being compared to orthogonal methods for total and viable cell count, such as Coulter counter, flow cytometry, and plate counting.
This method expands the applicability of ddPCR beyond quantifying total DNA in a sample to also quantifying only DNA from membrane-intact microbes and opens up a new suite of applications for DNA-based quantification methods.
Novel approaches to measure bacteria viability (J. Dunkers)
Microbes that are viable, or in other words living, should be capable of reproduction under favorable conditions. However, the vast majority of viable microbes cannot be grown under standard laboratory conditions and can be considered viable but not culturable (VBNC). Under favorable growth conditions, these microbes can resuscitate and initiate infection or contamination.
VBNC microbes, although not growing, do utilize stored energy to maintain membrane potential. The intensity of fluorescent membrane potential indicators has been used to measure the magnitude of membrane potential as well as differentiate viable from non-viable cells. Correlating fluorescence intensity with a biological or material property is complicated by confounding effects. For instance, fluorescence intensity scales with bacteria size, and many bacterial communities exist as doublets or larger aggregates. Fluorescence intensity measurements can become skewed from dye photobleaching and quenching. Moreover, background fluorescence and sample autofluorescence can make it difficult to separate out the desired signal.
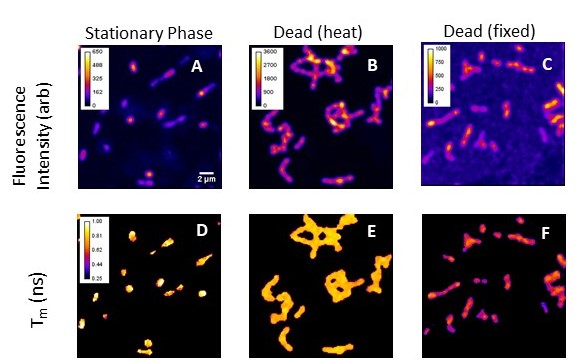
The use of fluorophore lifetime response can overcome many of the drawbacks of intensity measurements. Lifetime is independent of photobleaching and altered by local fluorophore environment, making it easier to differentiate sample fluorescence from background. Lastly, the units of fluorescence lifetime are nanoseconds, enabling direct comparison among experimental replicates and instruments.
The fluorescence lifetime microscopy (FLIM) images below show the intensity (A-C) and lifetime (D-E) responses of an anionic membrane voltage probe incubated with viable and non-viable Streptococcus mutans. FLIM in has the potential to serve as a foundational, quantitative measurement to which other viability assays can be benchmarked.