Summary
Damage to concrete structures in residential and commercial construction in central Connecticut have been attributed to the iron sulfide mineral pyrrhotite. Iron sulfides in concrete aggregate are not desirable as their relative instability results in decomposition with associated staining, expansion, and pop-outs near concrete surfaces or, in severe cases, cracking of the structure. There are no standardized test methods to assess pyrrhotite occurrence and abundance in aggregate or in concrete. Suggested limits on aggregate include 1 % S (Sulfide) by mass in Europe, a Canadian (and now Connecticut and Massachusetts) limit of 0.1 % S or 0.23 % pyrrhotite, are very challenging to meet using current standard guides for petrographic analyses. Developing a standard test method, including a set of calibration reference standards will provide a means for accurate, consistent analysis of pyrrhotite in concrete. The pyrrhotite/aggregate/concrete reactions and rates will be documented so that the most deleterious reactions can be efficiently reduced or eliminated. This reaction and rate data in combination with the evaluation of proposed mitigation strategies, on both materials and structural levels, will be used to manage the deleterious effect of the presence of the pyrrhotite mineral as a component of the aggregate in concrete.
Description
Objective
By 2026, develop a standard test method, a set of calibration reference materials to quantify pyrrhotite in concrete; document the pyrrhotite/aggregate/concrete reactions and rates so that the most deleterious reactions can be efficiently reduced or eliminated; and evaluate proposed mitigation strategies to manage the deleterious effect of the presence of the pyrrhotite mineral as a component of the aggregate in concrete.
Technical Idea
To meet the measurement challenges of assessing pyrrhotite occurrence in concrete and damage from cracking from the volume expansion from pyrrhotite and related reactions, new standardized test protocols and exploration of mitigation methods are necessary. NIST will develop research in each of the three categories:
- Pre-Concrete Materials (aggregate testing to reduce/eliminate pyrrhotite)
- Concrete Mix and Placement (water/cement ratio, additives, aggregate treatment, surface treatments on finished slab)
- Pre-Existing Foundations (structural assessments and treatments: surface coating, membrane application, permeable chemical solutions)
Simply, the new technical idea is to address mitigation strategies for crack damage caused by pyrrhotite at three phases of the concrete life cycle: before mixing, during placement, and existing structures. The approach for each phase is to first understand which reactions should be targeted by developing a model system and/or examining field specimens from which different mitigation strategies can be targeted for each phase in the concrete life cycle. A multiscale methodology from materials to structures that includes physics- based modeling of reactions and resulting expansion with a multi-disciplinary project team, including chemistry, geology, materials science, mechanical and civil engineering is critical for success.
Research Plan
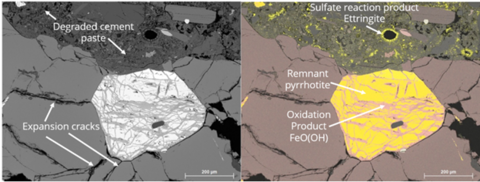
Pyrrhotite (Fe1-xS, where x = 0 – 0.2) is a common sulfide mineral exhibiting a brownish-bronze color with a metallic luster. Physical properties include a hardness of 4 on the Mohs scale, a specific gravity ranging between 4.58 and 4.65 and is magnetic, but with varying intensity depending upon the iron content. Pyrrhotite may occur as an iron-rich hexagonal high temperature form or an iron-poor monoclinic low temperature form. While not conclusive, studies have suggested that the monoclinic form oxidizes more rapidly than the hexagonal form. The oxidation of pyrrhotite in concrete aggregate results in the formation of ferrihydrite with an accompanying volume increase. Sulfate released from pyrrhotite oxidation initiates a secondary reaction with aluminosulfate phases in the hardened cement paste matrix resulting in a more substantial volume increase. Examples of pyrrhotite damage to concrete may be found in large dams in Europe, residential foundations in Quebec, Canada, Connecticut and now Massachusetts.
Fundamental reactions involving pyrrhotite and other iron sulfides in the aggregate and secondary reaction products with the surrounding cement matrix must be better understood to more effectively mitigate its deleterious effects. NIST will document the reactions and rates both experimentally and through physics-based modeling so that the most deleterious reactions can be efficiently reduced or eliminated.
NIST will develop plans for field studies to examine the extent of damage in the CT foundations. Fields studies are essential to characterize the extent of damage and minerology and better identify critical reactions on which to focus for mitigation. A field study for this project will benefit from both structural assessments and material/specimen collections. NIST will work with the UConn research group, who have established connections to the affected community, collected foundation specimens in the affected CT area, and completed some characterization. The NIST studies will include foundations with varying extents of deterioration, report structural assessments and materials collections. Field samples will also be used to test the use of coatings or other protection methods on the foundations. New test methods would be developed at NIST to examine foundation core specimens to determine the extent of reaction through the thickness of the core and efficiently quantify these changes at specific locations. Information from field samples will be used to determine a shortlist of mitigation strategies to investigation.
To facilitate development and evaluation of these test methods, a suite of reference samples for calibration and validation will be invaluable to ensure consistent, accurate testing. Reference standards for quantitative assessment of pyrrhotite in concrete in the Connecticut region should be representative of the pyrrhotite, the host rock, and the concrete matrix. Two distinctive lithologies are present in the Connecticut aggregate with a coarse and a fine-grained rock, which both may contain pyrrhotite. The coarse-grained lithology is thought to be more problematic because of its higher inter-granular porosity and permeability and the coarse nature of pyrrhotite. The fine-grained lithology also contains pyrrhotite, but it is thought to be less accessible because of the relative impermeability of this lithology, although pyrrhotite exposed on the surface will likely decompose.
To match the matrix of a typical concrete, in FY23 NIST completed a set of pyrrhotite reference materials, which includes pyrrhotite containing aggregate, sand, and hydrated cement. Because material characterization of the aggregate had given variable pyrrhotite concentration, synthesized pyrrhotite from iron and sulfur powder was added to the reference material in FY24. This will allow stakeholders to add known amounts of pyrrhotite to meet their applications. NIST is advancing tests methods in ASTM C09.50 Aggregate Reactions in Concrete (wavelength dispersive x-ray fluorescence (WD-XRF) and scanning electron microscopy/ energy dispersive spectroscopy (SEM/EDS)) to assess the proportions of sulfate and sulfide in samples.
All measurements are subject to random error and a lab-specific systematic error (bias). Four factors contribute to test variability: 1) operator, 2) equipment, 3) equipment calibration, and 4) testing environment. Within-laboratory repeatability excludes these four factors as they are generally constant for a single lab while between-laboratory reproducibility includes these four factors. A good test method has low variation on repeated tests of identical specimens for both single-operator and multi-laboratory precision. Fractional factorial experiment design facilitates development of robust test methods through identification and control of significant factors that are potential sources of variation in test determinations. This NIST set of reference calibration standards will minimize lab-specific systematic bias. NIST is proceeding with the WD-XRF test method initially. A refined test protocol with a documented within-lab precision statement will be drafted and promoted within the appropriate ASTM C09.50.
Mitigation strategies will be optimized by focusing on the two deleterious reactions found in the pyrrhotite containing crumbling foundations that result in volume expansion. The first expansion is from the reactivity of pyrrhotite leading to iron hydroxide formation. The second expansion is from the sulfate reaction, which is well documented. The presence of sulfate in solution reacts with aluminosulfate, a layered hydroxide, (AFm1) phases in concrete (roughly 10 % by volume) to create ettringite, which can cause up to a 150 % volume increase. Theoretical thermodynamic reaction modeling in combination with experimental reaction kinetic data will be used to document these reactions and the resulting volume expansion. Currently, NIST does not have in-house staff with thermodynamic modeling expertise and so we will utilize academic collaborations.
To understand the efficacy of any treatment, a “model/reference” synthesized aggregate will be developed to follow specific reaction mechanisms so that potential self-healing processes could be realized. These NIST model aggregates, or reference aggregates, with known masses of pyrrhotite and known PSD or surface area could be the basis of a standard test to evaluate the suitability of a treatment and/or to evaluate the upper limit concentrations of pyrrhotite. To synthesize the model aggregate, pyrrhotite must be incorporated consistently into a rock matrix that is both porous and permeable and to solidify the mass in a way that the pyrrhotite is not altered (oxidized). This synthesis will be a challenge. Approaches include sintering the matrix at temperatures that will not affect the pyrrhotite. The optimum matrix should be representative of a rock type found in the CT aggregates. To begin, clay and kaolinite will be assessed. Sintering clays may not be an option as the lower limit temps may exceed that for pyrrhotite. Kaolinite, which decomposes at 550 °C, may sinter around that temperature up to 750 °C. NIST experiments will test the synthesized aggregates incorporating known mass of specific particle size distribution (PSD) pyrrhotite as a standard test to assess 1) lower limit concentrations for crack initiation and 2) to assess propensity of proposed mitigation procedure effectiveness. The combination of pyrrhotite concentration and PSD will be assessed to identify their impact on rates of reaction and expansive stresses. Specifically, the NIST Empyrean XRD with the Mo source and large diameter capillaries will be used to examine reaction kinetics using the synthesized pyrrhotite that has been size graded and incorporated into a hardened cement paste. The reaction will use O2-saturated water flow and dissolution of the pyrrhotite, development of iron hydroxides (if crystalline), sulfate reactions (crystallization of ettringite) will be monitored. This would be an additional approach in investigating efficacy of a treatment by comparing to an untreated specimen.
Mitigation treatments will be explored for the three phases of the concrete life cycle (before mixing, during placement, and existing structures). Treatments could include reagents to alter the pyrrhotite reactions from within aggregate, through cement formulations used in concrete, or a coating barrier after placement or on the existing structure to prevent reactions within the concrete. An iron chelating agent may help to migrate away rather than precipitate these iron species in place. For new concrete, solutions to the volume increase include reformulation to change the size of the AFm inclusions and/or eliminate or reduce the amount of AFm. For current concretes these items have been fixed. Another possible mitigation method would be carbonation- AFm and the concrete- to reduce/eliminate this reaction regardless of any pyrrhotite reactions. Lithium salt treatments that have been reported to treat alkali-silica aggregate reactivity will also be assessed with caution as it is challenging to permeate concrete. Although the pyrrhotite-affected foundations contain lower-quality residential concrete where the permeability will be higher, transport mechanisms in concrete are slow and there is much unknown about what reactions are useful/helpful.
Given all mitigation treatment options, a coating barrier will be the initial focus as there are viable commercial products used for other applications that could apply to this pyrrhotite issue and there is an urgent need to find an alternative to the current practice of foundation replacement. To achieve a scientifically sound assessment of coating barriers, NIST will utilize newly prepared control and model/reference reactive aggregate concrete, respectively, to compare with field pyrrhotite containing foundation core specimens with no visible damage. Results using a well-defined model system compared with specimens from the field will be used to identify those coatings that seal the concrete and prevent/reduce moisture and oxygen penetration. Moreover, NIST will advance methods to test the permeability of coatings on concrete and cements to moisture and oxygen.
Major Accomplishments
A ceramic-based model aggregate system was synthesized using a clay body and additives to mimic the chemistry like aggregate. Synthesis parameters were modified to promote reactive polytypes of pyrrhotite. Experiments designs to scale up production to kg quantities for conventional expansion bars [(1 x 1x 12)] inch were completed. This model aggregate will be used to validate characterization methods for pyrrhotite and secondary products from pyrrhotite reactions with cementitious materials.
A finite element-based model of cracks due to expansion related to the presence of pyrrhotite was developed with the goal to predict the probability of failure due to amount of pyrrhotite in cement and published as a NIST IR. This model was applied to real-world cases from scanning electron microscopy (SEM) images of NIST field study CT foundations. Model crack propagation observations showed initiation at pyrrhotite sites, expansion to nearby pyrrhotite regions, propagation through aggregates into cement paste (CP), and continued growth within CP due to internal sulphate attack. The model also accounts for the diffusion of sulfates in cement matrix and determines change of modulus as a function of crack growth.
Studies assessing the effectiveness of concrete sealant to manage deleterious effects of the presence of the pyrrhotite mineral in the aggregate in concrete are showing that concrete sealant with most polymeric resin is more effective at mitigating water absorption. This study also illustrated that modifying standard ASTM air permeability equipment to accommodate 4-inch diameter specimens result in more accurate data. Assessment of additional deterioration to the field concrete for the aged, sealed specimens and measurement of aged control measurements to monitor transport of external conditions (e.g., moisture and oxygen) through concrete system are in progress.
Field core specimen characterization included new sample preparation methods using fluorescent dye and UV epoxy resin to quantify crack areas with optical microscopy. Backscatter electron scanning electron microscopy (BSE-SEM) data analysis methods were used to quickly estimate initial pyrrhotite fraction with fractions that oxidized than more time-consuming energy dispersion spectroscopy methods. In addition, these SEM data analysis methods were used to identify and measure isolated pyrrhotite inclusions to yield basic information on expansion during oxidation. All data was used as input for the expansion/crack simulation models.
Field studies were expanded to a foundation in Rutland, Massachusetts, where the NIST team collected > 30 core specimens (4 in. OD entire foundation thickness, ≤ 10 in.). The goal of field studies is to obtain specimens with varying extents of degradation and other structural engineering assessment information. NIST fields studies introduced new structural/crack inventory methods using a novel photogrammetric approach (S. Congress, MSU) and LIDAR techniques, separately. Field core characterization methods, including intake resonant frequency testing of field cores for dynamic Young’s modulus and traditional compressive strength tests were used in combination to the new structural assessment methods to map core location with mechanical properties using Random Field theory to address uncertainty probabilistically.
Several presentations of research from the project were given, including “Insights from Deteriorated Connecticut Foundations Containing Pyrrhotite-Bearing Aggregates to Improve Structural Durability”, RILEM 2025 Conference on Durability of Building Materials and Systems in Transportation Infrastructure, March 25-28, 2025, Mendrisio, Switzerland; “Field and Laboratory Analysis of Pyrrhotite-Affected Concrete Foundations”, Structures Congress 2025, April 9-11, Phoenix, AZ, USA; “Numerical Simulation and Analysis of Quasistatic Crack Propagation in Chemically-Attacked Cement-Based-Materials”, Engineering Mechanics Institute 2025 Conference (EMI 2025), May 27—30, 2025, Anaheim, CA, USA; an “Microstructures Causing Structural Instability: Applying Electron Backscatter Diffraction (EBSD) to Samples of Pyrrhotite Oxidation-Induced Concrete Degradation”, Microscopy and Microanalysis 2025, July 27-31, 2025, Salt Lake City, UT.

