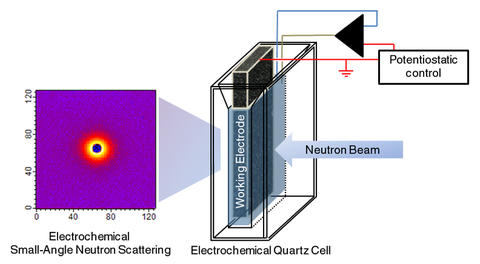
Schematic of NIST's "eSANS" (electrochemical Small-Angle Neutron Scattering) cell. A highly porous, sponge-like carbon electrode maximizes surface area for electrochemical reactions while structural details like particle size and configuration are measured using neutron scattering (image at left).
Researchers at the National Institute of Standards and Technology (NIST) have done a mash-up of two very different experimental techniques—neutron scattering and electrochemical measurements—to enable them to observe structural changes in nanoparticles as they undergo an important type of chemical reaction. Their recently published technique allows them to directly match up particle size, shape and agglomeration with the "redox" chemical properties of the particles. The measurements are important both for the design of nanoparticles for particular applications and for toxicology studies.
Nanoparticles present unique engineering challenges—and opportunities—because their extremely small size can give them physical properties quite unlike those they have in bulk quantities. The challenge for materials scientists is to determine just what those changes are and how they relate to particle size and structure.
The NIST team was interested in the oxidation-reduction—redox— properties of zinc oxide nanoparticles, which are used or being considered for a wide variety of applications ranging from sunscreens and antibacterial coatings to semiconductor and photoelectronic devices.
Redox reactions are one of the major divisions of chemical reactions, those that involve a transfer of electrons from one atom or molecule to another. Redox properties determine the path a chemical reaction will take. "They are the drivers of many biological processes," explains NIST materials researcher Vivek Prabhu. "There are many biochemical reactions that are well-defined oxidation-reduction reactions. There are tables of these. But there are no such tables that we know of on how nanoparticles can affect those reactions."
The NIST team knew they could monitor the size, shape and dispersion of nanoparticles in solution using SANS—small-angle neutron scattering. The scattering patterns from a SANS instrument, says Prabhu, give you not only those details but structural information about the solution itself, the size distribution of the particles and whether they clump together, all in "real" time as the experiment progresses.
Redox properties, on the other hand, are measured in electrochemical cells that are essentially half of a battery. Voltage and the amount of current flowing through the primary electrode depend on the reaction redox potential and the concentration of the test material.
The problem, Prabhu explains, is that SANS measures things in bulk, in a volume of space, but, "An electrochemical experiment is a very local experiment—it happens at an interface. What we needed was to maximize the interface." The answer, contributed by his partner, Vytas Reipa, is an exotic material called reticulated glassy carbon. "Like a very stiff household sponge or scouring pad made of pure carbon," Prabhu explains. The porous carbon electrode turned out to be an ideal terminal—lots of surface area to serve as a reaction interface; nearly transparent to neutrons, so it doesn't contribute much background noise; and best of all, it works well in water, enabling the study of nanoparticles in aqueous solutions, critical for biological reactions.
A big advantage of the "eSANS" technique, Prabhu says, is its generality. "You can apply our method to nearly any dispersed material that is of interest to redox chemistry—polymers, redox proteins, nucleic acids—at this nanoscale. Small polymer chains, for example. You can't really see them with electron microscopy, you can with neutrons."
V.M. Prabhu and V. Reipa. In situ electrochemical small-angle neutron scattering (eSANS) for quantitative structure and redox properties of nanoparticles. J. Phys. Chem. Lett. 2012, 3, 646-650 dx.doi.org/10.1021/jz300124t.

