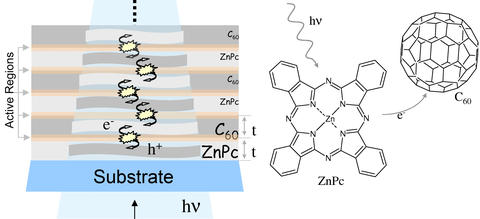
Light that strikes this organic solar cell causes electrons to flow between its layers, creating an electric current. Measurements made by the NIST/NRL research team determined the best thickness for the layers, a finding that could help optimize the cells performance.
Organic solar cells may be a step closer to market because of measurements taken at the National Institute of Standards and Technology (NIST) and the U.S. Naval Research Laboratory (NRL), where a team of scientists has developed a better fundamental understanding of how to optimize the cells' performance.*
Prototype solar cells made of organic materials currently lag far behind conventional silicon-based photovoltaic cells in terms of electricity output. But if even reasonably efficient organic cells can be developed, they would have distinct advantages of their own: They would cost far less to produce than conventional cells, could cover larger areas, and conceivably could be recycled far more easily.
The cells the team studied are made by stacking up hundreds of thin layers that alternate between two different organic materials—zinc phthalocyanine and C60, the soccer-ball shaped carbon molecules sometimes called buckminsterfullerenes, or "buckyballs." Light that strikes this multilayered film excites all its layers from top to bottom, causing them to give up electrons that flow between the buckyball and phthalocyanine layers, creating an electric current.
Each layer is only a few nanometers thick, and varying their thickness has a dramatic effect on how much electrical current the overall cell puts out. According to NIST chemist Ted Heilweil, determining the ideal thickness of the layers is crucial to making the best-performing cells.
"In essence, if the layers are too thin, they don't generate enough electrons for a substantial current to flow, but if they are too thick, many of the electrons get trapped in the individual layers," says Heilweil. "We wanted to find the sweet spot."
Finding that "sweet spot" involved exploring the relationship between layer thickness and two different aspects of the material. When light strikes the film, the layers generate an initial "spike" in current that then decays fairly quickly; the ideal cell would generate electrons as steadily as possible. Changing the layer thickness affects the initial decay rate, but it also affects the overall capacity of the material to carry electrons, so the team wanted to find the optimum combination of these two factors.
Paul Lane of NRL grew a number of films that had layers of different thickness, and the team made measurements at both labs that took the two factors into account, finding that layers of roughly two nanometers thick give the best performance. Heilweil says the results encourage him to think prototype cells based on this geometry can be optimized, though one engineering hurdle remains: finding the best way to get the electricity out.
"It's still unclear how to best incorporate such thin nanolayers in devices," he says. "We hope to challenge engineers who can help us with that part."
* P.A. Lane, P.D. Cunningham, J.S. Melinger, G.P. Kushto, O. Esenturk and E.J. Heilweil. Photoexcitation dynamics in films of C60 and Zn-Phthalocyanine with a layered nanostructure. Physical Review Letters, DOI: 10.1103/PhysRevLett.108.077402. Published 15 Feb 2012.

