SAT-TMMC: Liquid-vapor coexistence properties - Linear-Force Shifted Potential at 3.0σ
Liquid-vapor coexistence properties obtained by grand-canonical transition-matrix Monte Carlo and histogram re-weighting over the reduced temperature range 0.70 to 1.05 at increments of 0.05. Mean values of the saturation pressure, density, potential energy per molecule, and activity (chemical potential- see below) for each phase are reported.
| METHOD | Grand-canonical transition-matrix Monte Carlo and histogram re-weighting [1, 8-12] |
| V/σ3 | 512 |
| TRUNCATION | Linear Force Shifted at 3.0σ |
| Prob. of Disp. Move | 0.4 |
| Prob. of Ins/Del Move | 0.6 |
| Biasing Function Update Frequency | 1.0E6 trial moves |
| Simulation Length | 4.0E10 trial moves |
T* |
ρvap* |
+/- |
ρliq* |
+/- |
psat* |
+/- |
Uvap* |
+/- |
Uliq* |
+/- |
lnzsat* |
+/- |
| 0.70 | 7.501E-03 | 8.399E-07 | 7.852E-01 | 8.085E-05 | 4.931E-03 | 5.616E-07 |
-8.026E-02 |
1.200E-05 | -4.880E+00 | 1.079E-03 | -5.014E+00 | 3.579E-05 |
| 0.75 | 1.260E-02 | 9.754E-07 | 7.582E-01 | 5.995E-05 | 8.603E-03 | 9.141E-07 | -1.272E-01 | 2.613E-05 | -4.675E+00 | 5.116E-04 | -4.553E+00 | 1.019E-04 |
| 0.80 | 2.000E-02 | 3.176E-06 | 7.294E-01 | 4.892E-05 | 1.398E-02 | 1.457E-06 | -1.918E-01 | 6.606E-05 | -4.462E+00 | 6.670E-04 | -4.164E+00 | 7.423E-05 |
| 0.85 | 3.043E-02 | 2.874E-06 | 6.981E-01 | 6.105E-05 | 2.146E-02 | 2.037E-06 | -2.789E-01 | 3.193E-05 | -4.239E+00 | 6.362E-04 | -3.833E+00 | 3.196E-05 |
| 0.90 | 4.502E-02 | 1.127E-05 | 6.632E-01 | 4.728E-05 | 3.147E-02 | 3.614E-06 | -3.959E-01 | 1.371E-04 | -4.001E+00 | 4.514E-04 | -3.549E+00 | 7.106E-05 |
| 0.95 | 6.578E-02 | 9.238E-06 | 6.229E-01 | 8.581E-05 | 4.447E-02 | 4.035E-06 | -5.562E-01 | 8.421E-05 | -3.738E+00 | 6.128E-04 | -3.304E+00 | 4.753E-05 |
| 1.00 | 9.728E-02 | 3.577E-05 | 5.727E-01 | 9.086E-05 | 6.099E-02 | 8.989E-06 | -7.930E-01 | 3.541E-04 | -3.432E+00 | 5.604E-04 | -3.091E+00 | 6.316E-05 |
| 1.05 | 1.567E-01 | 1.848E-04 | 4.959E-01 | 1.935E-04 | 8.184E-02 | 9.093E-06 | -1.228E+00 | 1.810E-03 | -3.015E+00 | 8.225E-04 | -2.905E+00 | 3.964E-05 |
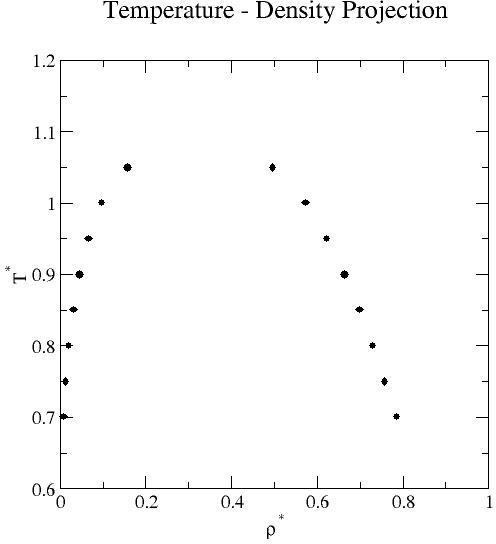
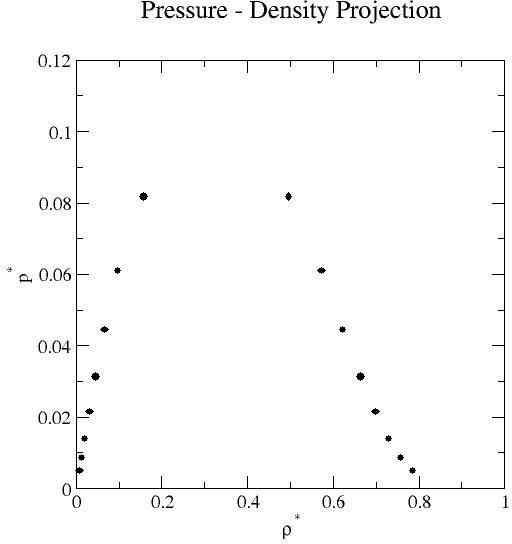
Remarks:
Uncertainties were obtained from five independent simulations and represent 95% confidence limits based on a standard t statistic. Liquid-vapor coexistence was determined by adjusting the activity such that the pressures of the liquid and vapor phases were equal. Here, the pressure is not the conventional virial pressure [2,3] but is the actual thermodynamic pressure, based on the fact that the absolute free energies can be obtained from the distributions determined from simulation [4]. Alternative methods, for example Gibbs-ensemble Monte Carlo and combination grand-canonical Monte Carlo and histogram re-weighting, can be used to determine liquid-vapor coexistence. A review of standard methods of phase equilibria simulations can be found in Ref. 5.
As introduced in Refs. 2 and 3, the activity, z, is defined as
$$ z = \dfrac{ \exp\left( \beta \mu \right)}{\lambda^3} $$
where Λ is the de Broglie wavelength, β = 1/(kBT) (where kB is Boltzmann's constant), and μ is the chemical potential. It is sometimes more convenient to work with ln z in the simulations and in post-processing. (NOTE: The reported activity is dimensionless, having been scaled by the LJ length cubed.)
Phase-coexistence energies were obtained by determining the mean potential energy at a given value of N for an additional 40 billion MC trials. Combining this information with the particle number probability distribution, the mean potential energy of the coexisting phases can be calculated [6].
For the Lennard-Jones fluid, linear force shifted at 3.0σ, the critical properties were estimated to be Tc*=1.079, ρc*=0.321, and pc*=0.096. Estimates were found via rectilinear diameter analysis of TMMC data computed with V*=512 close to the critical point [7]. (Finite-size scaling analysis has not been completed, so these critical properties should be taken simply as estimates.)
References
- J. R. Errington, J. Chem. Phys. 118, 9915 (2003).
- M. P. Allen and D. J. Tildesley, Computer Simulation of Liquids (Oxford University Press, New York, 1989).
- D. Frenkel and B. Smit, Understanding Molecular Simulation, 2nd ed. (Academic, San Diego, 2002)., pp.37-38.
- J. R. Errington and A. Z. Panagiotopoulos, J. Chem. Phys., 109, 1093 (1998).
- A. Z. Panagiotopoulos, J. Phys.: Condens. Matter, 12, R25-R52, (2000).
- J. R. Errington and V. K. Shen, J. Chem. Phys., 123, 164103 (2005).
- B. Smit and C. P. Williams, J. Phys.: Condens. Matter, 2, 4281-4288 (1990).
- V. K. Shen and D. W. Siderius, J. Chem. Phys., 140, 244106, 2014.
- V. K. Shen and J. R. Errington, JPC B 108, 19595, 2004.
- V. K. Shen and J. R. Errington, JCP 122, 064508, 2005.
- V. K. Shen, R. D. Mountain, and J. R. Errington, JPC B 111, 6198, 2007.
- D. W. Siderius and V. K. Shen, JPC C 117, 5681, 2013.

