Taking Measure
Just a Standard Blog
Optical frequency combs allow scientists to measure light—and our world—with great precision and accuracy. This device has led to innovations that scientists never imagined when it was created.
To many people, a measurement sounds mundane, like marking ticks on a ruler or reading the line on a thermometer. It’s a piece of data. And they tend to think that improved measurements look like finer and finer ticks on a ruler — which doesn’t seem very exciting.
But making new measurements is more than just making finer marks on a ruler. To measure something is to understand it, pull it apart and see how it works. New measurements can unlock possibilities that even scientists never thought of when they started out.
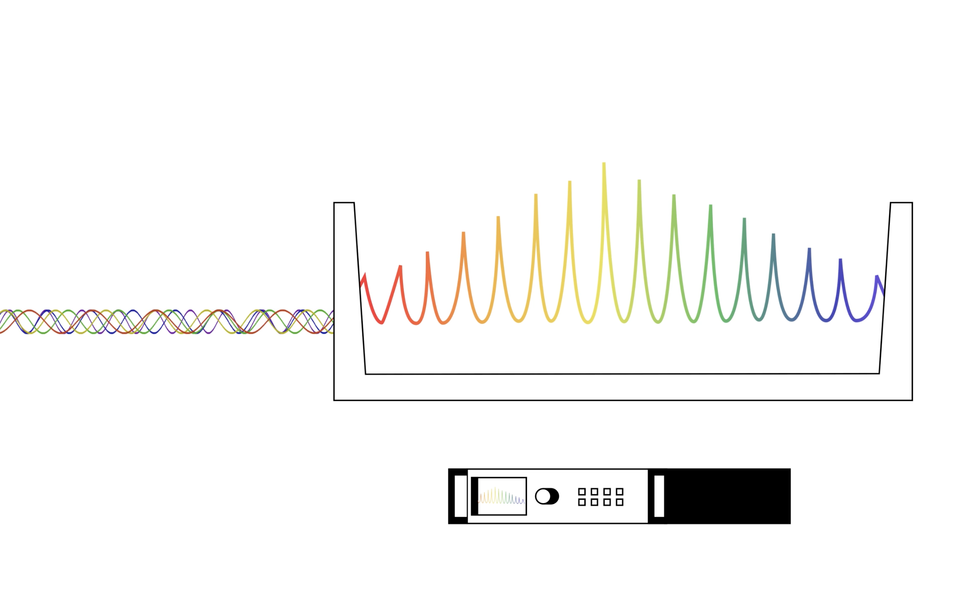
Perhaps there is no better example than the optical frequency comb. Very simply, this device is a ruler for light. Yet it’s so much more than a ruler.
Radio waves, microwaves, visible light, X-rays and infrared are all part of a spectrum of electromagnetic frequencies. They’re all waves, traveling at the speed of light, but the distance between the peaks of those waves can be kilometers apart, like some radio waves, or nanometers apart, like visible light and ultraviolet.
In the 1970s, scientists at the National Institute of Standards and Technology (NIST) were stuck. They wanted more precise and accurate atomic clocks, ones based on the very high optical frequencies of light released by atoms as their electrons jump between energy states, as opposed to the lower microwave frequencies they were using. Better clocks would give them a more precise definition of the second. A more precise second would give them a better definition of the meter, which is the distance light travels in a vacuum in a tiny fraction of a second. But all that relied on being able to measure these frequencies of light accurately and precisely.
There was a gap in measurement between these two ends of the electromagnetic spectrum. Scientists could measure radio and microwave frequencies accurately, but there were no electronics that could count fast enough to keep up with the atom’s optical frequencies. They could use a laser with a matching frequency to read the atom’s optical frequency. Scientists had lasers with known, exact frequencies, but they could only produce a single frequency or color. Without knowing the exact frequency of the atom, finding the right frequency laser to read the atom would take a lot of trial and error. NIST scientists tried daisy-chaining several lasers of different frequencies together to make a rudimentary optical ruler. That worked well enough to redefine the meter but wasn’t a long-term solution.
Enter the frequency comb, a Nobel Prize-winning device and the result of decades of research from NIST and others that you can read about here. The comb generates a billion pulses of light per second, which bounce back and forth inside an optical cavity. This creates millions of spikes of optical frequencies that look like rainbow-colored teeth on a comb (hence the name). The first tooth in that comb is set to a known frequency, which gives scientists a starting point to read the other frequencies. Much like a ruler, if you know the first marker is one millimeter and each marker is a millimeter apart, you can easily start measuring. Similarly, because they know exactly how far apart these frequencies are, scientists can translate these optical signals to microwaves with a simple mathematical formula, joining the two ends of the electromagnetic spectrum. This opens a lot of research doors.
Scientists used this new technology to make better clocks, eventually developing clocks that are 100 times better than the cesium clocks used for civilian time standards. More accurate and precise clocks are critical for GPS navigation, which relies on precise time signals to determine your location. Better clocks also have research advantages, from detecting tiny changes in gravity to studying phenomena of the quantum world and perhaps finding dark matter. These clocks may eventually change how we define a second. But scientists couldn’t have predicted all the other ways the comb would be used.
All atoms and molecules emit unique frequencies of light when they jump between energy states, not just the atoms used in clocks. If one of the frequencies from the comb hits an atom or molecule with the exact same frequency, scientists can identify what kind of atom or molecule they’ve hit. Using the optical frequency comb, scientists could study the composition of stars in exquisite detail. Astrophysicists can tell if they’ve found a new planet by measuring the changes in frequencies of the starlight as well. Using frequency combs, we can improve light-ranging systems, which bounce light off objects to detect them like radar or sonar. They can see objects through flames, helping NIST scientists study how structures fail during a fire.
The comb is also being used to detect even the smallest amounts of greenhouse gases in the air or look for disease in human breath.
All of that because we found a better way to measure light. Isn’t it amazing what a measurement can do?





