Summary
Protein drugs are the fastest-growing class of drugs in the pharmaceutical industry. However, they can be plagued by long-term stability issues, including aggregation and phase separation, which have yet to be fully understood. We have demonstrated an approach to characterize protein unfolding and aggregation and provide insight into destabilization mechanisms.
Description
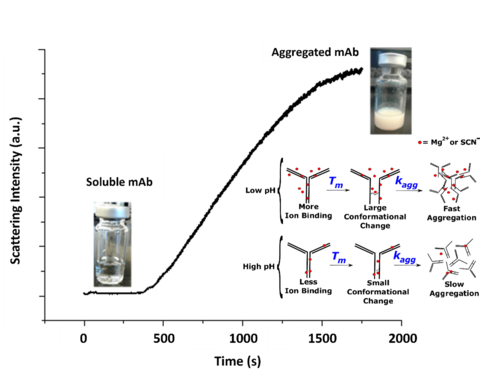
A stable protein formulation is essential to the safety of mAb drugs. In particular, mAb drug solutions formulated at high concentrations can undergo various biophysical instabilities, such as aggregation and liquid-liquid phase separation. These undesired solution properties are primarily driven by attractive protein-protein interactions. In fact, preventing such interactions during long-term storage is a major challenge faced by the biotech industry. Consequently, formulation conditions are critical to ensuring the robust and long-term shelf life of a protein drug solution.
A fundamental understanding of the impact of temperature, pH, viscosity, and salt addition on the biophysical stability of mAbs could help predict long-term stability. Our group systematically studies the effects of salt (e.g., the Hofmeister series), temperature and pH on the unfolding and aggregation kinetics of a low pI mAb. This approach allows for a detailed, mechanistic investigation of salt effects across various charged states of the mAb while maintaining relevant formulations and physiological conditions.
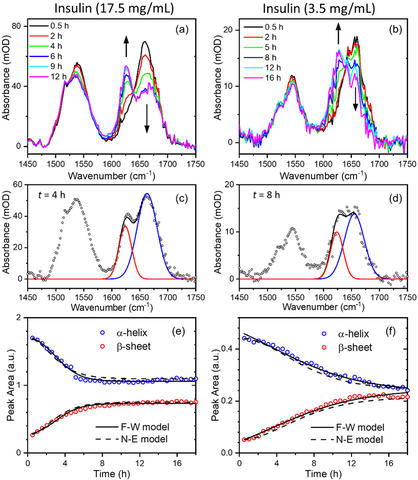
We have studied the biophysical stability of a therapeutic protein sample using various optical methods [1]. Dependences of the thermal stability on pH, salt, and temperature provide insights into detailed mechanisms of domain unfolding and solvent interactions. This mechanistic study can help improve the shelf-life of protein drug products and understand protein aggregation-related disease mechanisms. We used light scattering [1], thioflavin T[1], and recently developed solvent absorption compensation (SAC) IR spectroscopy [2], to investigate the aggregation kinetics in situ.
PUBLICATIONS
[1] K. B. Rembert, J. Zhang, Y. J. Lee, Effects of Salts and Surface Charge on the Biophysical Stability of a low pI Monoclonal Antibody, J. Pharmaceut. Sci. Published online (2022). https://doi.org/10.1016/j.xphs.2022.11.010
[2] B. Chon, S. Xu, Y. J. Lee, Compensation of Strong Water Absorption in Infrared Spectroscopy Reveals the Secondary Structure of Proteins in Dilute Solutions. Anal. Chem. 93, 2215 (2021). https://dx.doi.org/10.1021/acs.analchem.0c04091

