Understanding the Bioaccumulation of Gadolinium-Based Contrast Agents using Advanced Measurement Capabilities
Summary
Gadolinium-Based Contrast Agents (GBCA) are intravenous drugs used in diagnostic imaging procedures to enhance the quality of magnetic resonance imaging (MRI). The FDA currently considers GBCAs safe, however the development of nephrogenic systemic fibrosis (NSF) has been observed in patients with impaired renal function who are administered GBCAs. Recent research has emerged regarding the accumulation of gadolinium in tissues (bone and brain) of patients with normal renal function, with the retention of gadolinium increasing in patients who have repeated GBCA exposure. Despite these observations, very limited data regarding the form (e.g. species) of gadolinium accumulated and the mechanisms of accumulation and/or toxicity are known.
Description
MRI image of a human skull using a gadolinium-based contrast agent.
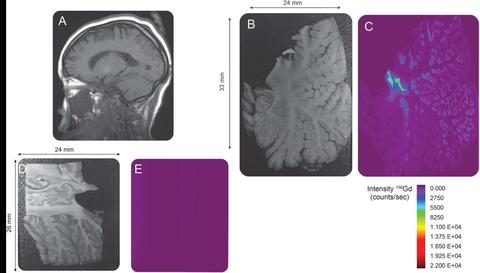
Researchers and staff from the Medical University of South Carolina are collaborating with NIST staff experienced in trace elemental speciation, sample preparation and instrumental analysis in order to demonstrate the bioaccumulation of intact GBCA in patients who have repeated GBCA exposure. Patients with as few as four GBCA exposures are shown to have detectable levels of gadolinium. The use of laser ablation coupled to inductively-coupled mass spectrometry permits detection of this bio-accumulation in specific regions of the brain. Additional research to determine the mechanism for the gadolinium accumulation is ongoing.
Associated Publication
1. Bandino, J. P., Gathings, R. M., Hinen, H. B., Hampton, M. T., Davis, W. C., and Elston, D. M., "Gadolinium PresenceWithin Cutaneous Sclerotic Bodies Confirmed by Laser Ablation Inductively Coupled Plasma Mass Spectrometry," Jama Dermatology, 154, 105-106 (2018).
2. Roberts, D. R., Welsh, C. A., and Davis, W. C., "Gadolinium Deposition in the Pediatric Brain," Jama Pediatrics, 171, 1229 (2017).
3. Roberts, D. R., Welsh, C. A., Lebel, D. P., and Davis, W. C., "Distribution Map of Gadolinium Deposition Within the Cerebellum Following Gbca Administration," Neurology, 88, 1206-1208 (2017).
4. Roberts, D. R., Lindhorst, S. M., Welsh, C. T., Maravilla, K. R., Herring, M. N., Braun, K. A., Thiers, B. H., and Davis, W. C., "High Levels of Gadolinium Deposition in the Skin of a Patient With Normal Renal Function," Investigative Radiology, 51, 280-289 (2016).
5. Cleveland, D., Long, S. E., Sander, L. C., Davis, W. C., Murphy, K. E., Case, R. J., Rimmer, C. A., Francini, L., and Patri, A. K., "Chromatographic methods for the quantification of free and chelated gadolinium species in MRI contrast agent formulations," Analytical and Bioanalytical Chemistry, 398, 2987-2995 (2010).

