In Situ Measurements of Thermodynamics and Reaction Kinetics During Synthesis and Functioning of Nanomaterials Using Transmission Electron Microscopy Based Techniques
Summary
Understanding and measuring synthesis-structure-property relationship at nanoscale is a fundamental step for designing nanoscale structures such as nanotubes, nanowires, plasmonic nanoparticles, quantum dots, etc. We employ our unique multiscale spectroscopy- environmental scanning transmission electron microscope (ESTEM), equipped with a monochromated electron source, an image corrector and a high-speed camera (frame rate of 1600 s-1) for quantifying dynamic processes at the nanoscale. We also continuously develop methods for in situ measurements of both structure and chemistry at the nanoscale and microscale, concurrently, during gas-solid interactions. Characterization of complex, time-dependent transformations require the use of advanced methodologies that enable the underlying chemical and physical processes to be identified and understood. Multiscale spectroscopy combined with electron diffraction as well as TEM/STEM imaging are employed for in situ observations and quantifications of dynamic processes, affecting both material structure and chemistry at the nano- and micro- scale during gas-solid interactions.
Description
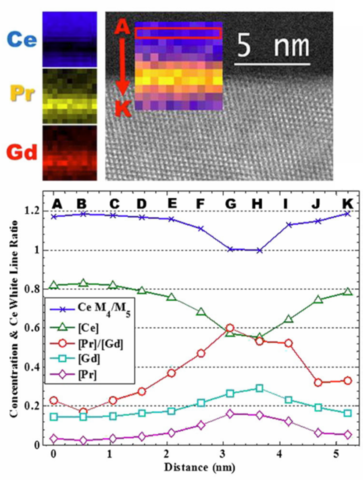
Annular dark-field image of a grain boundary with EELS maps showing the cation distribution at the interfacial region. Profiles of cation fractions and Ce M4/M5 white line ratio, suggesting the relationship between the cation fractions and Ce oxidation state.
Functional materials cover a broad range of materials, such as nanoparticles, atomically thin two-dimensional (2D) nanosheets, and thin films, for a variety of applications including catalysis and advanced electronics. Recent advances in nanotechnology enable a stringent control on synthesis of materials at the atomic level so that exotic property and functionality can emerge from specific structures. There is a growing need to identify and therefore develop these structures by allowing the structural components to interact in a controlled manner with external stimuli, including reactants, heat, pressure, electric field, stress, etc. At NIST, we advance in situ and operando methods for the ESTEM and implement a custom-built analytical spectroscopy platform to measure these complex transformations so that relevant dynamic processes can be understood electronically and optically.
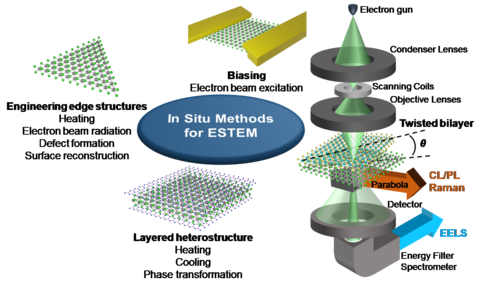
2D van der Waals heterostructures and device
2D materials have emerged as potentials candidates for various application, such as nanoelectronics, quantum, photovoltaic, energy storage and catalysis due to their unique electronic, optical and magnetic properties. These properties are not only dependent on the number of layers and their stacking order (e.g. twisted layers) but also on the chemistry and heterogeneity of component layers such as dopants, defects, edge structure etc. The edge structures and chemistry in 2D materials, play a critical role in controlling their construction, that enhance the electronic, magnetic, nonlinear optical, photoluminescence, and catalytic properties. It is conceivable that TEM should play an important role in deciphering the structure-function and property relationship. Moreover, in situ methods under different stimuli transform the measurements from “static” to “dynamic” and benefit the research focused on engineering novel nanoscale structures and understanding the functionality at the device level. A combination of multimodal techniques such as electron diffraction, imaging, and spectroscopies including electron energy-loss spectroscopy (EELS), Raman spectroscopy, energy dispersive X-ray spectroscopy (EDS), photoluminescence (PL) and cathodoluminescense (CL) are applied to obtain a complete picture of mechanisms involved by simultaneous measurement of structure, chemistry and properties.
Catalysts for Solid Oxide Fuel Cells
In this example, we measured the effect of working condition (temperature and pressure) on ceria-based anode materials for solid oxide fuel cell application in collaboration with the team at Arizona State University (ASU). We obtained time and temperature resolved high-resolution TEM (HRTEM) images and spectroscopy data to characterize morphological, structural and chemical changes. We used electron energy loss spectroscopy (EELS) to evaluate the reducibility of ceria as function of doping with aliovlent rare earth elements such as Gd and Pr. At the same time, we monitored the structure of carbon build-up on Ni catalysts using Raman spectroscopy when hydrocarbon gas was injected as a fuel source. The ESTEM capabilities allow us to probe the effect of heterogeneity, among the anode (interparticle) and within individual particles (intraparticle), on the catalytic properties of doped ceria.
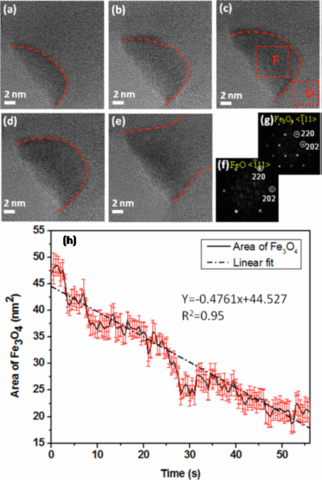
Redox Reactions in Transitional Metal Oxides
We also characterize the interplay of dynamical structural changes, phase evolution and metal oxidation state using ESTEM to provide insight into ways to optimize structure and chemistry for specific functions: (1) structure and dynamics of surface and subsurface transitions: TEM edge-on imaging are employed to determine the structural evolution of the surface and subsurface as a function of temperature and pressure of gases (e.g., H2, O2, methanol); (2) redox kinetics: both cross-sectional and planar TEM imaging are used to measure the oxidation and reduction kinetics including oxide nucleation and growth and oxide decomposition; (3) thermodynamics and phase evolution: in situ HRTEM imaging and electron diffraction are employed to measure the phase evolution upon oxidation and reduction under controlled temperature and gas pressure; (4) chemistry measurements: EELS data is used to determine the evolution of the oxidation states of metals during redox reactions. These atomic-scale in situ experimental data are fed into the density-functional theory (DFT) modeling for identifying the critical kinetic and thermodynamic factors controlling the interfacial processes of the redox reactions under technologically relevant reactive environments.
Plasmonic Nanoparticles for Localized Surface Plasmon Induced Reactions
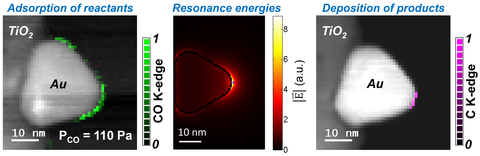
Heterogeneous catalysts are used to reduce activation energy (energy barrier) of chemical reactions by promoting gas adsorption on catalyst surfaces that is finally overcome using thermal energy. On the hand, localized surface plasmon (LSP) resonances, excited by photons on plasmonic nanoparticles, have been shown to induce gas dissociations at reduced temperatures, mimicking photocatalysis. Energy harnessed by plasmonic nanostructures and transferred to adsorbed reactants during the dephasing of LSP resonances is theorized to initiate such reactions by compensating for the thermal energy required otherwise. We have shown ESTEM is a powerful approach to capture the sub-nanoparticle level details of LSP induced reactions. In situ EELS allows us to identify the spatial distributions of LSP resonance energies and antinodes, and to measure the preferential gas adsorption sites on nanoparticle surfaces. We demonstrate that such sub-nanoparticle information is essential to identify catalytic active sites for LSP induced chemical reactions. We plan to continue to explore various hybrid nanoparticle systems that combine catalyst nanoparticles with plasmonic nanoparticles to initiate industrially relevant reactions at room temperature, thereby reducing the production costs of various chemicals, such as liquid fuels, ammonia, etc.
Major Accomplishments
2019
- Developed techniques to measure endothermic reactions at room temperature enabled by deep-ultraviolet localized surface plasmons.
2018
- Developed techniques to detect and employ localized surface plasmon resonances, excited by high energy electrons on plasmonic nanoparticles, to initiate room temperature reactions.
Additional Technical Details

Techniques available in the Multiscale Spectroscopy - ESTEM Laboratory:
High resolution transmission electron microscope (HRTEM): Imaging (0.06 nm spatial resolution) at the frame rates up to 1600 s-1 using direct electron detection camera.
Scanning transmission electron microscope (STEM): Angular dark-field(ADF) imaging (≈ 90 mrad) and at 0.135 nm spatial resolution
Electron energy-loss spectroscopy (EELS): Post-column dual EELS system with 0.08 eV energy resolution at 80 kV and hyperspectral imaging capability.
Energy dispersive x-ray spectrometer (EDS): Single Si(Li) detector with ≈ 150 eV energy resolution and hyperspectral imaging capability.
Raman Spectroscopy: Concurrent collection of Raman spectra using a 532-nm laser from the TEM samples is available for in situ microscale vibrational spectroscopy measurements at temperature, with or without gas environment concurrently with TEM observations. The laser can be used for local heating and Raman spectroscopy can be used to measure the temperature from 10-micron area of the sample.
Cathodoluminescence Spectroscopy: Optical response of the electron interactions with a wide range of materials, including plasmonic nanoparticles, nitrogen vacancy centers in diamond, solar cell materials (GaAs, CdTe), light-emitting diode materials (GaN), etc., can be collected in either TEM or STEM mode.
Sample heating in vacuum and in gas environment: Furnace based and MEMS based single and double tilt TEM sample holders for dynamic experiments. Heating rate for MEMS based holder is up to one degree ms-1 with a temperature limit of 1300 °C. Moreover, 532 nm laser can also be used for pulse heating ≈ 10 µm sample area. Several gases (H2, O2, N2, CO, CO2, C2H2, C2H4, CH4, alcohol and water vapors) can be introduced in the sample area, using mass flow controllers, up to 2000 Pa pressure around the sample.

