Summary
The NIST REFPROP database provides the most accurate thermophysical property models for a variety of industrially important fluids and fluid mixtures, including accepted standards. It has proven to be an extremely useful tool used by industry, government, and academia, and it is a primary method for the dissemination of work done by the Thermophysical Properties of Fluids Group.
Description
A collage of REFPROP screens
REFPROP is a computer program, distributed through the Standard Reference Data program of NIST, that provides thermophysical properties of pure fluids and mixtures over a wide range of fluid conditions including liquid, gas, and supercritical phases. It contains critically evaluated mathematical models, with the goal of representing the properties to within the uncertainty of the underlying experimental data used in their development. These properties (such as density, vapor pressure, viscosity, enthalpy, etc.) can used to design solutions to challenges presented by a changing climate, as well as to provide accurate thermophysical data to users in the chemical industry, the petroleum industry, and academia. Over the years the scope of the program has grown from the original focus on refrigerants to a much broader chemical slate; for example it currently contains standards vital to the natural gas industry (GERG-2008), fluids involved in carbon capture and sequestration processes, cryogens used in space applications, widely used industrial chemicals and solvents, air, water and steam, constituents of biofuels, and constituents of surrogates for complex fluids such as diesel and jet fuels. REFPROP is supplied as an easy-to-use graphical-user-interface for Windows users, FORTRAN source code, and a DLL callable from a wide range of platforms and programming environments, such as Excel, LabVIEW, and MatLab. The user can select from a variety of units, flash calculations, properties, tables and plots. More detailed information about the program and how to obtain it can be found at https://www.nist.gov/srd/refprop.
The relevance of REFPROP was validated by the recent 28th Meeting of the Parties to the Montreal Protocol in Kigali, Rwanda, where nearly 200 countries, including the United States, reached a historic agreement to limit the use of potent greenhouse gases known as hydrofluorocarbons (HFCs). These HFC fluids are used in many applications including refrigeration and air conditioning, the manufacture of insulation materials, and as working fluids in power cycles that are used in geothermal and solar applications. These “second generation” refrigerants came into widespread use, following the 1987 adoption of the Montreal Protocol which was implemented to protect the stratospheric ozone layer by phasing out the production and consumption of chlorinated fluorocarbons (CFC’s). The Montreal Protocol may be the most successful environmental agreement in history, with strong evidence for the recovery of the ozone layer. The HFCs are extremely potent greenhouse gases that can be hundreds to thousands of times more potent than carbon dioxide, contributing significantly to climate change and are now subject to regulation. NIST was at the forefront of research on the properties of the original replacement refrigerants, and continues to lead research in properties of new low-global warming potential (low-GWP) fluids that address climate change. REFPROP is at the heart of this research.
ADDITIONAL TECHNICAL DETAILS
Historical Information
The very first version of REFPROP, “REFrigerant PROPerties”, version 1.0, was released in 1989 and contained 15 pure fluids, only binary mixtures, and was based upon the pioneering work of Morrison and McLinden published in NIST Techical Note 1226, “Application of a Hard Sphere Equation of State to Refrigerants and Refrigerant Mixtures”. Customers received a 3.5-inch floppy disk containing an executable program, source code in FORTRAN, and a small binder containing the Users’ Guide. Windows was in its infancy, and the program ran under the DOS operating system. Although REFPROP was referred to as a “database,” throughout its history it has been a data system allowing a user to calculate data according to specific needs. Version 3.0, released in 1991, increased the number of pure fluids to 26 and saw the introduction of transport properties and the extended corresponding-states model. More accurate equations of state, in the form of modified Benedict-Webb-Rubin (MBWR) equations, appeared in version 4.0, released in 1993. Additional fluids continued to be added, and by 1996, version 5 was released with 43 pure fluids. At this point the original FORTRAN source code had become unwieldy and a complete overhaul of the source was done for version 6.0, in 1998. Also in this version Helmholtz-form equations of state were added, and a graphical user interface (written in Delphi) was introduced so that the program now ran under Windows. At this time, an economic assessment study indicated the program was having real impact. By version 7, in 2002, the focus had expanded to include fluids not related to the alternative refrigerants work, and the acronym REFPROP was reborn as REFerence fluid PROPerties. In addition, the GUI was rewritten under Visual Basic, and the ability to interface with Excel and Matlab was introduced. Version 8 was released in 2007 and contained the GERG 2004 model for natural gas properties, 85 pure fluids and values for interaction parameters for 303 binary mixtures. In 2010, version 9 updated the GERG model to GERG 2008, increased the number of pure fluids to 105, and increased the number of fitted binary mixtures to 639. The most recent release, version 9.1, in 2013, increased the number of pure fluids to 121, introduced a 64-bit DLL and improved the convergence of iterative calculations. The next release, anticipated in mid-2017, will contain even more fluids, improvements, and new features.

Associated Publications:
McLinden, M.O., "The history of NIST’s Refrigerants Program: II. Thermophysical Properties Research", ASHRAE Transactions: Symposia, 107, part 2: 699-709 (2001). https://www.nist.gov/node/735216
Economic Assessment of the NIST Alternative Refrigerants Research Program, 98-1 Planning Report, Prepared by TASC for the National Institute of Standards and Technology, Jan 1998 https://www.nist.gov/sites/default/files/documents/director/planning/report98-1.pdf
McLinden, M.O. and Didion, P.E., “Quest for alternatives”, ASHRAE Journal, 29(12): 32-42, 1987.Morrison, G., McLinden, M.O. “Application of a Hard Sphere Equation of State to Refrigerants and Refrigerant Mixtures”, NIST Technical Note 1226, August 1986 https://archive.org/details/applicationofhar1226morr
Low GWP fluids

As discussed above, NIST is very active in measuring and developing thermophysical property models of new “next generation” low-GWP fluids. The Thermophysical Properties of Fluids group is involved with world-wide collaborations to provide high-accuracy Helmholtz-based equations of state for thermodynamic properties, as well as individual wide-ranging fluid-specific correlations for transport properties such as viscosity and thermal conductivity. Some recent publications involving the low-GWP fluids such as R1234yf, R1234ze(Z), R1233zd(E), and Novec 649 as well as other refrigerants are given below.
Associated Publications:
Thol, M., Lemmon, E.W., "Equation of State for the Thermodynamic Properties of trans-1,3,3,3-Tetrafluoropropene [R1234ze(E)],"Int. J. Thermophys., 37(3):28, 2016. http://dx.doi.org/10.1007/s10765-016-2040-6
Huber, M.L. and Assael, M.J., "Correlations for the Viscosity of 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf) and trans-1,2,2,2-Tetrafluoropropene (R1234ze(E))”, Int. J. Refrig. 71: 39-45, 2016. http://dx.doi.org/10.1016/j.ijrefrig.2016.08.007
Perkins, R.A., Huber, M.L., Assael, M.J., "Measurements of the Thermal Conductivity of 1,1,1,3,3-Pentafluoropropane (R-245fa) and Correlations for the Viscosity and Thermal Conductivity Surfaces", J. Chem. Eng. Data, 61, 3286-3294, 2016. http://dx.doi.org/10.1021/acs.jced.6b00350
Akasaka, R. and Lemmon, E. W., "A Helmholtz energy equation of state for cis-1,1,1,4,4,4-hexafluoro-2-butene (R-1336mzz(Z))," The 8th Asian Conference on Refrigeration and Air-Conditioning, May 15-17, 2016, Taiwan.
Akasaka, R., Zhou, Y., Lemmon, E. W., "A Fundamental Equation of State for 1,1,1,3,3-Pentafluoropropane (R-245fa)", J. Phys. Chem. Ref. Data, 44, No. 1, 2015. https://www.nist.gov/node/554336
Lemmon, E. W., Zhou, Y., "Equation of State for the Thermodynamic Properties of 1,1,2,2,3-Pentafluoropropane (R-245ca)", Int. J. Thermophys. 37:27, 2016. http://dx.doi.org/10.1007/s10765-016-2039-z
Mondejar, M.E., McLinden, M.O., Lemmon, E.W., "Thermodynamic Properties of Trans-1-chloro-3,3,3-trifluoropropene (R1233zd(E)): Vapor Pressure, p-rho-T Data, Speed of Sound Measurements and Equation of State," J. Chem. Eng. Data, 60:2477-2489, 2015. http://dx.doi.org/10.1021/acs.jced.5b00348
Lemmon, E. W., Span, R., "Thermodynamic Properties of R-227ea, R-365mfc, R-115, and R-13I1", J. Chem. Eng. Data 60(12): 3745-3758, 2015. http://dx.doi.org/10.1021/acs.jced.5b00684
McLinden, M.O., Perkins, R.A., Lemmon, E.W., Fortin, T.J., “Thermodynamic Properties of 1,1,1,2,2,4,5,5,5-nonafluoro-4-(trifluoromethyl)-3-pentanone: Vapor Pressure, (p, ρ, T) Behavior, and Speed of Sound Measurements, and Equation of State,” J. Chem. Eng. Data 60:3636-3659, 2015. http://dx.doi.org/ 10.1021/acs.jced.5b00623
McLinden, M.O., Kazakov, A.F., Brown, J.S., and Domanski, P.A., “A thermodynamic analysis of refrigerants: Possibilities and tradeoffs for Low-GWP refrigerants”, Int. J. Refrig. 38:80-92, 2014. http://dx.doi.org/10.1016/j.ijrefrig.2013.09.032
Kazakov, A., McLinden, M.O. and Frenkel, M., “Computational design of new refrigerant fluids based on environmental, safety, and thermodynamic characteristics”, Ind. Eng. Chem. Res. 51(3): 12537-12548, 2012. http://dx.doi.org/10.1021/ie3016126
Richter, M., McLinden, M.O., and Lemmon, E.W., "Thermodynamic Properties of 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf): Vapor Pressure and p-rho-T Measurements and an Equation of State," J. Chem. Eng. Data, 56(7):3254-3264, 2011. http://dx.doi.org/10.1021/je200369m
Perkins, R.A. and Huber, M.L., "Measurement and Correlation of the Thermal Conductivity of 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf) and trans-1,3,3,3-Tetrafluoropropene (R1234ze(E))" J. Chem. Eng. Data , 56(12), pp. 4868-4874, 2011. http://dx.doi.org/10.1021/je200811n
Natural Gas
REFPROP contains standards used in the natural gas industry such as GERG-2008 and AGA-8. In addition, our researchers are involved in work to improve the representation of mixtures comprising fluids in natural gas. We also actively participate in and lead standards committees related to thermodynamic properties of natural gas.

Associated Publications:
GPA Standard 2145-16, Table of Physical Properties for Hydrocarbons and Other Compounds of Interest to the Natural Gas and Natural Gas Liquids Industries, GPA Midstream Association, 2016.
Richter, M. and McLinden, M.O., "Vapor-phase (p,ρ,T,x) behavior and virial coefficients for the (Methane + Propane) system", J. Chem. Eng. Data 59:4151-4164, 2014. http://dx.doi.org/10.1021/je500792x
Mansfield, E., Bell, I.H., Outcalt, S.L., “Bubble-Point Measurements of n-Propane + n-Decane Binary Mixtures with Comparisons of Binary Mixture Interaction Parameters for Linear Alkanes”, J. Chem. Eng. Data 61(7): 2573-2579, 2016. http://dx.doi.org/10.1021/acs.jced.6b00258
Lemmon, E.W., “Better defining the Uncertainties for the AGA-8 Equation”, Pipeline Research Council International Catalog No. PR-381-12604-R01. 2014.
Surrogate models for complex fluids

Complex mixtures such as aviation and transportation fuels, which are made up of hundreds or thousands of chemical species, may be approximated by a “surrogate mixture” where the properties of the complex fuel are represented by a relatively small (generally less than 10) number of constituent fluids whose properties are well known. The composition of the surrogate mixture is determined by fitting the real fuel data properties (such as density, cetane number, carbon type, viscosity, etc.). REFPROP has been used to model a variety of surrogates for fuels such as RP1/2, Jet-A, biodiesel, S8, and low-sulfur diesel.
Associated Publications:
Mueller, C.J., Cannella, W.J., Bays, J. T., Bruno, T. J., DeFabio, K., Dettman, H., Gieleciak, R. M., Huber, M. L., Kweon, C.-B., McConnell, S.S., Pitz, W.J., Ratcliff, M. A., “Diesel Surrogate Fuels for Engine Testing and Chemical-Kinetic Modeling: Compositions and Properties”, Energy & Fuels 30:1445-1461, 2016. http://dx.doi.org/10.1021/acs.energyfuels.5b02879
Mueller, C.J., Cannella, W. J., Bruno, T. J., Bunting, B., Dettman, H., Franz, J. A., Huber, M.L., Natarajan, M., Pitz, W.J., Ratcliff, M. A., Wright, K., “Methodology for Formulating Diesel Surrogate Fuels with Accurate Compositional, Ignition-Quality, and Volatility Characteristics”, Energy & Fuels 26:3284-3303, 2012. http://dx.doi.org/10.1021/ef300303e
Windom, B., Huber, M. L., Bruno, T.J., Lown, A.L., Lira, C.T., "Measurements and Modeling Study on a High-Aromatic Diesel Fuel", Energy & Fuels 26:1787-1797, 2012. http://dx.doi.org/10.1021/ef201861b
Huber, M.L., Lemmon, E.W., and Bruno, T.J., “Surrogate mixture model for the thermophysical properties of aviation fuel Jet-A”, Energy & Fuels, 24:3565-3571, 2010. http://dx.doi.org/10.1021/ef100208c
Huber, M.L., Lemmon, E.W., Ott, L. S., and Bruno, T. J., "Preliminary Surrogate Mixture Models for the Thermophysical Properties of Rocket Propellants RP-1 and RP-2", Energy & Fuels, 23:3083-3088, 2009. http://dx.doi.org/10.1021/ef900216z
Huber, M. L., Lemmon, E.W., Kazakov, A., Ott, L.S., and Bruno, T.J., "Model for Thermodynamic Properties of a Biodiesel Fuel", Energy & Fuels, 23:3790-3797, 2009. https://pubs.acs.org/doi/10.1021/ef900159g
Huber, M.L., Lemmon, E.W., Diky, V., Smith, B.L., and Bruno, T. J., "Chemically Authentic Surrogate Mixture Model for the Thermophysical Properties of a Coal-Derived-Liquid Fuel", Energy & Fuels, 22:3249–3257, 2008. http://dx.doi.org/ 10.1021/ef800314b
Huber, M.L., Smith, B.L., Ott, L. S., and Bruno, T. J., "Surrogate Mixture Model for the Thermophysical Properties of Synthetic Aviation Fuel S-8: Explicit Application of the Advanced Distillation Curve", Energy & Fuels, 22:1104-1114, 2008. http://dx.doi.org/10.1021/ef700562c
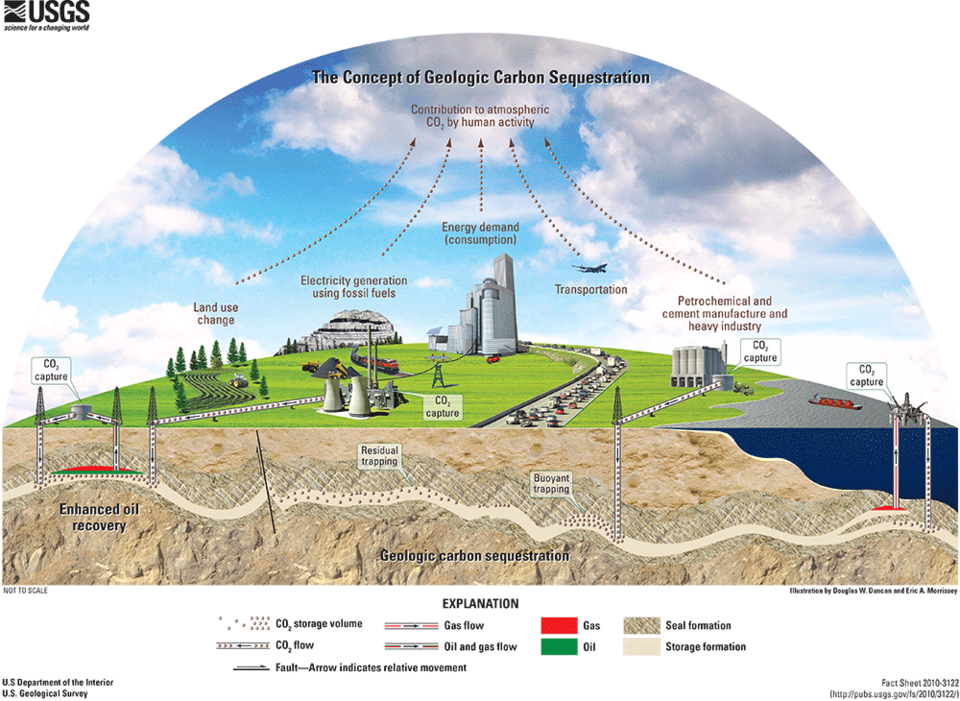
Carbon Capture and Sequestration
If carbon capture and sequestration (CCS) becomes widely used, it will be necessary to compress and transport large amounts of CO2 through pipelines. The pipelines may have significant impurities, including water. It is important to know the thermophysical properties, including the location of the phase boundary (dew-point line) of carbon dioxide and its mixtures. REFPROP contains models for such mixtures, and continuing research (both experimental and theoretical) is being performed to improve these models.
Associated Publications:
Harvey, A.H., Bell, I.H., Huber, M.L., Laesecke, A., Lemmon, E.W., Meyer, C.W., Muzny, C.D., Perkins, R.A.,"Thermophysical Properties of Carbon Dioxide and CO2-Rich Mixtures", Report to US Department of Energy, 2016. https://www.nist.gov/node/1077901
Bell, I.H. , Lemmon, E.W., Harvey, A.H., "Algorithms for the calculation of psychrometric properties from multi-fluid Helmholtz-energy- explicit models", International Journal of Refrigeration, 87, 2017. https://doi.org/10.1016/j.ijrefrig.2017.10.001
Schultz, A.J., Kofke, D.A., Harvey, A.H., “Molecular-based virial coefficients of CO2-H2O Mixtures”, AIChE J., 61(9): 3029-3037, 2015. http://dx.doi.org/10.1002/aic.14880
Huber, M.L., Sykioti, E.A., Assael, M.J., Perkins, R.A., “Reference Correlation of the Thermal Conductivity of Carbon Dioxide from the Triple Point to 1100 K and up to 200 MPa”, J. Phys. Chem. Ref. Data 45(1): 03102. http://dx.doi.org/10.1063/1.4940892
Meyer, C. W., Harvey, A.H., "Dew-point Measurements for Water in Compressed Carbon Dioxide", AIChE J., 61(9):2913-2925, 2015. http://dx.doi.org/10.1002/aic.14818
Space Applications
Much of the work on properties of cryogens, such as oxygen, done in the 1960s as part of the space program was done at NIST. While the early equations of state for these fluids have been updated, the new EOS and transport models in REFPROP today are still largely based on the early experimental data. REFPROP contains fluids that can be used as propellants, and fluids that can be used to model the atmosphere on our neighboring planets.

Associated Publications:
Richardson, I. A., Leachman, J. W., Lemmon, E. W., “Fundamental Equation of State for Deuterium”, J. Phys. Chem. Ref. Data, 43(1):013103, 2014. http://dx.doi.org/10.1063/1.4864752
Muzny, C.D., Huber, M.L., and Kazakov, A.F.,"Correlation for the Viscosity of normal hydrogen obtained from symbolic regression", J. Chem. Eng. Data, 58(4):969-979, 2013. http://dx.doi.org/10.1021/je301273j
Assael, M.J., Assael. J.-A.M., Huber, M.L., Perkins, R.A. and Takata, Y., "Correlation of the Thermal Conductivity of Normal and Parahydrogen from the Triple Point to 1000 K and up to 100 MPa", J. Phys. Chem. Ref. Data, 40(3):033101, 2011. http://dx.doi.org/10.1063/1.3606499
Leachman, J.W., Jacobsen, R.T, Penoncello, S.G., Lemmon, E.W., "Fundamental Equations of State for Parahydrogen, Normal Hydrogen, and Orthohydrogen,"J. Phys. Chem. Ref. Data, 38(3):721-748, 2009. http://dx.doi.org/10.1063/1.3160306
Common Industrial Fluids

REFPROP contains models for the thermodynamic and transport properties of common industrial fluids and their mixtures. We strive to include additional fluids in response to customer needs. Currently REFPROP contains common solvents such as benzene and toluene, small alcohols (methanol, ethanol, propanol, etc.) and working fluids such as ammonia and steam. REFPROP contains the current IAPWS standards for the properties of water and steam. Current research also is underway to improve the representation of mixtures of fluids using Helmholtz-energy explicit models.
Associated Publications:
Gao, K., Wu, J., Zhang, P., Lemmon, E.W.,"A Helmholtz Energy Equation of State for Sulfur Dioxide", J. Chem Eng. Data, 61(8):2859-2872, 2016. http://dx.doi.org/10.1021/acs.jced.6b00195
Gedanitz, H., Davila, M.J., Lemmon, E.W., “Speed of sound measurements and a fundamental equation of state for cyclopentane”, J. Chem. Eng. Data, 60(5):1331-1337, 2015. http://dx.doi.org/10.1021/je5010164
Bell, I. H., Lemmon, Eric W., "Automatic fitting of binary interaction parameters for multi-fluid Helmholtz-energy-explicit mixture models", J. Chem. Eng. Data, 61 (11):3752-3760, 2016. http://dx.doi.org/10.1021/acs.jced.6b00257
Koutian, A., Assael, M.J., Huber, M.L., Perkins, R.A., "Reference Correlations of the Thermal Conductivity of Ethene and Propene", J. Phys. Chem. Ref. Data, 45:033104, 2016. http://dx.doi.org/10.1063/1.4958984
Vassiliou, C.M., Assael, M.J., Huber, M.L., Perkins, R.A., "Reference Correlations of the Thermal Conductivity of Cyclopentane, iso-Pentane, and n-Pentane", J. Phys. Chem. Ref. Data, 44(3):033102, 2015. http://dx.doi.org/10.1063/1.4927095
Avgeri, S., Assael, M.J., Huber, M.L., Perkins, R.A., " Reference Correlation of the Viscosity of Toluene from the Triple Point to 675 K and up to 500 MPa", J. Phys. Chem. Ref. Data 44(3):033101, 2015. http://dx.doi.org/10.1063/1.4926955
Avgeri, S., Assael, M.J., Huber, M.L., and Perkins, R.A., "Reference Correlation of the Viscosity of Benzene from the Triple Point to 675 K and up to 300 MPa", J. Phys. Chem. Ref. Data, 43(3): 033103, 2014. http://dx.doi.org/10.1063/1.4892935
Mylona, S.K., Antoniadis, K.D., Assael, M.J., Huber, M.L., Perkins, R.A., “Reference correlations for the thermal conductivity of o-xylene, m-xylene, p-xylene, and ethylbenzene from the triple point to 700 K and moderate pressures”, J. Phys. Chem. Ref. Data, 43(4):043104, 2014. http://dx.doi.org/10.1063/1.4901166
Assael, M.J., Koini, I.A., Antoniadis, K.D., Huber, M.L., Perkins, R.A., "Reference correlation of the thermal conductivity of sulfur hexafluoride from the triple point to 1000 K and up to 150 MPa", J. Phys. Chem. Ref. Data, 41:023104. http://scitation.aip.org/content/aip/journal/jpcrd/41/2/10.1063/1.4708620. (Please also see the erratum: http://scitation.aip.org/content/aip/journal/jpcrd/43/3/10.1063/1.4885454).
Michailidou, E. K.; Assael, M. J.; Huber, M. L., Perkins, R.A., “Reference correlation for the viscosity of n-heptane from the triple point to 600 K and up to 248 MPa”, J. Phys. Chem. Ref. Data, 43(2):023103, 2014. http://dx.doi.org/10.1063/1.4875930
Huber, M.L., Perkins, R.A., Friend, D.G., Sengers, J.V., Assael, M.J., Metaxa, I.N., Miyagawa, K., Hellmann, R., Vogel, E., "New International Formulation for the Thermal Conductivity of H2O", J. Phys. Chem. Ref. Data,.41(3): 1-23, 2011. http://dx.doi.org/10.1063/1.4738955
Disclaimers
Certain commercial equipment, instruments, materials, systems, software, and trade names may be identified throughout this site in order to specify or identify technologies adequately. Such identification is not intended to imply recommendation or endorsement by NIST or any other party, nor is it intended to imply that the systems or products identified are necessarily the best available for the purpose. All data and other information posted on this site is provided as a public service and is provided “AS IS.” NIST MAKES NO WARRANTY OF ANY KIND, EXPRESS, IMPLIED OR STATUTORY, INCLUDING, WITHOUT LIMITATION, THE IMPLIED WARRANTY OF MERCHANTABILITY, FITNESS FOR A PARTICULAR PURPOSE, NON-INFRINGEMENT AND DATA ACCURACY.
By selecting external links, you will be leaving NIST webspace. Links to other websites are provided because they may have information that would be of interest to you. No inferences should be drawn on account of other sites being referenced, or not, from this page. There may be other websites that are more appropriate for your purpose.

