Summary
By using machine learning approaches, we seek to elucidate underlying universal characteristics of fluids and fluid mixtures that enable property prediction. Namely, thermodynamic properties of these systems which play a significant role in applications ranging from medicine to industrial manufacturing. A deeper understanding is expected to facilitate improved pharmaceutical development, cheaper chemical separations, and lower energy alternatives to chemical processes.
Description
Machine learning approaches are being studied by NIST for enable property prediction of fluids and fluid mixtures.
Understanding the thermodynamic properties of fluids and fluid mixtures is of central importance in many fields of science and engineering ranging from medicine to consumer products. The nature of the particles in a fluid can vary greatly depending on the type of interactions present, e.g., dipole-dipole, (screened) electrostatics, van den Waals, etc. However, virial expansions offer a “universal” description of fluids by expressing properties as a series expansion involving coefficients which are related to this interaction but are not specific to its form. This effectively coarse-grains out the details of such interactions, and it is known that certain universal behaviors of fluids can be observed based on the value of low order coefficients, for example, the Noro-Frenkel “Extended” Law of Corresponding States. [1] Using advanced sampling methods, it is now possible to compute many higher order virial coefficients for a wide range of fluids. [2] This project aims to search for universal properties of fluids using virial coefficients as physically-informed descriptors in machine learning (ML) algorithms. By combining previously developed “extrapolation” methodologies and flat-histogram simulations we can obtain a sufficient amount of data to train large, complex ML models. [3]
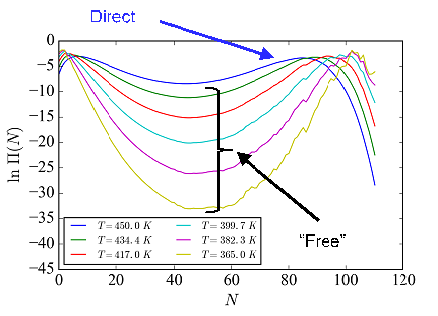
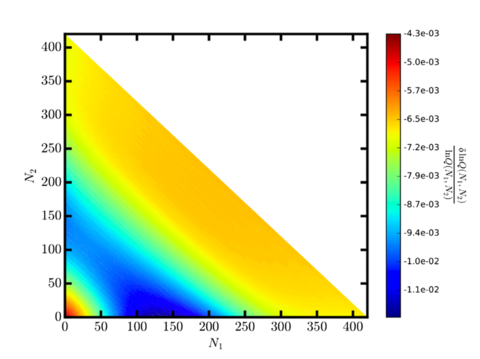
This approach can also be extended to the study of confined fluids, a major area of importance in chemical separations and industrial manufacturing, and one which is critically needed to meet the “Energy Intensity of Chemical Processing” Grand Challenge laid out the National Research Council. NIST provided a $500,000 Advanced Manufacturing Technology Consortia (AMTech) Program Planning Award to initiate a collaborate effort with the American Chemical Society Green Chemistry Institute (ACS GCI) Chemical Manufacturers Roundtable to reduce the energy requirements of industrially-relevant separations by finding viable alternatives. As part of this ALTSEP project we have sought to simulate different fluids with a variety of different interactions and types of confinement. This project also aims to investigate how much ML can be used to find universal fluid properties and other thermodynamic characteristics of such fluids.
References
- Extended Corresponding-states Behavior for Particles with Variable Range Attractions
- Mayer Sampling: Calculation of Cluster Integrals using Free-Energy Perturbation Methods
- Flat-histogram Extrapolation as a Useful Tool in the Age of Big Data
RELATED PUBLICATIONS
1. Mahynski, N. A., Jiao, S., Hatch, H. W., Blanco, M. A., and Shen, V. K., "Predicting structural properties of fluids by thermodynamic extrapolation," Journal of Chemical Physics, 148, (2018).
2. Witman, M., Mahynski, N. A., and Smit, B., "Flat-Histogram Monte Carlo as an Efficient Tool To Evaluate Adsorption Processes Involving Rigid and Deformable Molecules," Journal of Chemical Theory and Computation, 14, 6149-6158 (2018).
3. Hatch, H. W., Mahynski, N., Medina, M. A. B., Shen, V. K., and Jiao, S., "Predicting virial coefficients and alchemical transformations by extrapolating Mayer-sampling Monte Carlo simulations," (2017).
4. Mahynski, N. A., Errington, J. R., and Shen, V. K., "Multivariable extrapolation of grand canonical free energy landscapes," Journal of Chemical Physics, 147, (2017).
5. Mahynski, N. A., Blanco, M. A., Errington, J. R., and Shen, V. K., "Predicting low-temperature free energy landscapes with flat-histogram Monte Carlo methods," Journal of Chemical Physics, 146, (2017).
6. Day, R. D., Roseneau, D. G., Vander Pol, S. S., Hobson, K. A., Donard, O. F. X., Pugh, R. S., Moors, A. J., and Becker, P. R., "Regional, temporal, and species patterns of mercury in Alaskan seabird eggs: Mercury sources and cycling or food web effects?," Environmental Pollution, 166, 226-232 (2012).
7. Day, R. D., Roseneau, D. G., Berail, S., Hobson, K. A., Donard, O. F. X., Vander Pol, S. S., Pugh, R. S., Moors, A. J., Long, S. E., and Beckert, P. R., "Mercury Stable Isotopes in Seabird Eggs Reflect a Gradient from Terrestrial Geogenic to Oceanic Mercury Reservoirs," Environ. Sci. Technol., 46, 5327-5335 (2012).
8. Point, D., Sonke, J. E., Day, R. D., Roseneau, D. G., Hobson, K. A., Vander Pol, S. S., Moors, A. J., Pugh, R. S., Donard, O. F. X., and Becker, P. R., "Methylmercury photodegradation influenced by sea-ice cover in Arctic marine ecosystems," Nature Geoscience, 4, 188-194 (2011).

