Summary
Biomarkers play an increasingly important role in disease detection and treatment. As one of the Biomarker Reference Laboratories (BRLs) in the Early Detection Research Network (EDRN) - an initiative of the National Cancer Institute (NCI), NIST is developing biomarker-based systems, employing front-end technologies and novel materials such as single-walled carbon nanotubes (SWCNTs) and quantum dots (Qdots), which are potentially useful for the detection and treatment of cancers.
Description
Intended impact
Although significant progress has been made in both the understanding and treatment of cancer during the last thirty years, it remains the second leading cause of death in the US. The cancer community has set a goal to eliminate cancer-related suffering and death by 2015. To achieve this goal, not only better therapies are required but also improved methods to assess an individual's risk of developing cancer, to detect cancers at early stages when they can be treated more effectively, to distinguish between aggressive and nonaggressive cancers, and to monitor recurrence and response to therapy. In the past several years, there has been increasing interest and enthusiasm in molecular biomarkers as tools for cancer early detection, diagnosis, prognosis and treatment. Biomarkers are cellular indicators of physiologic state and/or of change during a disease process. Such markers, including DNA/RNA, protein, and metabolites, provide information on the occurrence and progression of the disease. Recent advances in high-throughput technologies in genomics, proteomics, and metabolomics have facilitated biomarker discovery. As more potential biomarkers are discovered, further studies are needed to validate these markers. The ultimate use of these biomarkers is in clinical applications for cancer detection and treatment.
Objective
Conduct research and development on biomarkers and technologies for early cancer detection and treatment.
Goals
- Discovery of new cancer biomarkers using high-throughput technologies;
- Validation of newly identified cancer biomarkers in clinical settings.
- Translation of validated biomarkers into potentially useful clinical applications.
Research activities and technical approach
NIST is actively involved in various stages of cancer biomarker development in three lines of research. The first line of research focuses on discovery of new cancer biomarkers. Mitochondrial DNA (mtDNA) is considered more susceptible to environmental damages than genomic DNA. As a result, mtDNA lesions may accumulate in the cell and ultimately lead to malignant transformations. Recently, mtDNA lesions have been discovered to be characteristic of many cancers. We have recently initiated a project to identify oxidatively induced mtDNA lesions correlated with mitochondrial function changes in cancer cell lines as potential biomarkers for early cancer detection. We use LC/MS and GC/MS to measure the levels of DNA base lesions in the mitochondrial genome by collaborating with scientists within the same division.
In the second line of research, we are validating newly identified cancer biomarkers with clinical tissue samples. Many of these biomarkers are proteins for which accurate quantitation is required to provide meaningful information for clinical decision-making. Immunohistochemistry (IHC) has been traditionally used for analyzing proteins at the single-cell level. However, the photobleaching of the organic fluorophores used in conventional IHC methods has made quantitation problematic. To overcome this hurdle, we have developed a quantitative immunohistochemical (IHC) assay method using quantum dots (Qdots), a semiconductor nanocrystal, as the fluorophore. To improve assay specificity and sensitivity, we have also developed novel IgY antibodies for two biomarkers, HER2 and telomerase, and demonstrated their superiority over mammalian IgGs in a Qdot-based IHC system. We are currently using this system to validate multiple biomarkers of the PI3K/AKT/mTOR pathway including PTEN, mTOR, and E4-BP1, which are potential markers of prostate, breast and colon cancer progression.
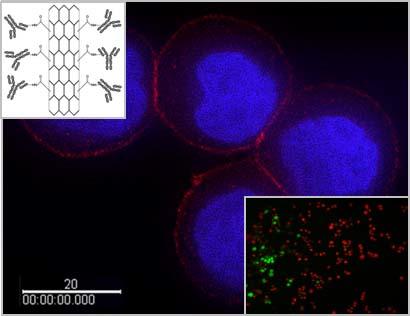
Our third line of research involves translation of established biomarkers into potentially useful clinical applications. HER2 is an example of such a biomarker whose status plays a pivotal role in therapeutic decision making for patients with breast cancer and in determining their clinical outcomes. We are developing HER2-based systems employing front-end technologies and novel materials. One such technology is single-walled carbon nanotubes (SWCNTs). SWCNTs have strong Raman signals and near infrared (NIR) absorption, which can be exploited to facilitate cancer detection and therapy. In collaboration with researchers at Rutgers University, we have constructed a HER2 IgY-SWCNT complex by covalently functionalizing SWCNTs with anti-HER2 IgY antibodies. We have demonstrated the dual functionality of this complex for both detection and selective photothermal ablation of breast cancer cells in an in vitro model. An advantage of this system is that it does not require internalization by the cancer cells to achieve the selective destruction. We are currently extending the application of this system to other cancers.
Major Accomplishments
- Developed IgY antibodies for HER2 and telomerase.
- Developed a HER2 IgY-SWCNT complex and demonstrated its use for detection and selective destruction of breast cancer cells in an in vitro model.
- Developed a Qdot-based quantitative IHC assay system.

