Summary
Due to their small size, nanoparticles can enter living organisms in previously unforeseen ways and their immense surface area results in higher reactivity compared to corresponding bulk materials. New toxicological testing methods, accurate measurement methods, reference materials and documentary standards are urgently needed to support risk assessments and regulatory policy decisions regarding materials containing nanoparticles.
Description
Impact
Nanomaterials have already been incorporated into a large range of commercial products including pharmaceuticals, sunscreens, automobile additives, personal care products, detergents and plastics. Although nanotechnology is changing the way we live, the accidental release of nanomaterials into the environment poses an unknown public health risk. Due to the significant physical and chemical differences between materials in nanoparticle form and bulk material form, new methods are needed to assess nanomaterial safety.
Workers, consumers and living organisms are exposed to nanomaterials through inhalation, dermal contact or water exposure. Understanding the environmental, health and safety effects of nanomaterials through scientifically-based and consensus-based standard test methods is critical for assessment of nanotechnology safety and will lead to increased commerce on a global level. A common issue encountered in such studies is accurate nanoparticle concentration expression, given the critical importance of the dose-response relationship. Furthermore, particle concentration can be alternatively expressed as mass, number of particles (molar) or surface area units, often complicating inter-study comparisons. Accurate gravimetric measurement using analytical balances may not be practical in the sub-microgram range, as this is approaching the sensitivity limit for most instruments.
Approach
The quartz crystal microbalance (QCM) is a nanogram-resolution mass-sensing technique based on the piezoelectric effect. The technique is quite versatile, as it possesses a wide detection range and can be applied to deposited mass measurements in the gas or liquid phase. Deposition of a liquid drop on one of the faces of an oscillating crystal causes a shift in its resonant frequency, enabling accurate residue film mass measurements following solvent evaporation. We apply quartz crystal microgravimetry to accurately quantify nanoparticle concentrations in size-selected colloidal suspensions in a low boiling point solvent. By depositing a fixed volume of the nanoparticle colloidal solution and determining the dry residue mass after solvent evaporation, we measured the nanoparticle concentration and expressed it in mass per unit volume. Concentration data on size-selected nanoparticle batches enables the calculation of the nanoparticle extinction coefficient and will facilitate practical applications of absorption-based nanoparticle concentration determination when accurate quantitation of a small nanoparticle sample is required.
Silver nanomaterials are increasingly being used as antimicrobial agents in medical devices. A recent joint study with the Food and Drug Administration's Center for Devices and Radiological Health assessed the in vitro hemolytic potential of silver particles in human blood to determine which physical and chemical particle properties contribute to mechanisms of red blood cell damage.
NIST staff are active participants in ISO TC 229 (Nanotechnologies) technical groups.
Major Accomplishments
A colloidal nanoparticle concentration measurement method using quartz microgravimetry was developed with partners from MIT and the University of Massachusetts. Application of a small drop of the nanoparticle suspension in a volatile solvent to the crystal surface leaves a dry nanoparticle residue after solvent evaporation, then the shift in the crystal resonant frequency is recorded. The microbalance was calibrated to determine the mass-resonance frequency shift relationship applicability range. First, a stock solution was prepared from a dry nanoparticle powder weighed on an analytical balance and dispersed in methanol using mild sonication. Next, a series of 20 μL pure methanol drops were deposited and evaporated to determine instrument noise level and reproducibility. Finally, a series of 20 μL Si and Ag nanoparticle suspension drops (particle concentration ranged from 5 to 6000 μg/ml) in methanol were deposited and plateau frequencies were recorded (Figure 1).
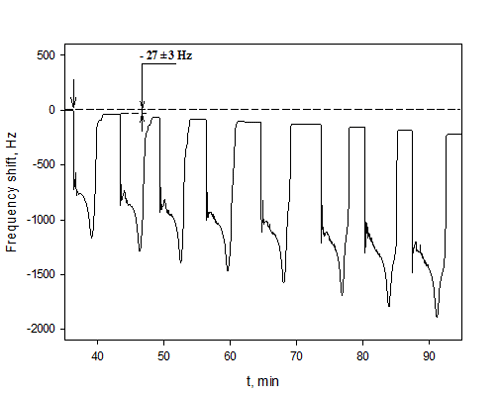
| Figure 1. Quartz microbalance resonance frequency trace recorded during the sequential injection of 20 μl drops of methanol containing 120 μg/ml silicon nanoparticles. The presence of dry nanoparticle residue after methanol evaporation decreases the crystal resonance frequency by 27 Hz. |
A standard curve (Figure 2) indicated that the QCM frequency responded linearly to dry nanoparticle loading up to c = 1300 μg/ml. Utility of QCM microgravimetry was shown in determining the molar concentrations of five size-selected Si nanocrystal samples with median diameters of 1.1, 1.4, 5.2, 7.1, and 14.8 nm and calculating size-dependent nanocrystal extinction coefficients that could be utilized for colloidal nanoparticle concentration measurements using ultraviolet-visible spectrophotometry.
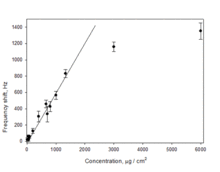
| Figure 2. Quartz crystal microbalance calibration obtained from dilution series of Si and Ag nanoparticles in methanol. |
Four silver particle powders (two in the nanometer-diameter ranged, two in the micrometer-diameter range) were dispersed in water and characterized using transmission electron microscopy, dynamic light scattering, surface-enhanced Raman spectroscopy, and surface charge measurement. Particle size and agglomeration were dependent on the suspension media. While significant size changes occurred to the silver particles due to interaction with media components, the results indicated that the higher level of hemolysis observed with nanoparticles compared to larger particles is related to an increased silver ion release rate and a greater surface area. Our in vitro hemolysis results suggest that commercially available blood-contacting medical devices containing silver nanoparticles should be carefully evaluated for their hemolytic potential.
We are leading the work on ISO Technical Report 16197 "Compilation and Description of Toxicological and Ecotoxicological Screening Methods for Engineered and Manufactured Nanomaterials" with partners from Evonik, the National Institute for Occupational Safety and Health, BASF, DuPont and General Electric. This report will aid the process of toxicological screening of engineered and manufactured nanomaterials prior to full-scale toxicological analysis. The eagerly awaited technical report will provide information on methods that can be used to screen nanomaterials in order to determine whether to continue development of a product, take on the cost of performing a comprehensive toxicological analysis, or determine if appropriate controls are in place to continue nanomaterials research in the laboratory.
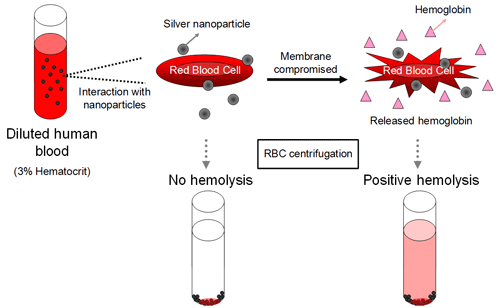
Figure 3. Measurement of silver nanoparticle hemolytic properties.
Associated Product(s)
Project Brief (PDF)

