Bryant C. Nelson (Fed)
Research Interests
Extracellular vesicles for drug delivery and gene therapy applications
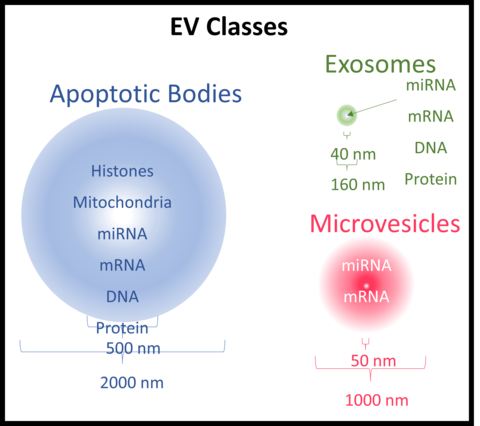
Extracellular vesicles (EVs) are non-replicating lipid bilayer vesicles that are shed by almost all types of eukaryotic and prokaryotic cells under both physiological and pathological conditions. Extending in size over the nanometer to micrometer size range (Figure 1), EVs are biological vectors intimately involved in cell-to-cell communication via the lateral transfer of coding and non-coding RNAs, small amounts of DNA, surface and cytoplasmic proteins, phospholipids and soluble small molecules to recipient cells. They have been shown, in many fundamental and preclinical studies, to possess low immunogenicity, minimal complement activation potential, low toxicity and excellent biocompatibility. In the context of EV technology development for the delivery of nucleic acid- or protein-based therapeutics, there exists a critical need to develop robust and reproducible methods for isolating, purifying and processing EV samples from a variety of sources for downstream analytical characterization. Please visit: https://pubmed.ncbi.nlm.nih.gov/32885720/. Our team focuses on developing and standardizing analytical methods and developing reference materials (RMs) for characterizing the physicochemical and biological key quality attributes of mammalian cell-derived EVs. Using mass spectrometry (MS)-based proteomics and other analytical techniques, one of our current goals is to positively identify and quantify key EV protein surface markers (tetraspanins) that will enable discrimination of marker-positive EV populations from non-EV particles, contaminants and other cellular debris. This work will facilitate comparability and harmonization of EV measurements across different instruments and laboratories, ensuring the quality and reliability of EV-based therapeutic products.
Other Research Areas of Significance:
- Development of standardized methods and measurements for isolating, purifying and processing EVs to enable their downstream utility in next-generation regenerative medicines and advanced therapies (RMATs) and as general drug delivery vectors.
- Development of standardized methods and measurements that allow the scalable use of EVs as clinical biomarkers in plasma and/or as in vitro biomarkers of cell induced toxicity (liquid biopsies). Both processes measure expression levels of key biological molecules. A key challenge is the low level of biomarkers (e.g., cancer biomarkers: proteins, miRNAs) and the elevated levels of biological contaminants (e.g., lipoproteins, platelet-derived EVs) in plasma samples that may preclude early detection of the targeted biomarker.
- Development of standardized methods and measurements that will allow the production and/or characterization of EV-based biological reference materials. Please visit: https://doi.org/10.3390/biom16010066
- For additional information on all of these ongoing research areas, please visit: Gene Delivery Systems
Lipid nanoparticle (LNP) documentary standards and analytical test methods
LNPs have emerged as an important nanoscale drug delivery technology for the in vivo intracellular delivery of therapeutic proteins and nucleic acids. The safe and efficient delivery of therapeutic proteins and nucleic acids to cells and tissues can enable the effective treatment and prevention of a wide range of human diseases (e.g., cancers, infectious diseases, etc.) and facilitate innovations in regenerative medicine and other advanced therapies. Documentary standards and robust analytical methodology for establishing high quality characterization of ribonucleic acids - RNAs (specifically mRNA) encapsulated in LNPs are critically needed by mRNA-LNP product manufacturers and regulatory authorities. Our lab is co-leading along with ASTM International, the FDA and key industry and academic partners, the development of both documentary standards and analytical test methods that will enable the characterization of mRNA encapsulation efficiency (e.g., the amount of free versus total mRNA encapsulated in LNP vectors), the qualitative or quantitative extraction of LNP encapsulated mRNA and the characterization of identity, integrity and purity of mRNA before and after encapsulation in LNP vectors. It is expected that at the conclusion of this multiyear project, a comprehensive and impactful standard guide that describes the key technical considerations and recommended best practices, including suitable analytical methods and tools, will be available to industry, academia and regulatory stakeholders worldwide. The standard will enable stakeholders to make informed decisions regarding the best analytical methodology that is fit-for-purpose for their individual mRNA-LNP product formulation measurement needs. The development of innovative and reproducible test methods based on the consensus recommendations presented in the guide are currently under discussion with stakeholders. For more information, please visit mRNA-LNP Standard and mRNA-LNP Standard Commentary.
Awards and Honors
NIST Outstanding Mentor Award (2017)
Sigma Xi Distinguished Lecturer (2015 to 2017)
Sigma Xi Member (2005 to present)
Professional Committees and Service
Chair of the ACS – Division of Analytical Chemistry, (2025 to present)
American Chemical Society (ACS) Councilor – Division of Analytical Chemistry (2019 to 2023)
ACS Project SEED Awards Committee Co-Chair (2022 to present)
Member – Interagency Coordinating Committee on the Validation of Alternative Methods (ICCVAM) Nano Working Group (since 2018)
ASTM International E56.08 Chair – Nano-Enabled Medical Products (2017 to present)
Member - NIST Research Advisory Committee (2007 to 2010)
NIST Sigma Xi President (2008 to 2009)
Member – American Chemical Society (1998 to present)
Advisor – Biomedical Science Careers Program – Harvard Medical School (1996 to present)

