Problem
patent description
Short Tandem Repeat (STR) markers is a method for genetic profiling. It involves four to five nucleotide repeats allowing scientists to obtain relatively precise and accurate information. For example, several genetic conditions are linked to three nucleotide repeats.
Cell line defines population of cells that can be maintained in culture for an extended period while retaining stability of certain phenotypes and functions. Cell lines are usually clonal, meaning the entire population originated from a single common ancestor cell.
At this time, additional (14) mouse STR markers are needed to address the limitations of the current mouse cell line authentication assay to increase resolution and discrimination between same strain mouse samples. These additional STR markers will either be added to the existing assay or will stand alone as a separate assay.
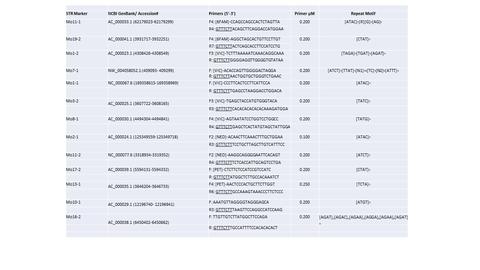
Invention
A method, kit and primers for determining alleles present in a DNA sample that is sourced from either a mouse or a cell line derived from a mouse. A set of STR marker loci are selected that can be amplified together in a multiplex amplification reaction. The set of STR marker loci are selected from the group consisting of: 18-3, 4-2, 6-7, 15-3, 6-4, 12-1, 5-5, X-1, Mo11-1, Mo19-2, Mo1-2, Mo7-1, Mo1-1, Mo3-2, Mo8-1, Mo2-1, Mo11-2, Mo17-2, Mo13-1, D8S1106 and D4S2408. A set of oligonucleotide primer pairs are provided. Each oligonucleotide primer pair flanks a single locus in the set of STR marker loci, which are co-amplified in a multiplex amplification reaction, with the product(s) comprising a mixture of amplified alleles from each of the co-amplified loci in the set of STR marker loci. The products are evaluated to determine the alleles present at each of the loci analyzed.
Features
These additional 14 mouse STR markers will add resolution between mouse cell lines derived from the same strain, allowing a more accurate STR profile. These additional markers will add confidence to researchers trying to discriminate between same strain samples for cell line authentication.

