NIST 2D NMR ‘Fingerprinting’ Study Gives Biopharmaceutical Sector New Power to Assure High Quality Monoclonal Antibody Therapeutics
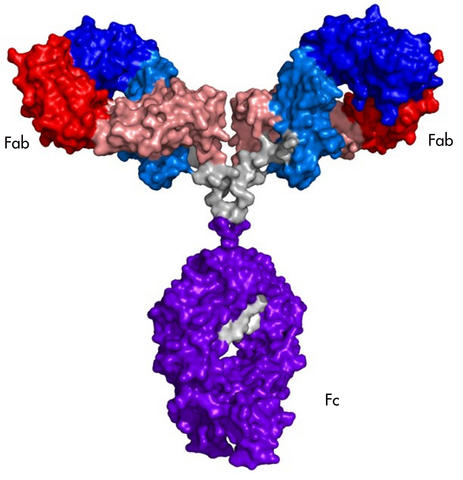
A schematic showing the NISTmAb monoclonal antibody, an immunoglobulin G molecule developed by NIST as a reference material. The labels mark the fragments Fab and Fc that were used in a novel NIST two-dimensional NMR fingerprinting method to measure the structural configuration of the entire antibody.
Researchers at the National Institute of Standards and Technology (NIST) recently completed a large international study* that establishes two-dimensional nuclear magnetic resonance (2D NMR) as a reliable way to compare the three-dimensional structures of monoclonal antibody biopharmaceuticals (mAbs), the largest and fastest growing class of therapeutics.
Because a mAb’s three-dimensional shape, also known as its higher order structure, is linked to its function, it is considered a “critical quality attribute:” a key property used to assess biopharmaceutical product quality. The 2D NMR method for mAbs, originally demonstrated at NIST, gives developers, manufacturers, and regulators a robust way to assess this critical characteristic linked to drug quality and performance.
Biopharmaceuticals—large, complex protein molecules—are used to treat serious and life-threatening conditions such as cancer, autoimmune disorders, infectious diseases, and other illnesses. Global sales of biopharmaceuticals reached nearly $153 billion dollars in 2017. Therapies composed of monoclonal antibodies are the most common type of biopharmaceutical.
Any change in the higher order structure of a protein can prevent it from functioning properly: Biopharmaceutical proteins that deviate from their designed shape will not hit their intended therapeutic targets. This compromises the effectiveness of the therapy, and might lead to an unwanted and potentially dangerous immune response.
The NIST researchers and their international collaborators used 2D NMR to examine the same mAb, the molecule that is the basis for NIST reference material (RM) 8671—widely known as the NISTmAb. Each mAb has a unique 2D NMR spectral fingerprint that directly correlates to its higher order structure. While NIST researchers had previously demonstrated that 2D NMR could precisely measure the NISTmAb’s spectral fingerprint, broad adoption of the new NMR technology requires a demonstration that members of the biopharmaceutical research community can achieve the same, reliable results with a variety of NMR instruments, across different laboratories and operators.
Twenty-five institutions from around the world participated in this largest-ever inter-laboratory study using 2D NMR. There was nearly equal representation from industry, academia, and government institutions, including three regulatory agencies. Countries represented included Australia, Brazil, Canada, Germany, Japan, Slovenia, Sweden, Switzerland and the United States.
According to Carlos Amezcua, a senior research scientist at Baxter Healthcare, the study is a “game changer” for industry. "The results from this study demonstrate that 2D-NMR spectroscopy can be used for the accurate, precise, and robust assessment of the higher order structure in protein therapeutics. This study will surely pave the way for NMR to become a mainstream analytical technique during the physicochemical characterization of biopharmaceuticals and biosimilars.”
Looking forward, robust and precise structural assessment—like that provided by 2D NMR—could help bring generic versions of biopharmaceuticals, known as biosimilars, to the market by providing a universally accepted approach for judging the similarity of protein structures with great precision and confidence. For patients, this could accelerate the speed with which these lifesaving drugs can be approved for clinical use, leading to wider access to mAb-based therapies.
* R.G. Brinson, J.P. Marino, F. Delaglio et al. Enabling adoption of 2D-NMR for the higher order structure assessment of Monoclonal Antibody Therapeutics. mAbs, https://doi.org/10.1080/19420862.2018.1544454

