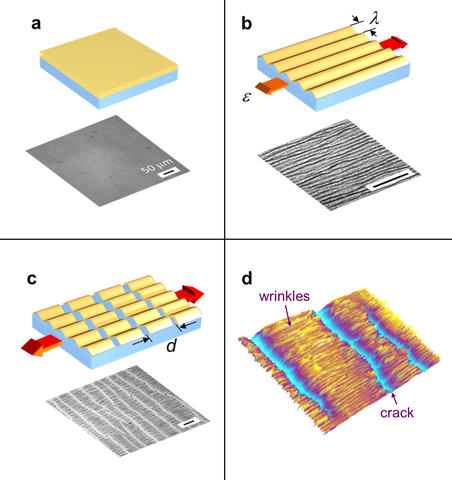
Nanomechanical measurements (model system and microimage of typical specimen). a) thin rigid film on elastic substrate b) initial strain induces surface wrinkles parallel to stress c) additional strain induces regular pattern of cracks in the film d) typical specimen imaged with optical profilometer (280 X 210 micrometers.)
Researchers at the National Institute of Standards and Technology (NIST) have demonstrated a measurement technique that reliably determines three fundamental mechanical properties of near-nanoscale films. The technique, which highlights the challenge of making mechanical measurements on an object with at least one dimension comparable to the size of a virus, should enable better design and engineering for a variety of thin-film technologies, particularly reverse-osmosis membranes for water purification.
Reverse-osmosis membranes, explains NIST researcher Chris Stafford, are an interesting challenge for the materials scientist. The membranes are used in water purification systems—a polyamide film no more than 200 nanometers thick backed by a thicker, porous support layer. Water holding dissolved salts or other contaminants is forced against one side of the membrane at substantial pressures up to about a thousand psi (roughly 7 megapascal), and comes out the other side leaving most of the impurities behind. The mechanical integrity of the membrane is obviously essential—it can't tear or develop pinhole leaks under the pressure—but engineers lacked a good way to measure the strength and breaking point, under stress, of these extremely thin films.
The NIST technique builds on earlier work by the team that demonstrated that you can reliably determine Young's modulus—a measure of stiffness or elasticity—for thin and ultrathin films by bonding it to a piece of silicon rubber, and then carefully stretching it in one direction. The film will develop a regularly spaced pattern of wrinkles (try it with a piece of plastic wrap), and the spacing of the wrinkles, the amount of stretch and some math gives you the modulus. In the new work, they basically pull harder until the film starts developing minute cracks crosswise to the tension. These too, it turns out, occur in regular patterns, and the spacing can be analyzed to determine both the fracture strength and the onset fracture strain, or the failure point, of the film.
Applying their technique to study the effect of chlorine on reverse-osmosis membranes, the team uncovered a puzzle. Chlorine in the water is known to cause a progressive deterioration in membrane performance, generally thought to be the result of prolonged chemical attack by the chlorine. Not so, according to the NIST team. "Chemically the chlorine attack is pretty quick," says Stafford. Spectroscopic chemical analysis showed that all the chemical damage from chlorine exposure happens in the first few hours. Tests using the wrinkle-crack method, however, show that the mechanical properties degrade continuously—the material becoming more and more stiff, brittle and weak—up to the longest duration tested, 10 days. "It may be an aging effect in polymers," says Stafford. "We're continuing to study that to figure out what's going on in there, because it's a real measurement challenge to get in on that length scale to follow the structure over time."
The project is part of a broader NIST program to study materials issues related to sustainable technologies like water purification, but the research team notes that the wrinkle-crack method itself would be broadly applicable to mechanical studies of almost any nanoscale thin film in fields as diverse as artificial skin, flexible electronics, thin-film sensors, fuel cells and photovoltaics.

